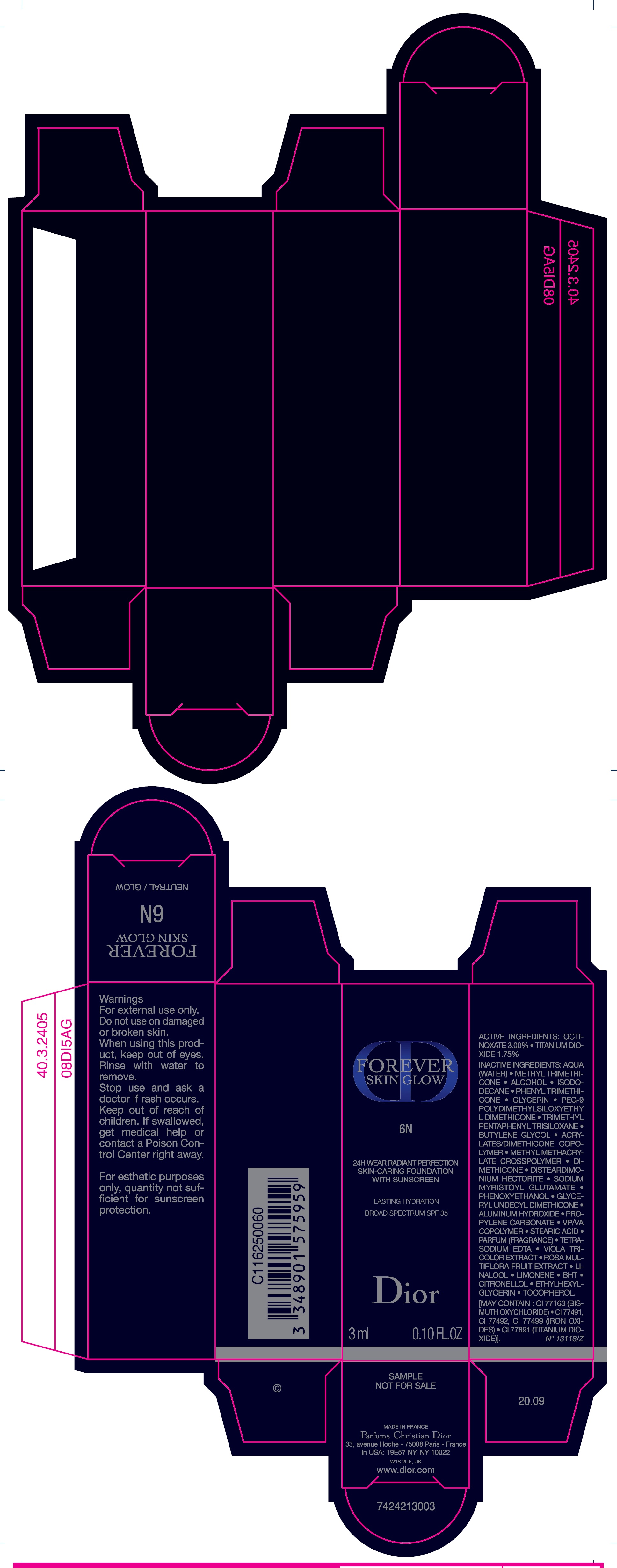
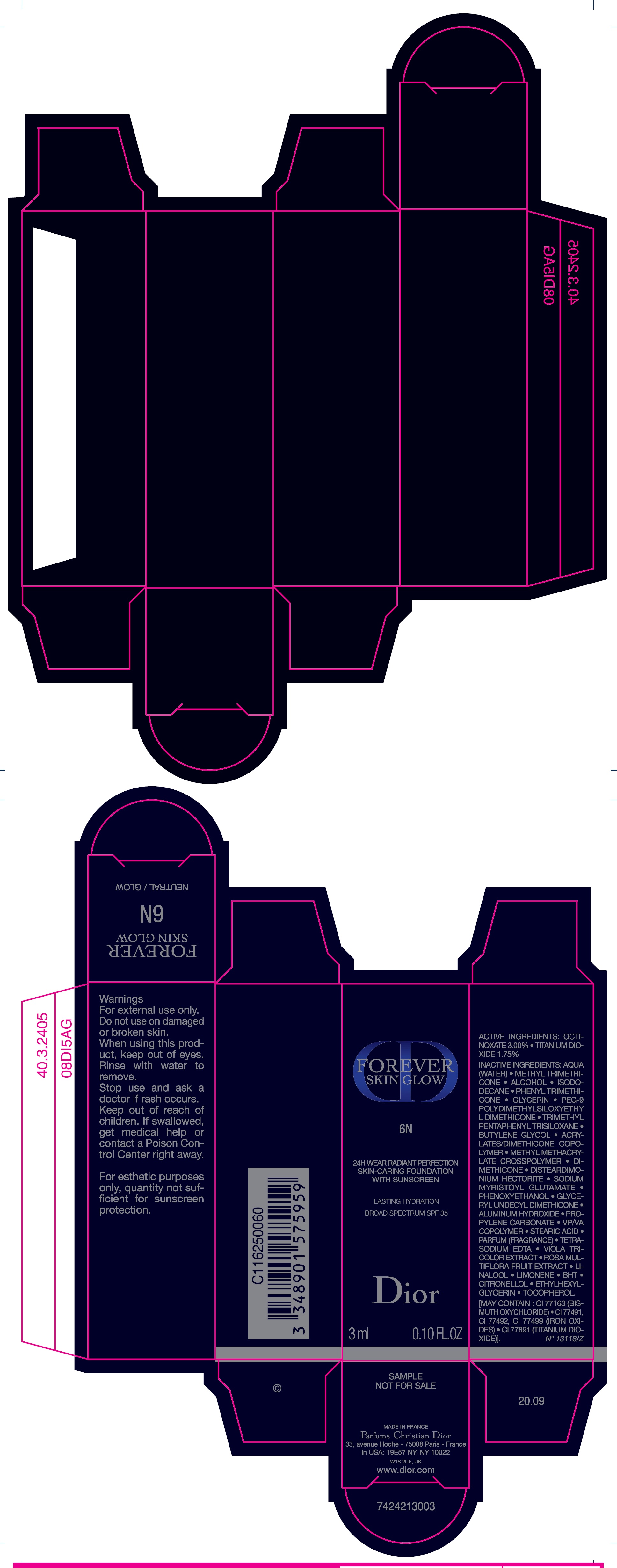
Label: FOREVER SKIN GLOW 24H WEAR RADIANT PERFECTION SKIN CARING FOUNDATION WITH SUNSCREEN BROAD SPECTRUM SPF 35 LASTING HYDRATION 6N- octinoxate, titanium dioxide emulsion
- NDC Code(s): 61957-2937-0, 61957-2937-1
- Packager: PARFUMS CHRISTIAN DIOR
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 3, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredients
- Uses
- Warnings
-
Directions
- Apply liberally 15 minutes before sun exposure.
- Reapply at least every 2 hours.
- Use a water-resistant sunscreen if swimming or sweating.
- Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including: Sun Protection Measures.
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses.
- Children under 6 months of age: ask a doctor.
- Other information
-
Inactive ingredients
AQUA (WATER), METHYL TRIMETHICONE, ALCOHOL, ISODODECANE, PHENYL TRIMETHICONE, GLYCERIN, PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE, TRIMETHYL PENTAPHENYL TRISILOXANE, BUTYLENE GLYCOL, ACRYLATES/DIMETHICONE COPOLYMER, METHYL METHACRYLATE CROSSPOLYMER, DIMETHICONE, DISTEARDIMONIUM HECTORITE, SODIUM MYRISTOYL GLUTAMATE, PHENOXYETHANOL, GLYCERYL UNDECYL DIMETHICONE, ALUMINUM HYDROXIDE, PROPYLENE CARBONATE, VP/VA COPOLYMER, STEARIC ACID, PARFUM (FRAGRANCE), TETRASODIUM EDTA, VIOLA TRICOLOR EXTRACT, ROSA MULTIFLORA FRUIT EXTRACT, LINALOOL, LIMONENE, BHT, CITRONELLOL, ETHYL HEXYGLYCERIN, TOCOPHEROL. [MAY CONTAIN : CI 77163 (BISMUTH OXYCHLORIDE), CI 77491, CI 77492, CI 77499 (IRON OXIDES), CI 77891 (TITANIUM DIOXIDE)].
- Package Labeling:
