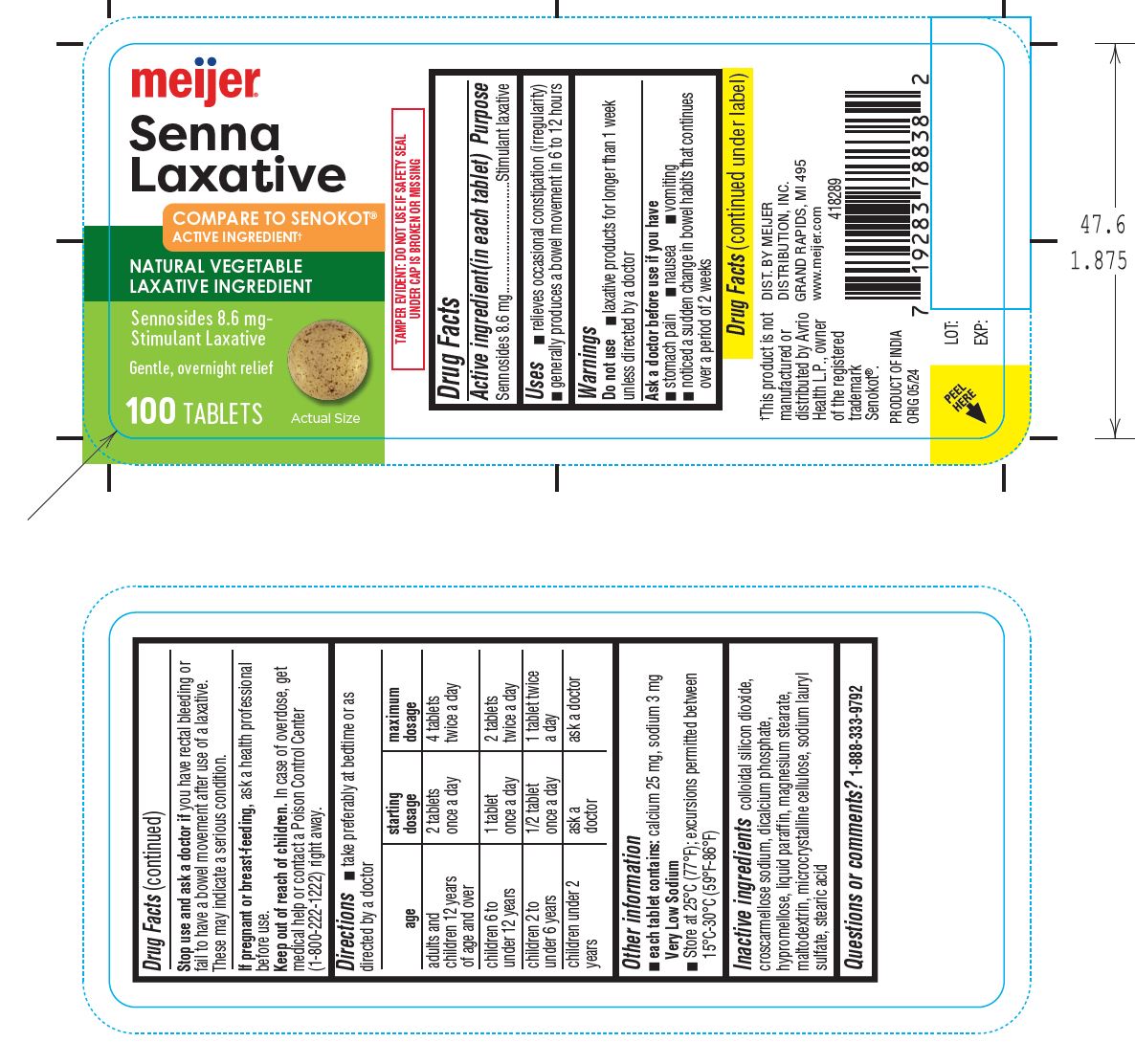
Label: SENNA LAXATIVE- sennosides tablet, film coated
- NDC Code(s): 79481-0059-1
- Packager: Meijer, Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated June 17, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient(in each tablet) Sennosides 8.6 mg
- Purpose Stimulant laxative
- Uses ■ relieves occasional constipation (irregularity) ■ generally produces a bowel movement in 6 to 12 hours
- Warnings
- Do not use■ laxative products for longer than 1 week unless directed by a doctor
- Ask a doctor before use if you have ■ stomach pain ■ nausea ■ vomiting ■ noticed a sudden change in bowel habits that continues over a period of 2 weeks
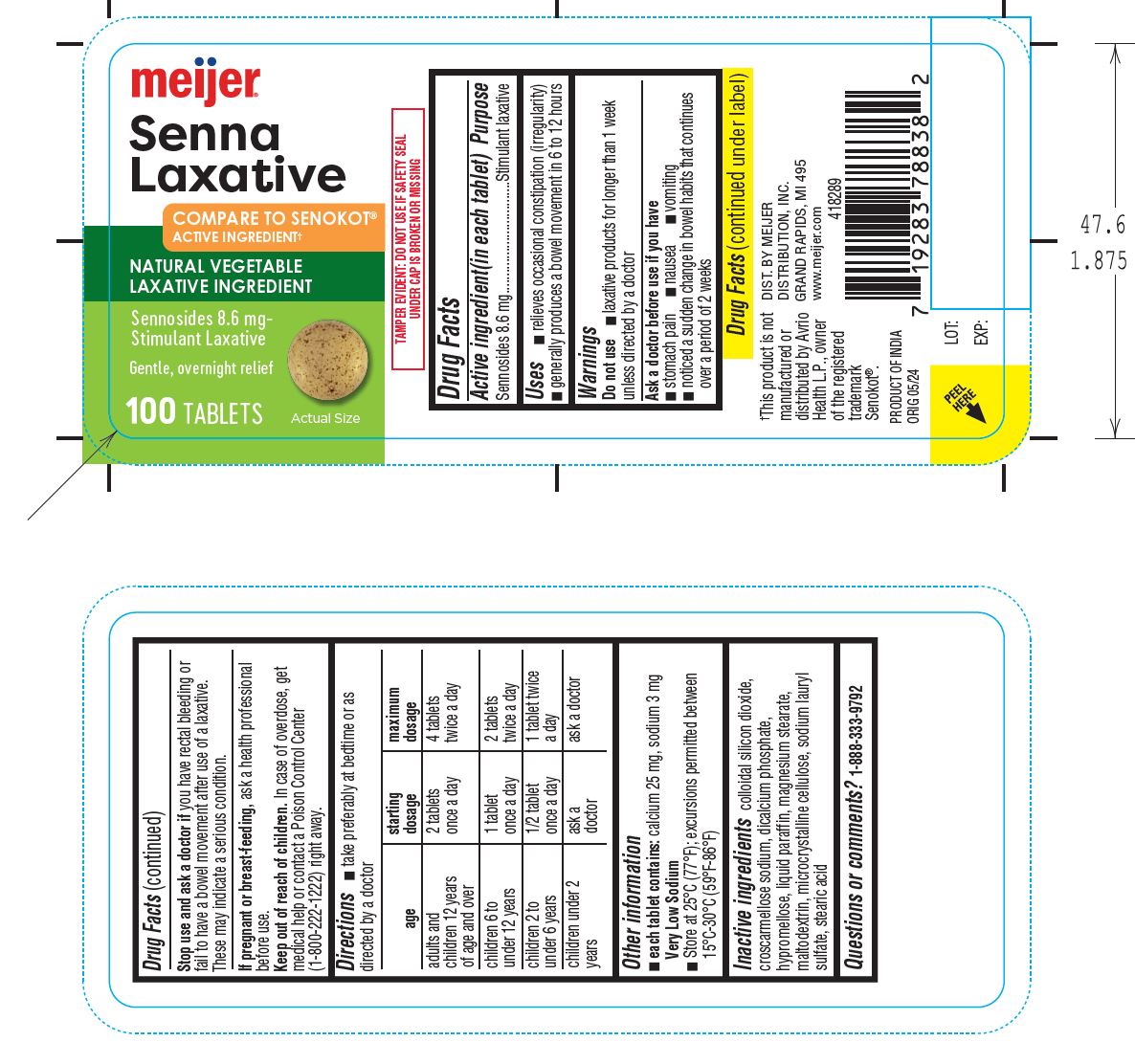
- Stop use and ask a doctor if you have rectal bleeding or fail to have a bowel movement after use of a laxative. These may indicate a serious condition.
- If pregnant or breast-feeding,ask a health professional before use.
- Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
- Directions■ take preferably at bedtime or as directed by a doctor age starting dosage maximum dosage adults and 2 tablets once a day 4 tablets twice a day children 12 years of age and over children 6 to 1 tablet once a day 2 tablets twice a day under 12 years children 2 to 1/2 tablet once a day 1 tablet twice a day under 6 years children under 2 ask a doctor ask a doctor years
- Other information ■ each tablet contains:calcium 25 mg, sodium 3 mg Very Low Sodium ■ Store at 25°C (77°F); excursions permitted between 15°C-30°C (59°F-86°F)
- Inactive ingredientscolloidal silicon dioxide, croscarmellose sodium, dicalcium phosphate, hypromellose, liquid paraffin, magnesium stearate, maltodextrin, microcrystalline cellulose, sodium lauryl sulfate, stearic acid
- Questions or comments? 1-888-333-9792
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SENNA LAXATIVE
sennosides tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:79481-0059 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SENNOSIDES (UNII: 3FYP5M0IJX) (SENNOSIDES - UNII:3FYP5M0IJX) SENNOSIDES 8.6 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CALCIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: L11K75P92J) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MINERAL OIL (UNII: T5L8T28FGP) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SODIUM LAURYL SULFATE (UNII: 368GB5141J) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color brown Score no score Shape ROUND Size 9mm Flavor Imprint Code S8 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:79481-0059-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 06/14/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M007 06/14/2024 Labeler - Meijer, Inc. (006959555)