Label: CETIRIZINE HYDROCHLORIDE tablet
- NDC Code(s): 21130-640-03, 21130-640-09, 21130-640-18
- Packager: SAFEWAY
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated December 18, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
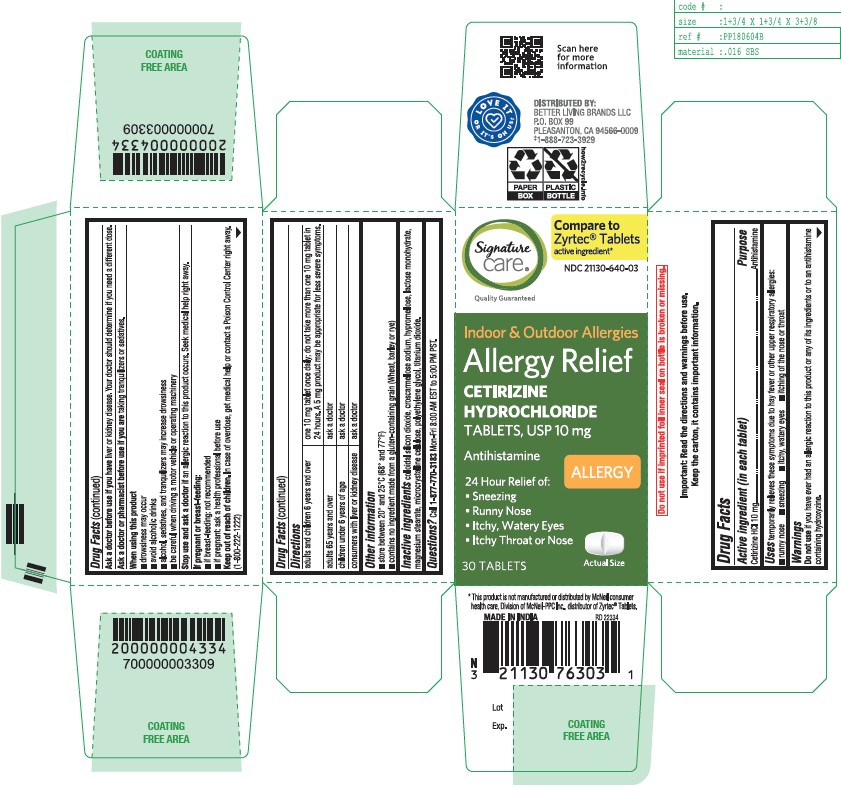
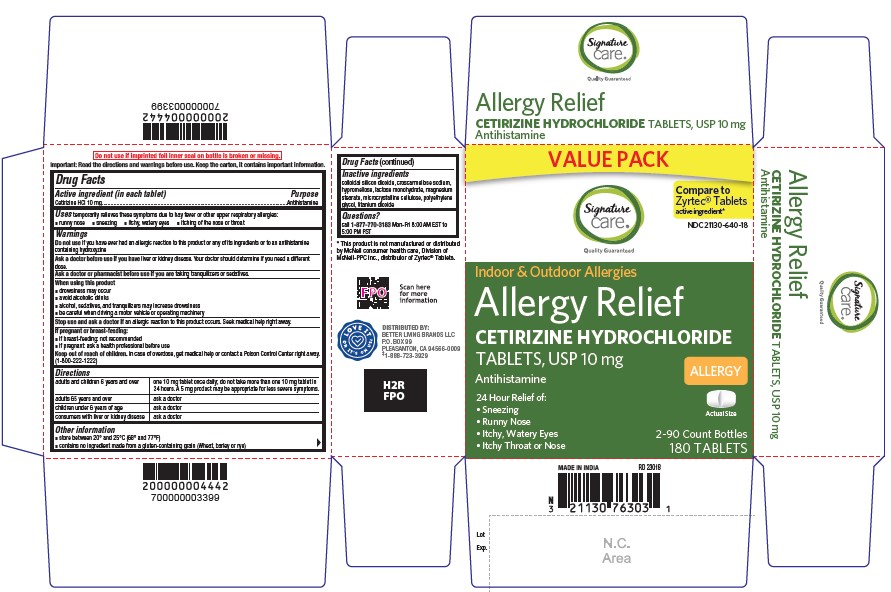
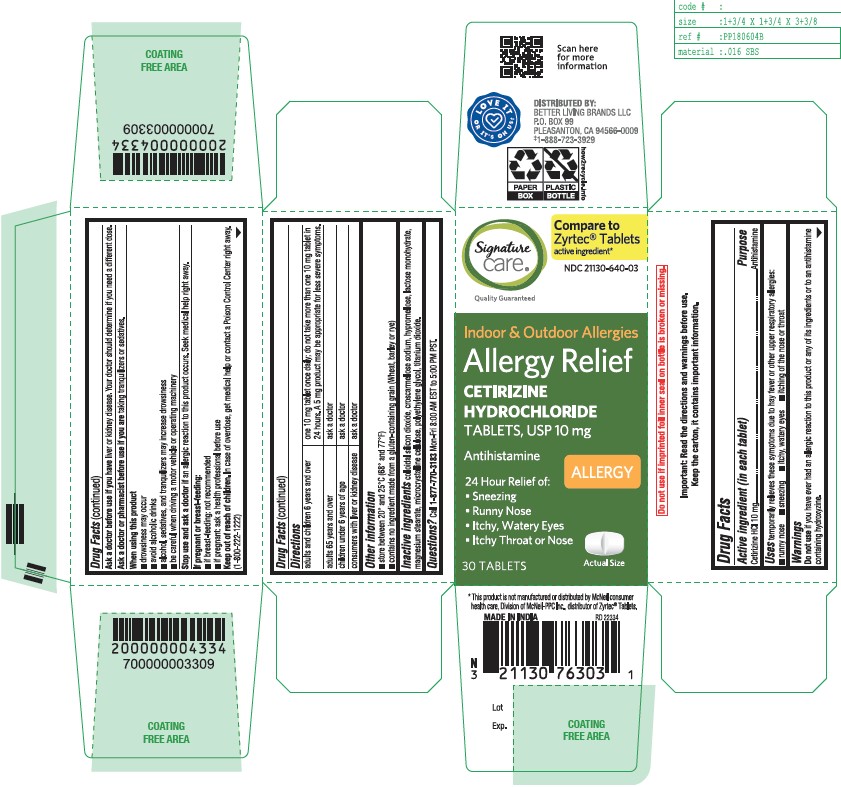
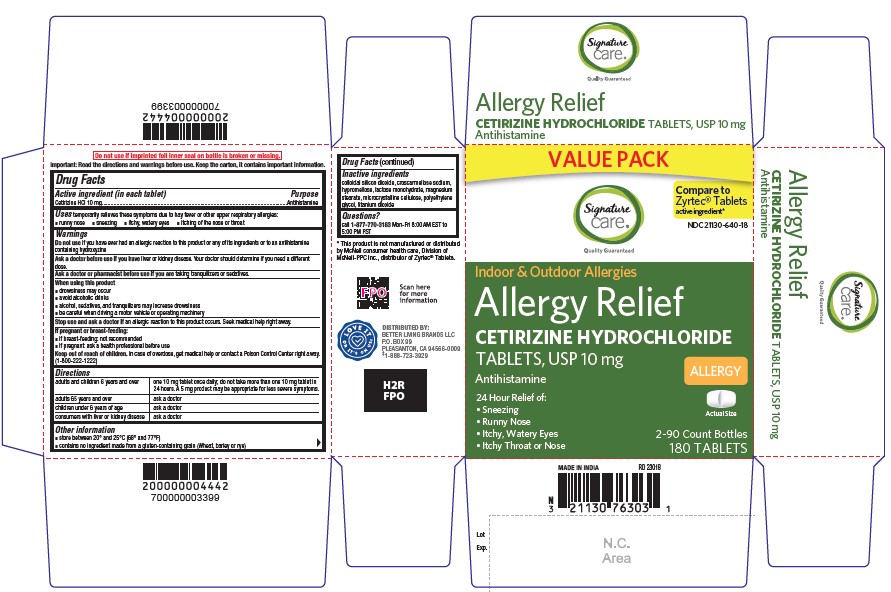
- Active ingredient (in each tablet)
- Purpose
- Uses
- Do not use
- Ask a doctor before use if you have
- Ask a doctor or pharmacist before use if you are
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding
- Keep out of reach of children
-
Directions
adults and children 6 years and over
one 10 mg tablet once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms.
adults 65 years and over
ask a doctor
children under 6 years of ageask a doctor
consumers with liver or kidney disease
ask a doctor - Other information
- Inactive ingredients
- Questions?
- Principal Display Panel
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
CETIRIZINE HYDROCHLORIDE
cetirizine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21130-640 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CETIRIZINE HYDROCHLORIDE (UNII: 64O047KTOA) (CETIRIZINE - UNII:YO7261ME24) CETIRIZINE HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) Product Characteristics Color white (White to off-white) Score 2 pieces Shape RECTANGLE (rounded off rectangular) Size 9mm Flavor Imprint Code G;4 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21130-640-03 30 in 1 BOTTLE; Type 0: Not a Combination Product 06/30/2023 2 NDC:21130-640-18 180 in 1 CARTON; Type 0: Not a Combination Product 06/30/2023 3 NDC:21130-640-09 90 in 1 BOTTLE; Type 0: Not a Combination Product 06/30/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209274 06/30/2023 Labeler - SAFEWAY (009137209)