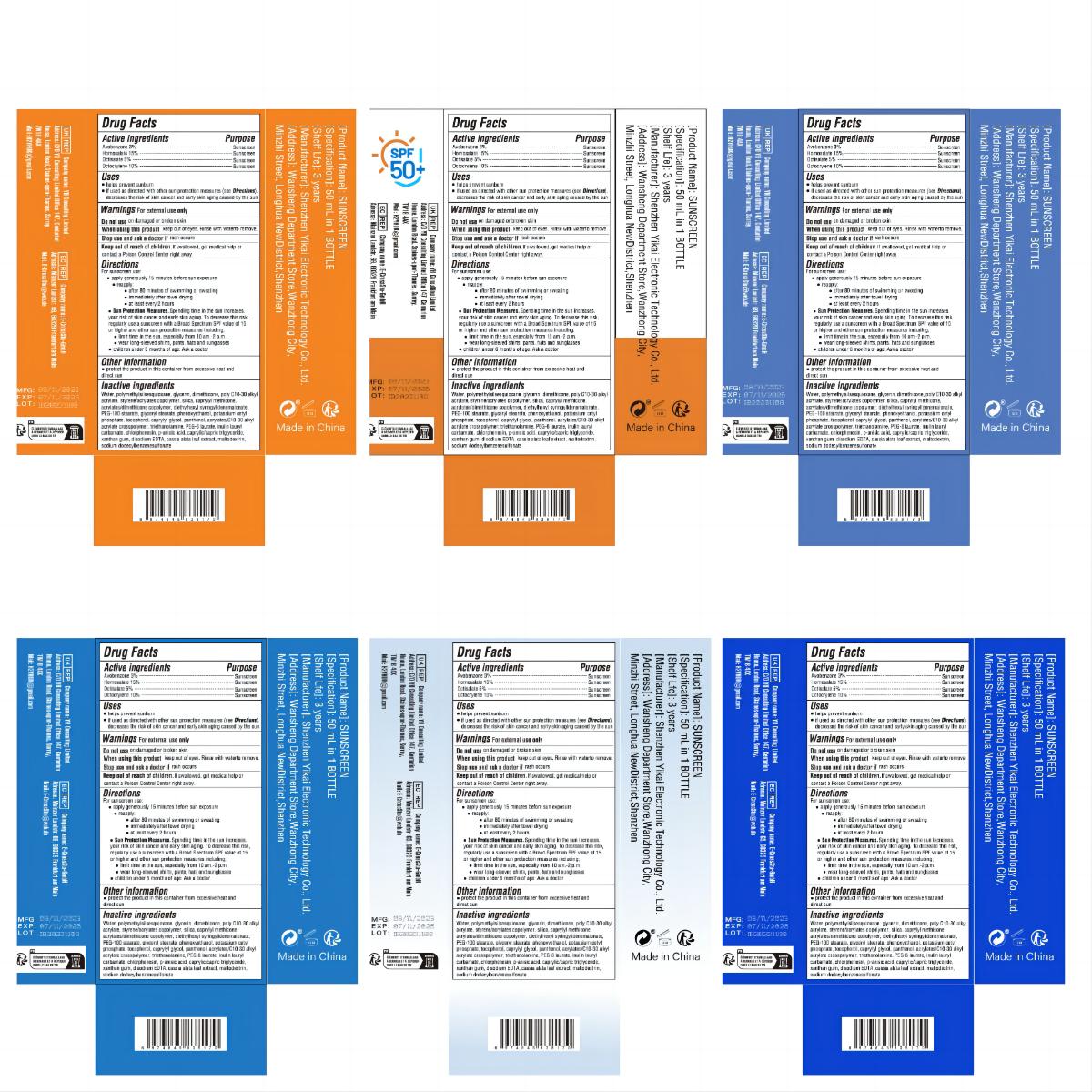
Label: SUNSCREEN cream
- NDC Code(s): 83809-020-01, 83809-020-02
- Packager: Shenzhen Yikai Electronic Technology Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 1, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
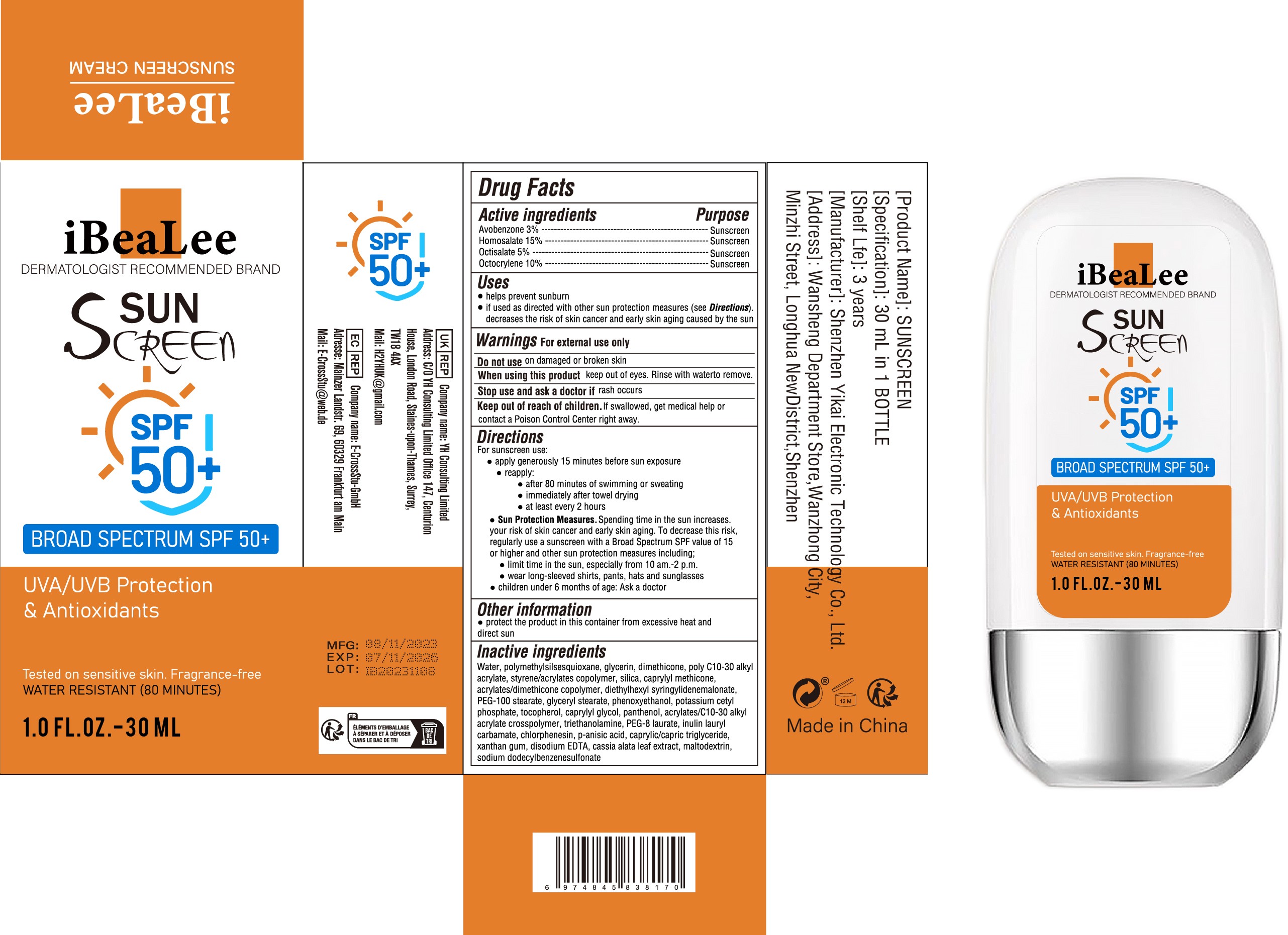
