Label: TRIPLE ANTIBIOTIC AND PAIN RELIEF- bacitracin, neomycin, polymyxin b, pramoxine hydrochloride ointment
- NDC Code(s): 69842-562-17, 69842-562-58, 69842-562-64
- Packager: CVS Pharmacy
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 6, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each gram)
- Purpose
- Uses
-
Warnings
For external use only.
Do not use
- •
- if you are allergic to any of the ingredients
- •
- in the eyes
- •
- over large areas of the body
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
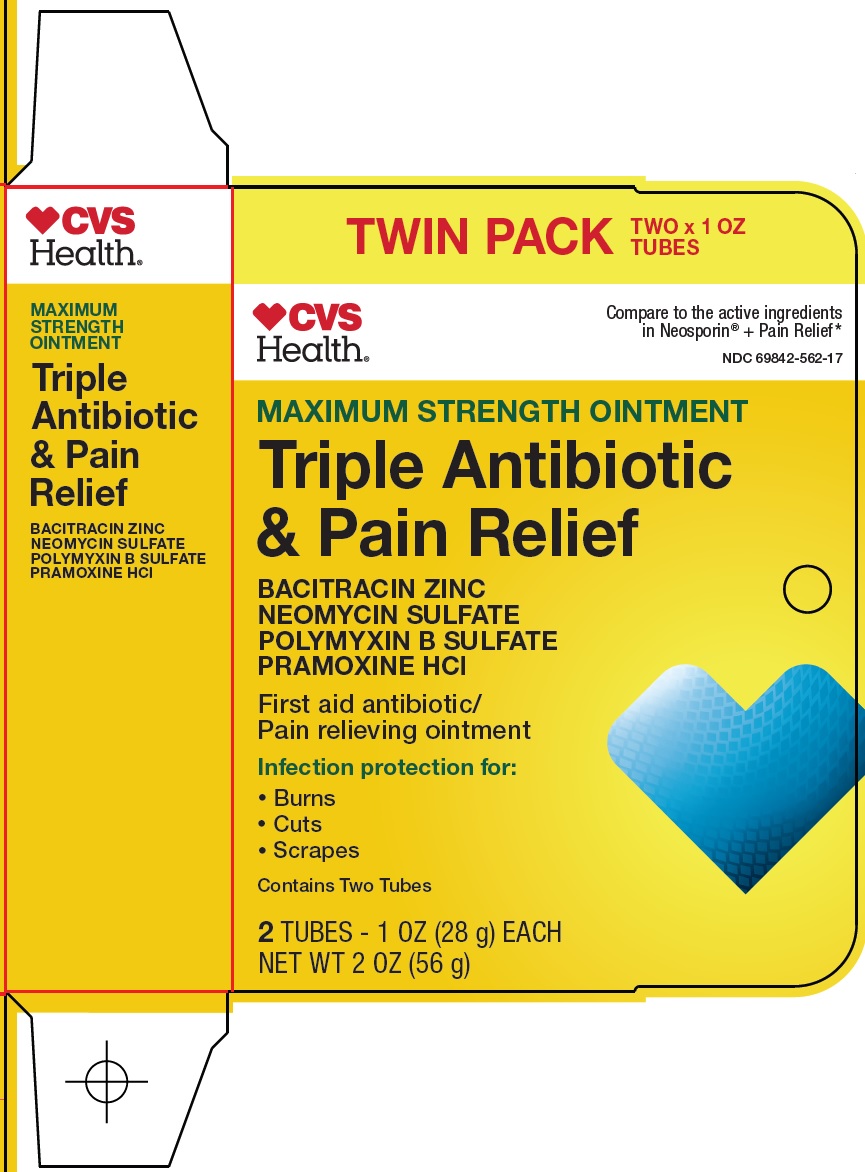
Principal Display Panel
TWIN PACK
TWO x 1 OZ TUBES
CVS Health®
Compare to the active ingredients in Neosporin® + Pain Relief
MAXIMUM STRENGTH OINTMENT
Triple Antibiotic & Pain Relief
BACITRACIN ZINC
NEOMYCIN SULFATE
POLYMYXIN B SULFATE
PRAMOXINE HCl
First aid antibiotic/pain relieving ointment
Infection protection for:
● Burns
● Cuts
● Scrapes
Contains Two Tubes
2 TUBES – 1 OZ (28 g) EACH
NET WT 2 OZ (56 g)
-
INGREDIENTS AND APPEARANCE
TRIPLE ANTIBIOTIC AND PAIN RELIEF
bacitracin, neomycin, polymyxin b, pramoxine hydrochloride ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-562 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 500 [USP'U] in 1 g NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 g POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 10000 [USP'U] in 1 g PRAMOXINE HYDROCHLORIDE (UNII: 88AYB867L5) (PRAMOXINE - UNII:068X84E056) PRAMOXINE HYDROCHLORIDE 10 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) Product Characteristics Color YELLOW Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-562-64 1 in 1 CARTON 09/09/2020 1 28 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:69842-562-58 1 in 1 CARTON 09/09/2020 2 14 g in 1 TRAY; Type 0: Not a Combination Product 3 NDC:69842-562-17 2 in 1 CARTON 08/13/2023 3 28 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M004 09/09/2020 Labeler - CVS Pharmacy (062312574)