Label: CETIRIZINE HYDROCHLORIDE (ALLERGY)- cetirizine hydrochloride capsule
- NDC Code(s): 58602-806-04, 58602-806-12, 58602-806-16, 58602-806-53
- Packager: Aurohealth LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated September 14, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
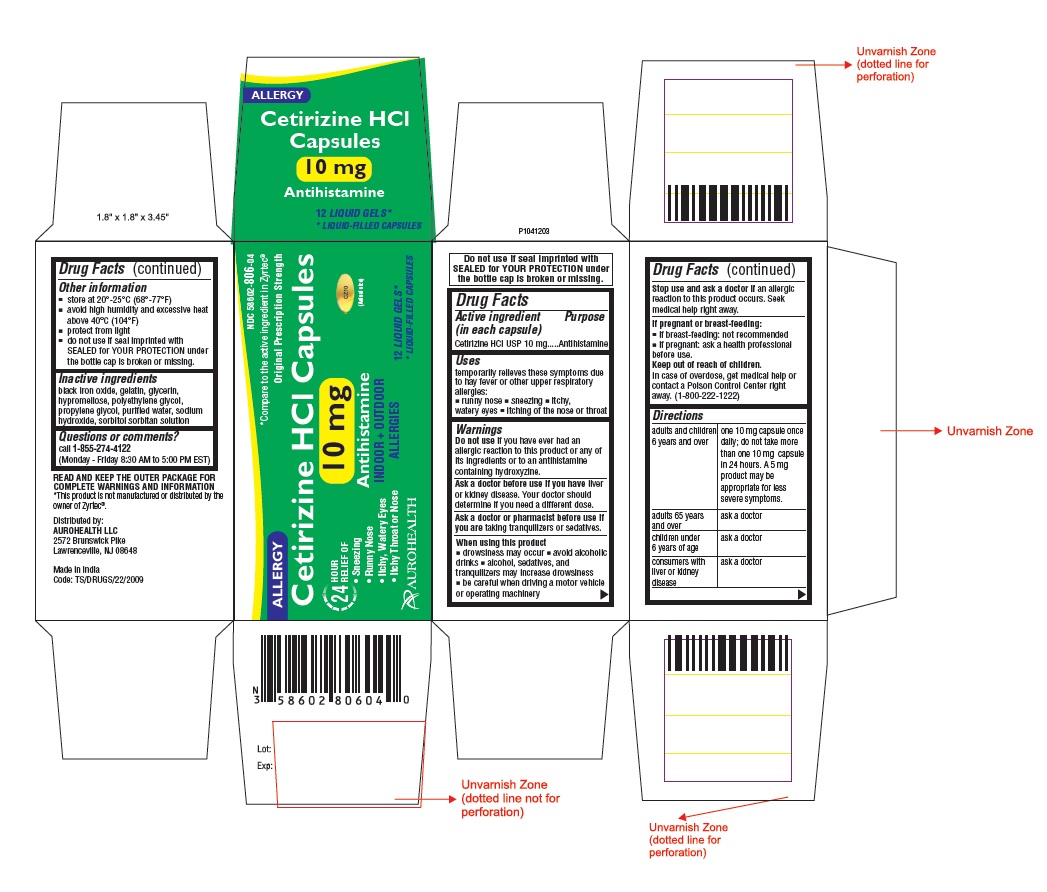
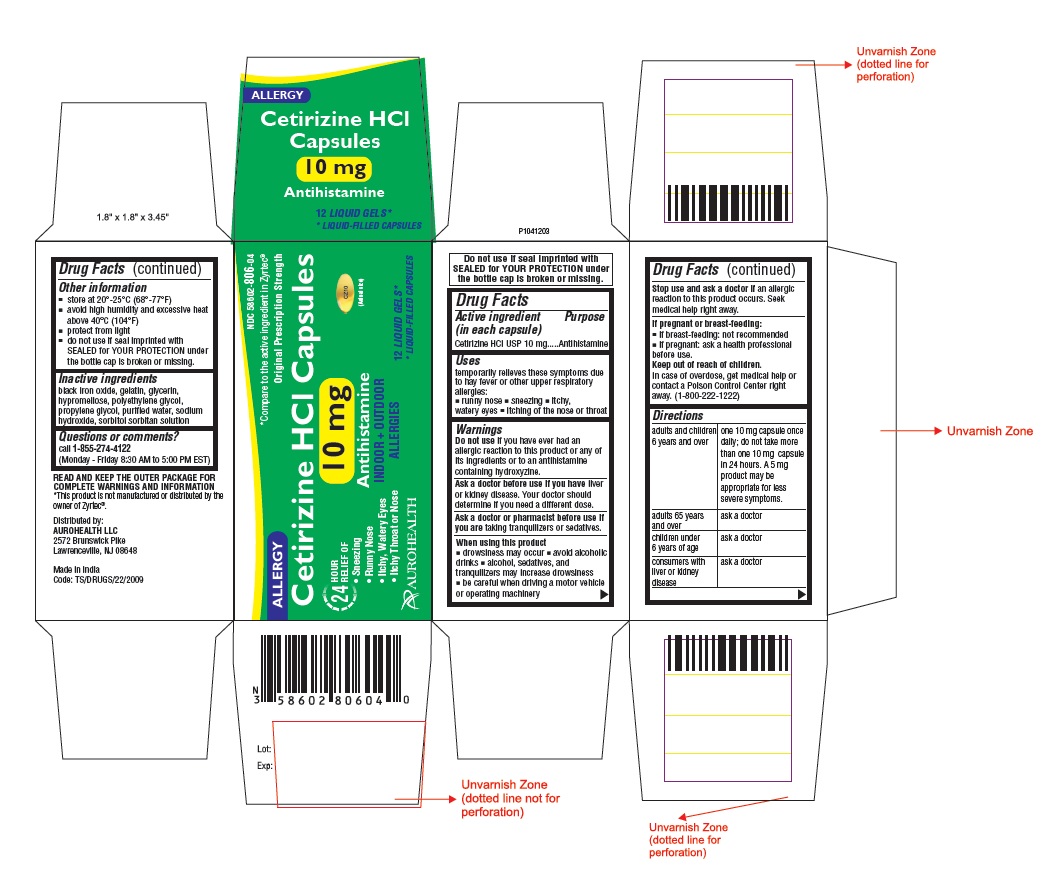
- Active ingredient (in each capsule)
- Purpose
- Uses
- Warnings
- ASK DOCTOR
- ASK DOCTOR/PHARMACIST
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding:
- Keep out of reach of children.
-
Directions
adults and children
6 years and over
one 10 mg capsule once daily;
do not take more than one 10 mg
capsule in 24 hours. A 5 mg
product may be appropriate for
less severe symptoms.
