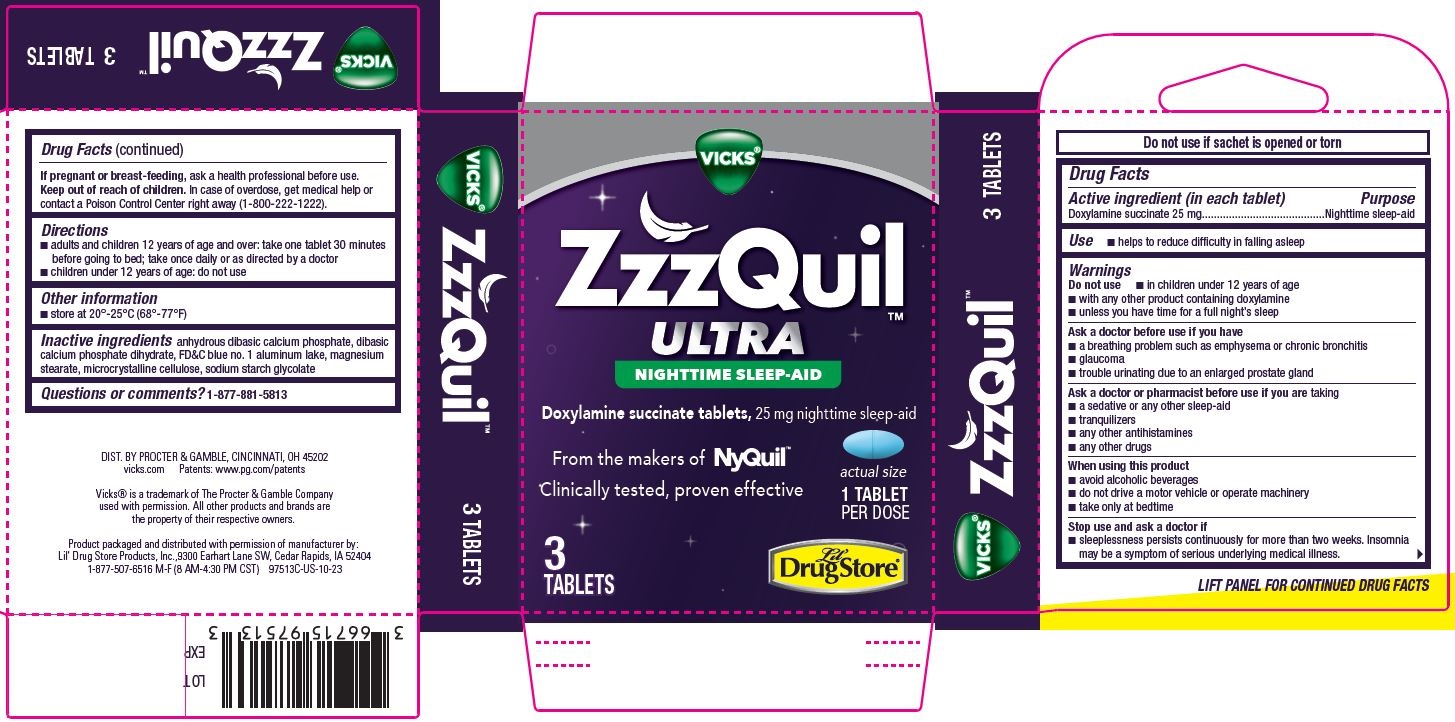
Label: ZZZQUIL ULTRA, LIL DRUG STORE- doxylamine succinate tablet
- NDC Code(s): 66715-9751-3
- Packager: Lil' Drug Store Products, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 22, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Use
-
Warnings
Warnings
Do not use
Do not use
- in children under 12 years of age
- with any other product containing doxylamine
- unless you have time for a full night’s sleep
Ask a doctor
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Ask a doctor or pharmacist before use if you are taking
Ask a doctor or pharmacist before use if you are taking
- a sedative or any other sleep-aid
- tranquilizers
- any other antihistamines
- any other drugs
When using this product
When using this product
- avoid alcoholic beverages
- do not drive a motor vehicle or operate machinery
- take only at bedtime
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Distributor information
DIST. BY PROCTER & GAMBLE, CINCINNATI, OH 45202
vicks.com Patents: www.pg.com/patents
Vicks ® is a trademark of The Procter & Gamble Company
used with permission. All other products and brands are
the property of their respective owners.
Product packaged and distributed with permission of manufacturer by:
Lil' Drug Store Products, Inc.,9300 Earhart Lane SW, Cedar Rapids, IA 52404
1-877-507-6516 M-F (8 AM-4:30 PM CST) - ZzzQuil TM ULTRA, Lil' Drug Store ® - PDP/Package
-
INGREDIENTS AND APPEARANCE
ZZZQUIL ULTRA, LIL DRUG STORE
doxylamine succinate tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:66715-9751 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOXYLAMINE SUCCINATE (UNII: V9BI9B5YI2) (DOXYLAMINE - UNII:95QB77JKPL) DOXYLAMINE SUCCINATE 25 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) FD&C BLUE NO. 1 ALUMINUM LAKE (UNII: J9EQA3S2JM) MAGNESIUM STEARATE (UNII: 70097M6I30) SODIUM STARCH GLYCOLATE TYPE A (UNII: H8AV0SQX4D) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) Product Characteristics Color blue Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code L441 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66715-9751-3 3 in 1 CARTON 01/26/2024 1 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M010 01/26/2024 Labeler - Lil' Drug Store Products, Inc. (093103646)