Label: POVIDON IODINE PREP PADS- povidone-iodine swab
- NDC Code(s): 49953-0725-1
- Packager: Dalian Rongbang Medical Healthy Devices Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated December 28, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
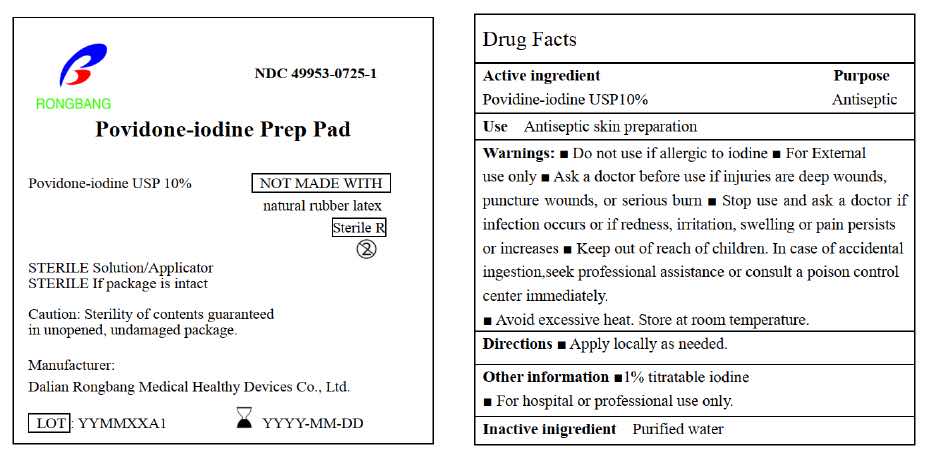
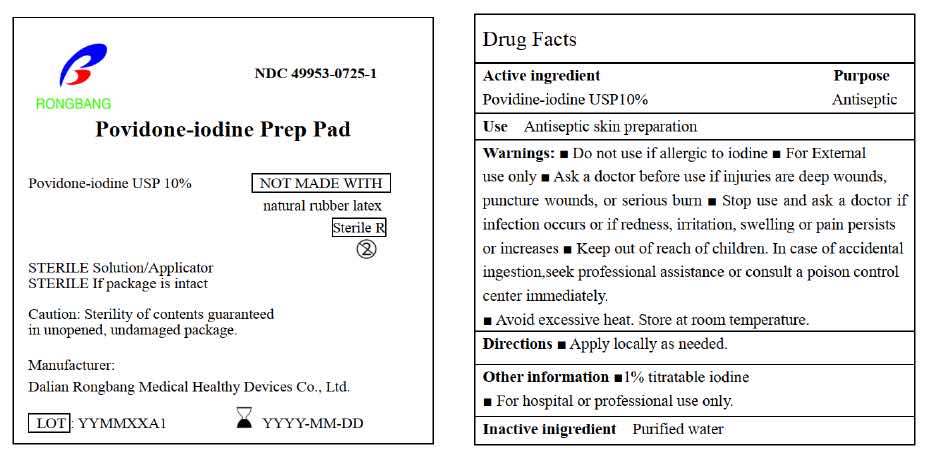
- ACTIVE INGREDIENT
- PURPOSE
- USE
- WARNINGS
- DO NOT USE
