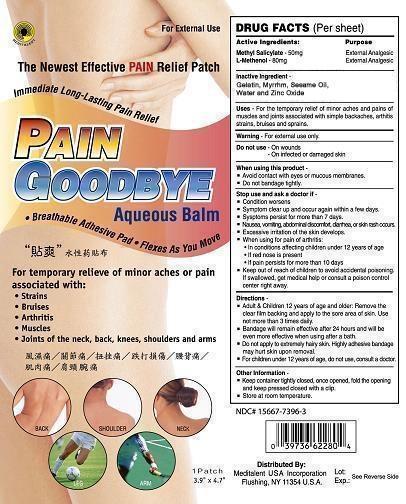
Label: PAIN GOODBYE AQUEOUS PATCH- methyl salicylate patch
-
Contains inactivated NDC Code(s)
NDC Code(s): 15667-7396-3, 15667-7396-5 - Packager: Austin Biotechnology Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 10, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS
- PURPOSE
- USES
- WARNINGS
- Stop use and ask a doctor if
- Stop use and ask a doctor if
- Stop use and ask a doctor if
- WARNINGS
-
Directions
Adult and children 12 years of age and older; Remove the clear film backing and apply to the sore area of skin. Use not than 3 times daily.
Bandage will remain effective after 24 hours and will be even more effective when using after a bath.
Do not apply to extremely hairy skin. Highly adhesive bandage may hurt skin upon removal.
Other Information:
Keep container tightly closed. Once opened, fold the opening and keep pressed. Close with a clip.
Store at room temperature.
- INACTIVE INGREDIENTS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PAIN GOODBYE AQUEOUS PATCH
methyl salicylate patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:15667-7396 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYL SALICYLATE (UNII: LAV5U5022Y) (SALICYLIC ACID - UNII:O414PZ4LPZ) METHYL SALICYLATE 50 mg in 1000 mg 2-(L-MENTHOXY)ETHANOL (UNII: 5ZWW23169H) (2-(L-MENTHOXY)ETHANOL - UNII:5ZWW23169H) 2-(L-MENTHOXY)ETHANOL 80 mg in 1000 mg Inactive Ingredients Ingredient Name Strength GELATIN (UNII: 2G86QN327L) MYRRH (UNII: JC71GJ1F3L) SESAME OIL (UNII: QX10HYY4QV) WATER (UNII: 059QF0KO0R) ZINC OXIDE (UNII: SOI2LOH54Z) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:15667-7396-5 5 in 1 BAG 1 NDC:15667-7396-3 1000 mg in 1 BAG Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 10/26/2011 Labeler - Austin Biotechnology Inc (656001949) Registrant - Austin Biotechnology Inc (656001949) Establishment Name Address ID/FEI Business Operations Austin Biotechnology Inc 656001949 manufacture(15667-7396)