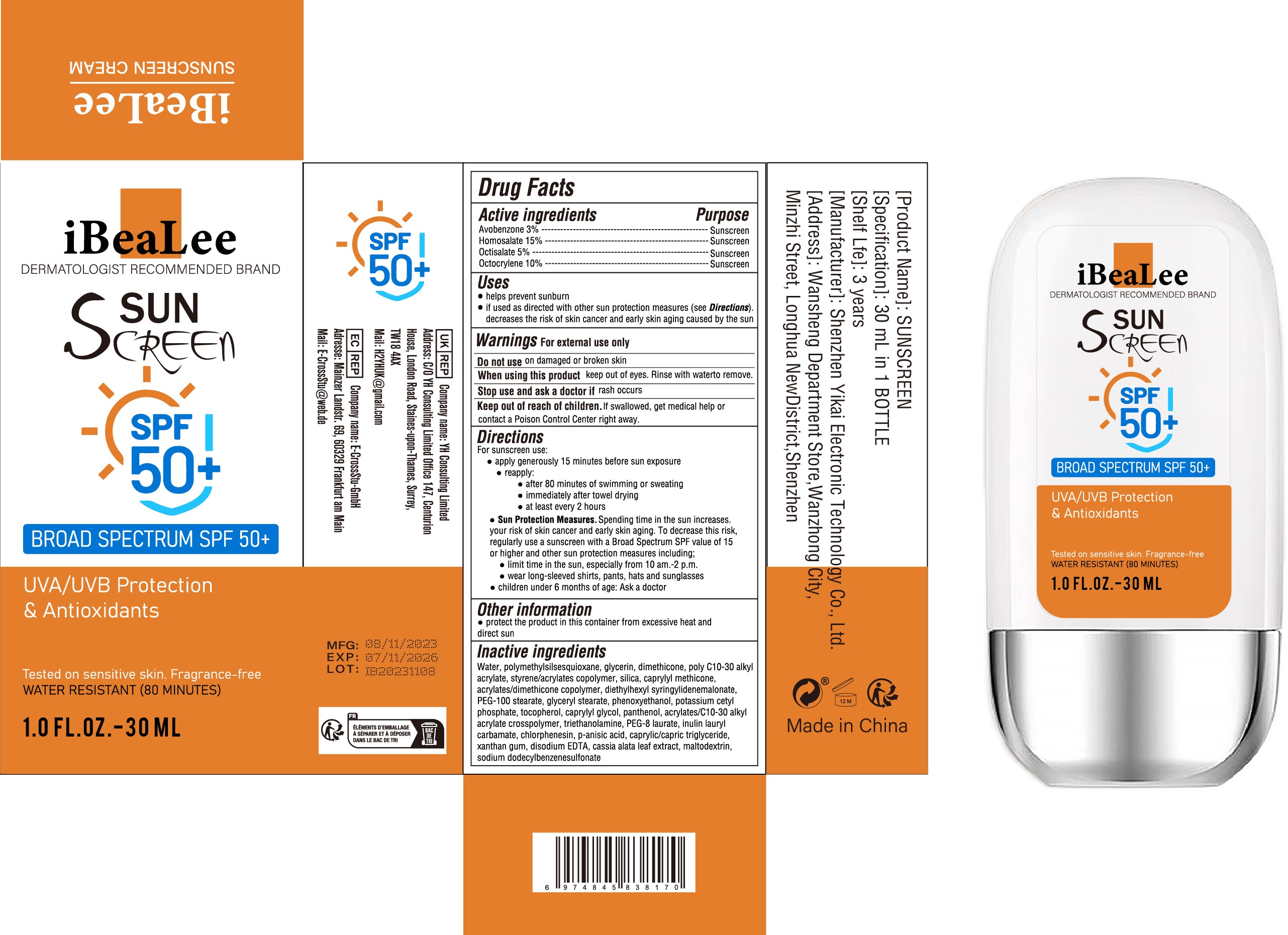
- Active Ingredient
- Purpose
- Use
- Warnings
- Do not use
- When Using
- Stop Use
- Keep Oot Of Reach Of Children
-
Directions
For sunscreen use:
● apply generously 15 minutes before sun exposure
● reapply:
● after 80 minutes of swimming or sweating
● immediately after towel drying
● at least every 2 hours
● Sun Protection Measures. Spending time in the sun increases. your risk of skin cancer and early skin aging. To decrease this risk,
regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including;
● limit time in the sun, especially from 10 am.-2 p.m.
