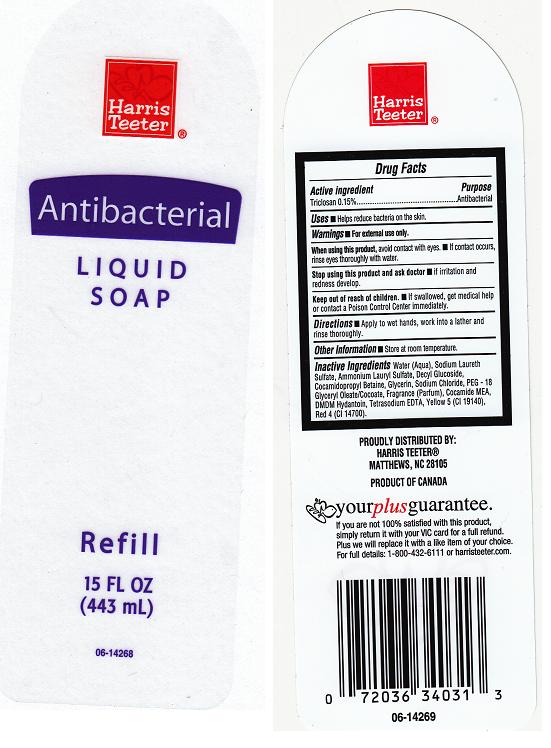
Label: HARRIS TEETER ANTIBACTERIAL- triclosan soap
-
Contains inactivated NDC Code(s)
NDC Code(s): 72036-220-15 - Packager: HARRIS TEETER
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 7, 2010
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Warnings
- Directions
- Uses
- Inactive Ingredients
- Package Front and Back Labels
-
INGREDIENTS AND APPEARANCE
HARRIS TEETER ANTIBACTERIAL
triclosan soapProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72036-220 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRICLOSAN (UNII: 4NM5039Y5X) (TRICLOSAN - UNII:4NM5039Y5X) TRICLOSAN 0.15 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SODIUM LAURETH SULFATE (UNII: BPV390UAP0) AMMONIUM LAURYL SULFATE (UNII: Q7AO2R1M0B) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) GLYCERIN (UNII: PDC6A3C0OX) SODIUM CHLORIDE (UNII: 451W47IQ8X) COCO MONOETHANOLAMIDE (UNII: C80684146D) DMDM HYDANTOIN (UNII: BYR0546TOW) EDETATE SODIUM (UNII: MP1J8420LU) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) FD&C RED NO. 4 (UNII: X3W0AM1JLX) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72036-220-15 443 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 10/07/2010 Labeler - HARRIS TEETER (047279351)