Label: WONDERBELLY MAXIMUM STRENGTH ANTACID- calcium carbonate tablet, chewable
- NDC Code(s): 82431-105-03, 82431-105-12, 82431-105-60
- Packager: Ginger Health Company
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 28, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Ask a doctor or pharmacist before use if you are presently taking a prescription drug. Antacids may interact with certain prescription drugs.
When using this product
do not take more than 7 tablets in 24 hours
do not use the maximum dosage for more than 2 weeks, except under the advice and supervision of a doctor
if pregnant or breast-feeding, ask a health professional before use - KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
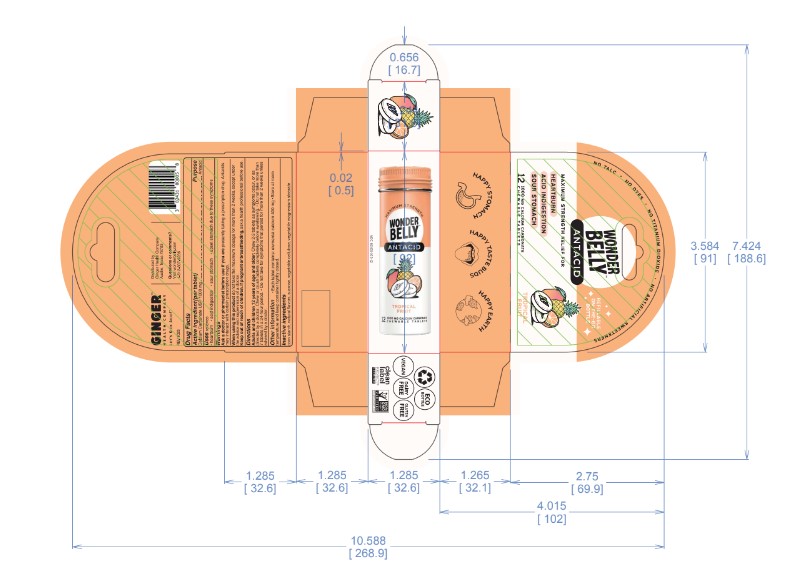
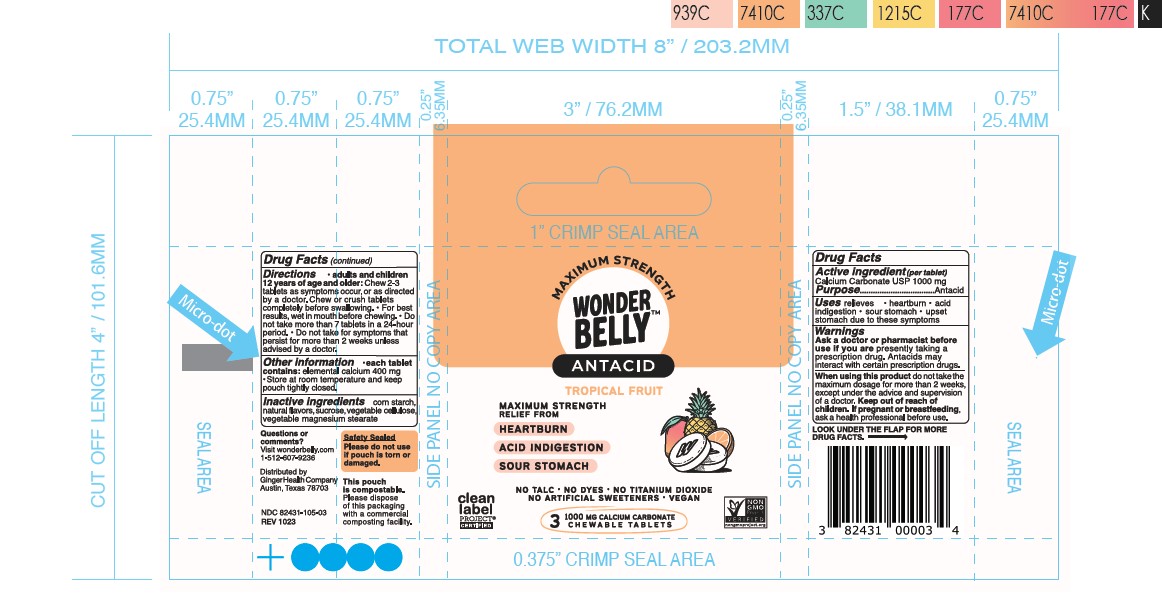
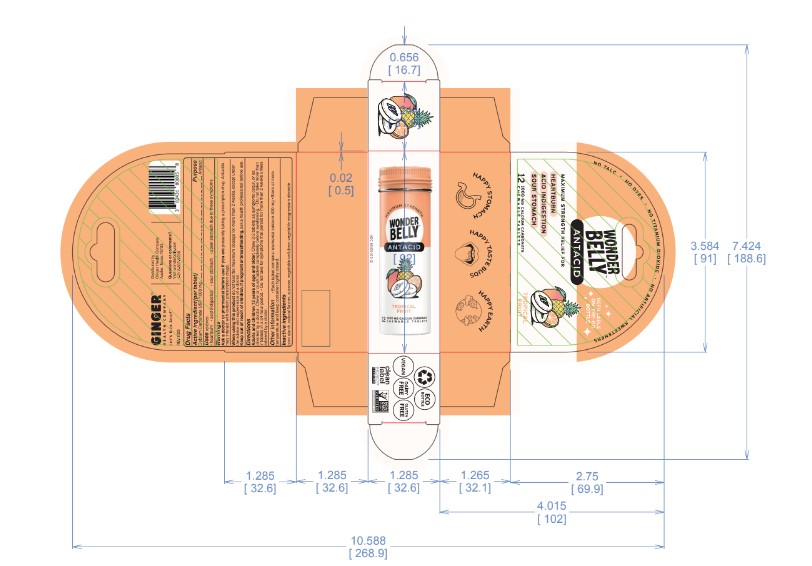
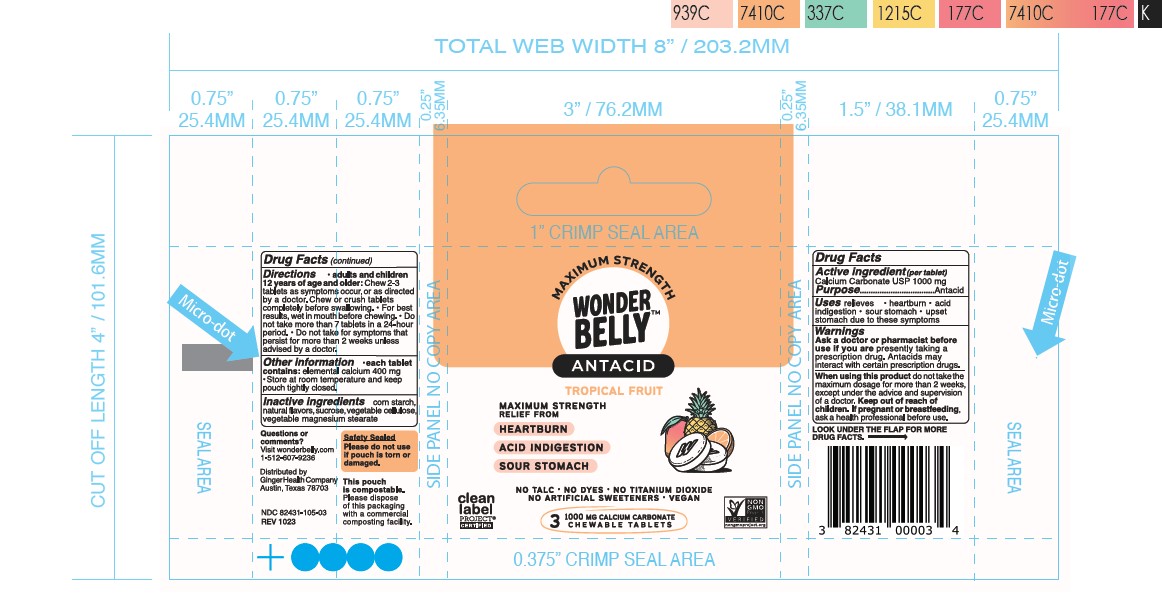
- Principal Display Panel-60ct rev 0523 Principal Display Panel-12ct rev 1123 Principal Display Panel-3ct V2 Rev 1023 Principal Display Panel-12ct UC rev 1023 Principal Display Panel-60ct rev 1023
-
INGREDIENTS AND APPEARANCE
WONDERBELLY MAXIMUM STRENGTH ANTACID
calcium carbonate tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82431-105 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB, CARBONATE ION - UNII:7UJQ5OPE7D) CALCIUM CARBONATE 1000 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) MAGNESIUM STEARATE (UNII: 70097M6I30) SUCROSE (UNII: C151H8M554) Product Characteristics Color white Score no score Shape ROUND Size 19mm Flavor TROPICAL FRUIT PUNCH Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82431-105-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 06/30/2023 2 NDC:82431-105-12 12 in 1 BOTTLE; Type 0: Not a Combination Product 09/01/2023 3 NDC:82431-105-03 3 in 1 POUCH; Type 0: Not a Combination Product 09/01/2023 
Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M001 06/30/2023 Labeler - Ginger Health Company (118431122)