Label: EUPHRASIA OFFICINALIS 30C- euphrasia officinalis pellet
- NDC Code(s): 76472-4049-1
- Packager: SEVENE USA
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated January 25, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
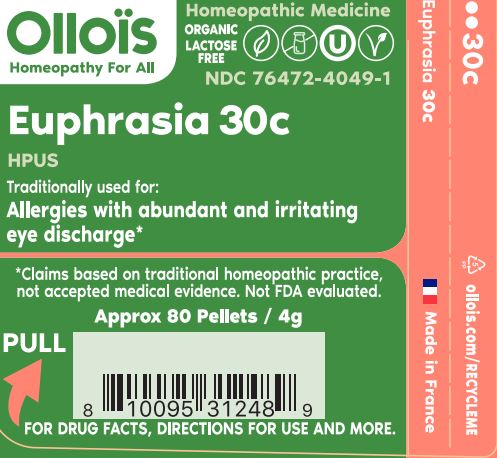
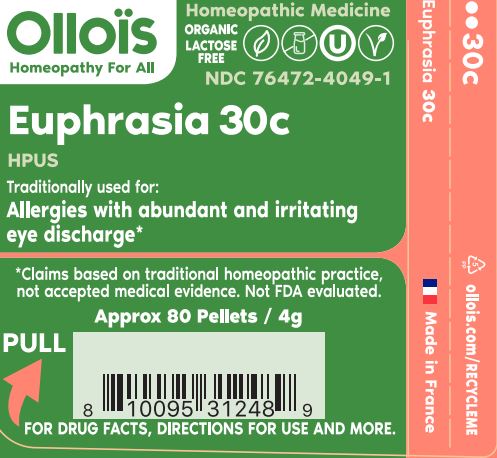
- TRADITIONALLY USED FOR:
- DOSAGE & ADMINISTRATION
- USE
- WARNINGS
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- OTHER INFORMATION
- DO NOT USE
- INACTIVE INGREDIENT
- QUESTIONS?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EUPHRASIA OFFICINALIS 30C
euphrasia officinalis pelletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76472-4049 Route of Administration SUBLINGUAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength EUPHRASIA STRICTA (UNII: C9642I91WL) (EUPHRASIA STRICTA - UNII:C9642I91WL) EUPHRASIA STRICTA 30 [hp_C] Inactive Ingredients Ingredient Name Strength SUCROSE (UNII: C151H8M554) Product Characteristics Color white Score Shape ROUND Size 4mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76472-4049-1 80 in 1 CYLINDER; Type 0: Not a Combination Product 07/08/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 07/08/2022 Labeler - SEVENE USA (969332936)