adults 65 years and over
ask a doctor
children under 6 years of age
ask a doctor
consumers with liver or
kidney disease
ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
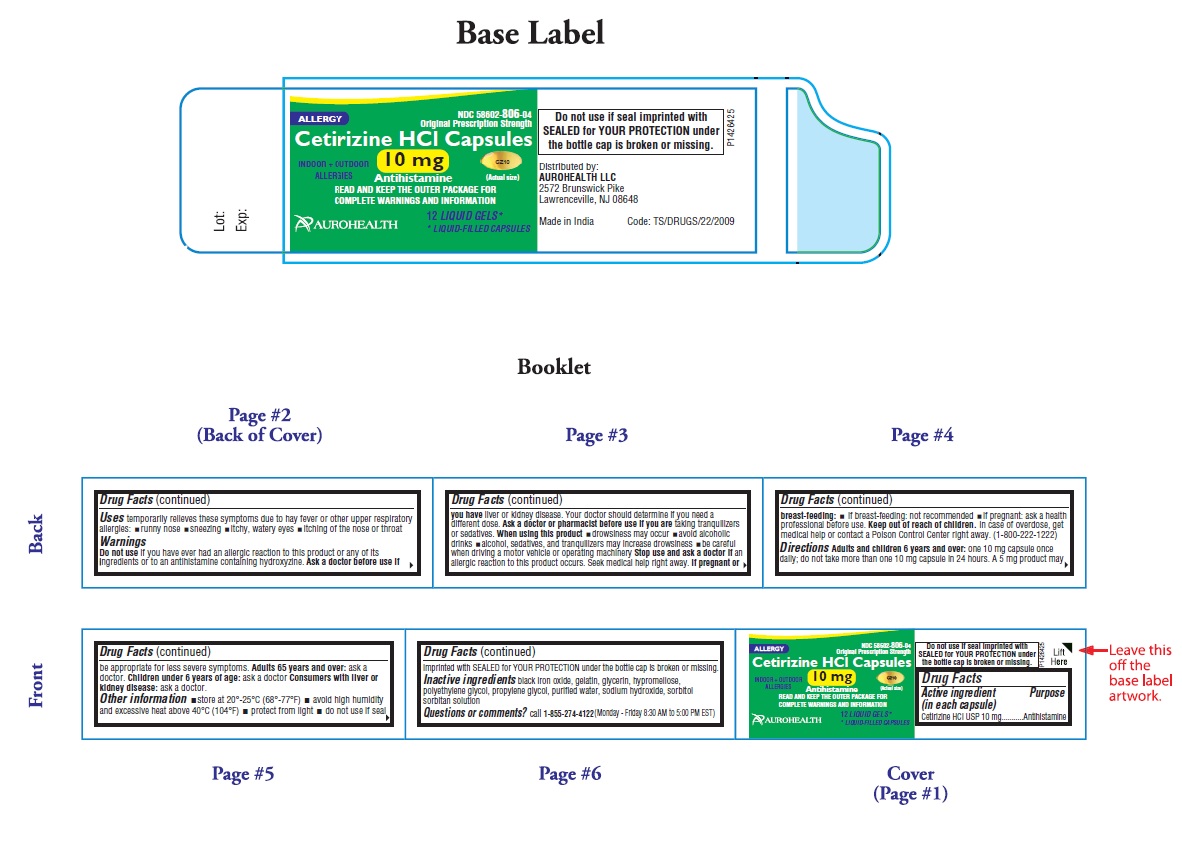
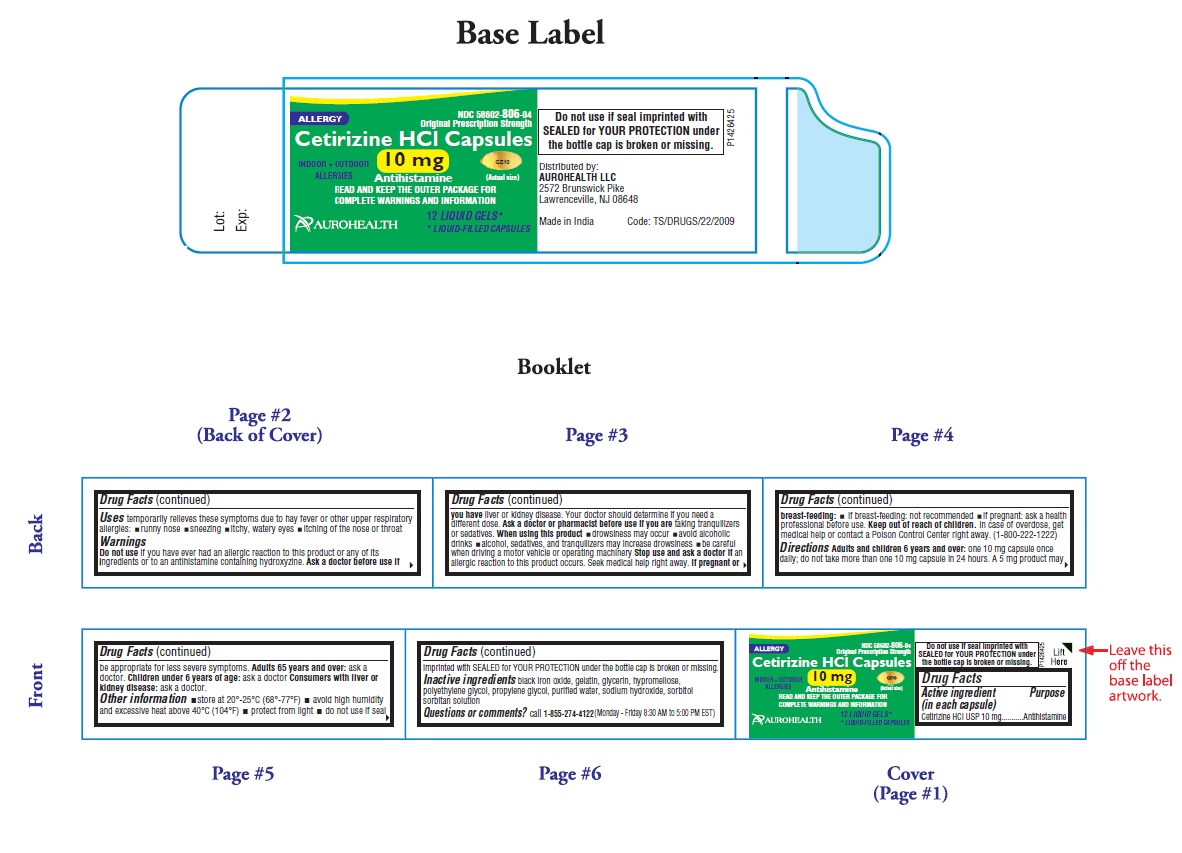
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 10 mg (12's Capsule Container Label)
-
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL -10 mg (12's Capsule Container Carton Label)
NDC 58602-806-04
*Compare to the active ingredient in Zyrtec®
Original Prescription Strength
ALLERGY
Cetirizine HCl Capsules
10 mg
Antihistamine
INDOOR +OUTDOOR CZ10
ALLERGIES (Actual Size)
24 HOUR
RELIEF OF- Sneezing
- Runny Nose
- Itchy, Watery Eyes
- Itchy Throat or Nose
12 LIQUID GELS*
* LIQUID-FILLED CAPSULES
AUROHEALTH
-
INGREDIENTS AND APPEARANCE
CETIRIZINE HYDROCHLORIDE (ALLERGY)
cetirizine hydrochloride capsuleProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58602-806 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CETIRIZINE HYDROCHLORIDE (UNII: 64O047KTOA) (CETIRIZINE - UNII:YO7261ME24) CETIRIZINE HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) SORBITOL (UNII: 506T60A25R) Product Characteristics Color YELLOW (Clear colourless to pale yellow viscous liquid) Score no score Shape OVAL Size 13mm Flavor Imprint Code CZ10 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58602-806-04 1 in 1 CARTON 07/20/2018 1 12 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:58602-806-53 1 in 1 CARTON 07/20/2018 2 25 in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:58602-806-12 1 in 1 CARTON 07/20/2018 3 40 in 1 BOTTLE; Type 0: Not a Combination Product 4 NDC:58602-806-16 65 in 1 CARTON 07/20/2018 4 65 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209107 07/20/2018 Labeler - Aurohealth LLC (078728447) Establishment Name Address ID/FEI Business Operations APL HEALTHCARE LIMITED 650844777 ANALYSIS(58602-806) , MANUFACTURE(58602-806) Establishment Name Address ID/FEI Business Operations Aurobindo Pharma Limited 650381903 ANALYSIS(58602-806) , MANUFACTURE(58602-806)