● wear long-sleeved shirts, pants, hats and sunglasses
● children under 6 months of age: Ask a doctor - Other information
-
Inactive ingredients
Water, polymethylsilsesquioxane, glycerin, dimethicone, poly C10-30 alkyl
acrylate, styrene/acrylates copolymer, silica, caprylyl methicone,
acrylates/dimethicone copolymer, diethylhexyl syringylidenemalonate,
PEG-100 stearate, glyceryl stearate, phenoxyethanol, potassium cetyl
phosphate, tocopherol, caprylyl glycol, panthenol, acrylates/C10-30 alkyl
acrylate crosspolymer, triethanolamine, PEG-8 laurate, inulin lauryl
carbamate, chlorphenesin, p-anisic acid, caprylic/capric triglyceride,
xanthan gum, disodium EDTA, cassia alata leaf extract, maltodextrin,
sodium dodecylbenzenesulfonate - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SUNSCREEN
sunscreen creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83809-020 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5 g in 100 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 15 g in 100 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 100 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 10 g in 100 mL Inactive Ingredients Ingredient Name Strength GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) 2-ETHYLHEXYL ACRYLATE, METHACRYLATE, METHYL METHACRYLATE, OR BUTYL METHACRYLATE/HYDROXYPROPYL DIMETHICONE COPOLYMER (30000-300000 MW) (UNII: S7ZA3CCJ4M) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) BEHENYL ACRYLATE POLYMER (UNII: D64PM5UT4U) POLYOXYL 100 STEARATE (UNII: YD01N1999R) TOCOPHEROL (UNII: R0ZB2556P8) TROLAMINE (UNII: 9O3K93S3TK) SENNA ALATA LEAF (UNII: 4BXR6YZN92) GLYCERIN (UNII: PDC6A3C0OX) DIMETHICONE (UNII: 92RU3N3Y1O) ACRYLIC ACID/2-ETHYLHEXYL ACRYLATE/STYRENE COPOLYMER (500000 MW) (UNII: 3K8D4YRM4R) ACRYLATES/C10-30 ALKYL ACRYLATE CROSSPOLYMER (60000 MPA.S) (UNII: 8Z5ZAL5H3V) XANTHAN GUM (UNII: TTV12P4NEE) WATER (UNII: 059QF0KO0R) POTASSIUM CETYL PHOSPHATE (UNII: 03KCY6P7UT) PANTHENOL (UNII: WV9CM0O67Z) PEG-8 LAURATE (UNII: 762O8IWA10) INULIN LAURYL CARBAMATE (UNII: 48RFF58ESG) P-ANISIC ACID (UNII: 4SB6Y7DMM3) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) MALTODEXTRIN (UNII: 7CVR7L4A2D) SODIUM DODECYLBENZENESULFONATE (UNII: 554127163Y) POLYMETHYLSILSESQUIOXANE (4.5 MICRONS) (UNII: 59Z907ZB69) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CAPRYLYL TRISILOXANE (UNII: Q95M2P1KJL) PHENOXYETHANOL (UNII: HIE492ZZ3T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83809-020-01 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2024 2 NDC:83809-020-02 50 mL in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 02/01/2024 Labeler - Shenzhen Yikai Electronic Technology Co., Ltd. (700426808) Establishment Name Address ID/FEI Business Operations Shenzhen Yikai Electronic Technology Co., Ltd. 700426808 manufacture(83809-020) , label(83809-020)