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENT
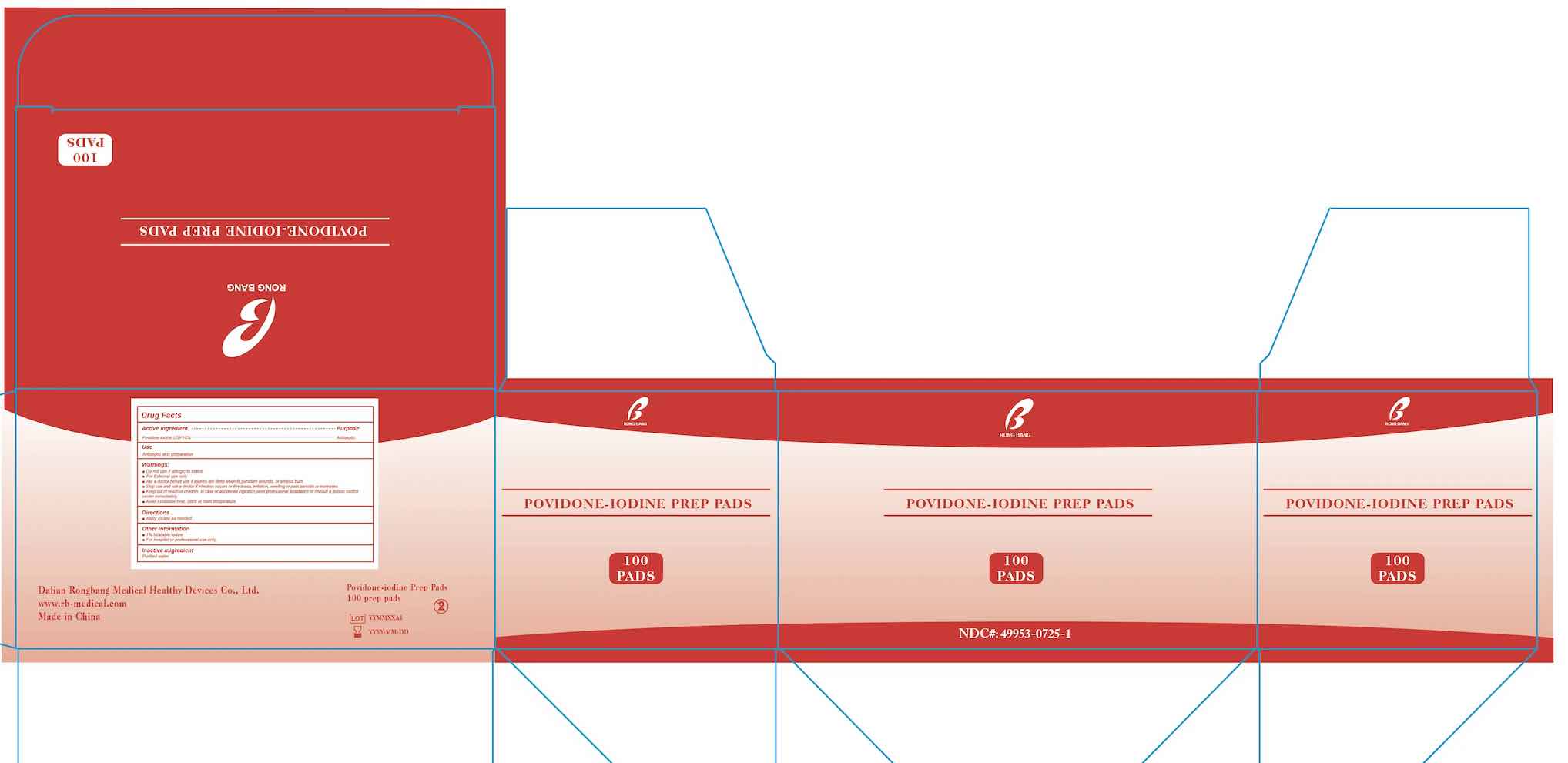
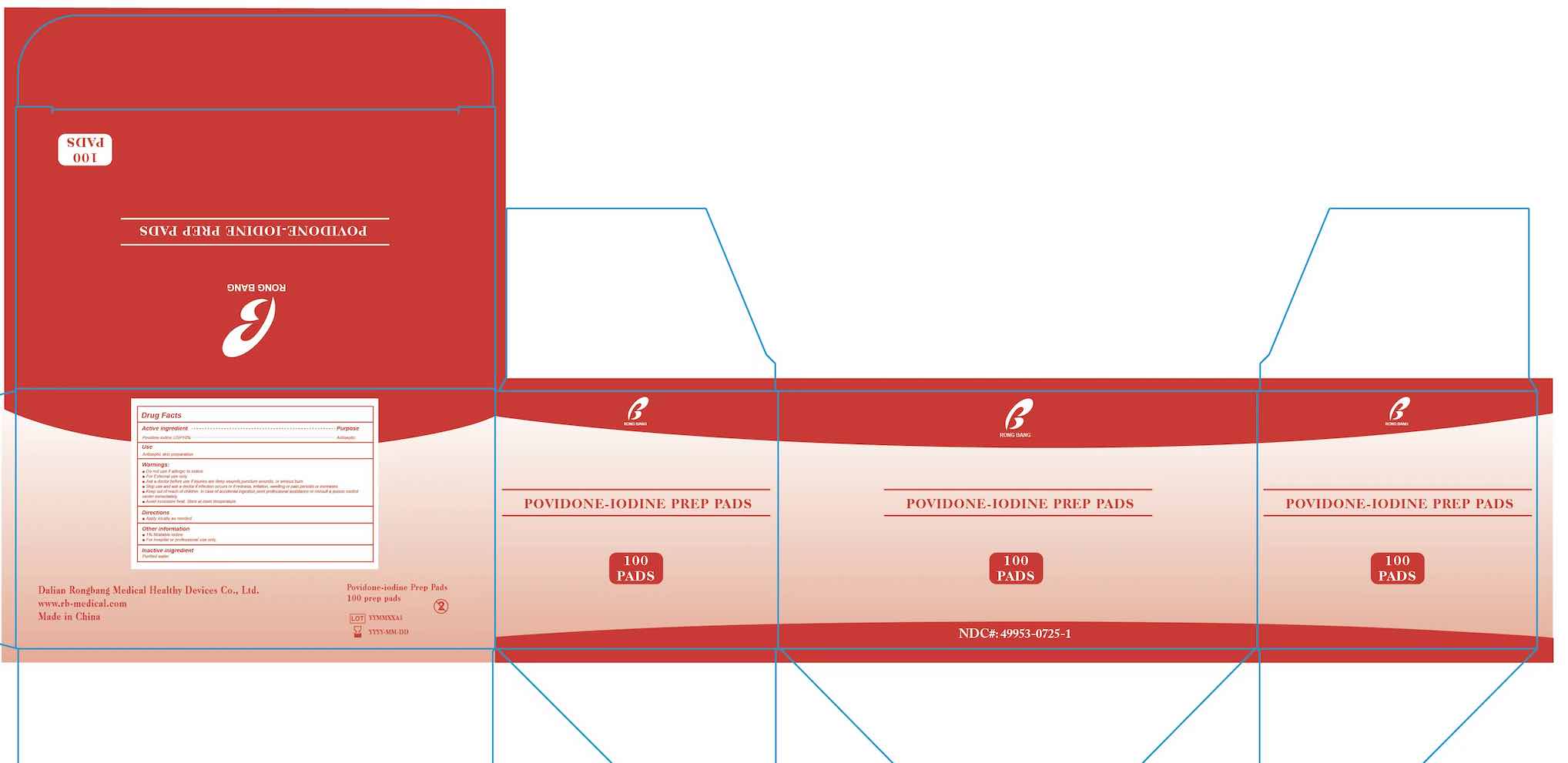
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
POVIDON IODINE PREP PADS
povidone-iodine swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49953-0725 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE-IODINE (UNII: 85H0HZU99M) (IODINE - UNII:9679TC07X4) IODINE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49953-0725-1 100 in 1 BOX 02/10/2023 1 1 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 02/10/2023 Labeler - Dalian Rongbang Medical Healthy Devices Co., Ltd. (529882730) Establishment Name Address ID/FEI Business Operations Dalian Rongbang Medical Healthy Devices Co., Ltd. 529882730 manufacture(49953-0725)