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
FOREVER SKIN GLOW 24H WEAR RADIANT PERFECTION SKIN CARING FOUNDATION WITH SUNSCREEN BROAD SPECTRUM SPF 35 LASTING HYDRATION 6N
octinoxate, titanium dioxide emulsionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:61957-2937 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 30 mg in 1 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 17.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) METHYL TRIMETHICONE (UNII: S73ZQI0GXM) ALCOHOL (UNII: 3K9958V90M) ISODODECANE (UNII: A8289P68Y2) PHENYL TRIMETHICONE (UNII: DR0K5NOJ4R) GLYCERIN (UNII: PDC6A3C0OX) PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE (UNII: TYP81E471F) TRIMETHYL PENTAPHENYL TRISILOXANE (UNII: HE7ABK56N5) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) METHYL METHACRYLATE/GLYCOL DIMETHACRYLATE CROSSPOLYMER (UNII: EG97988M5Q) DIMETHICONE (UNII: 92RU3N3Y1O) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) SODIUM MYRISTOYL GLUTAMATE (UNII: AYU7QD893W) PHENOXYETHANOL (UNII: HIE492ZZ3T) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) PROPYLENE CARBONATE (UNII: 8D08K3S51E) STEARIC ACID (UNII: 4ELV7Z65AP) EDETATE SODIUM (UNII: MP1J8420LU) LINALOOL, (+/-)- (UNII: D81QY6I88E) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) TOCOPHEROL (UNII: R0ZB2556P8) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61957-2937-0 1 in 1 CARTON 01/01/2019 1 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:61957-2937-1 3 mL in 1 TUBE; Type 0: Not a Combination Product 01/01/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2019 Labeler - PARFUMS CHRISTIAN DIOR (275252245)