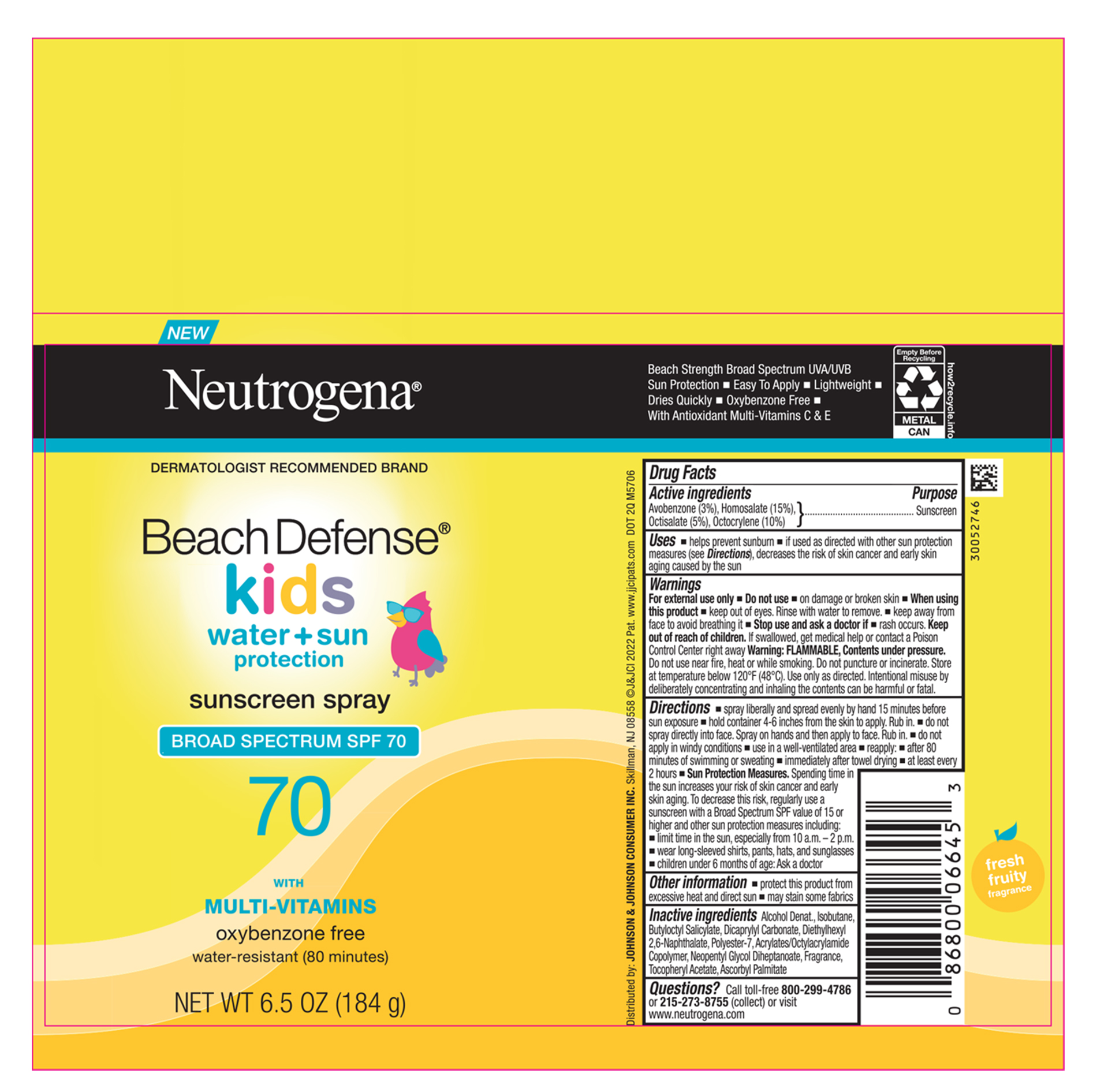
Label: NEUTROGENA BEACH DEFENSE KIDS SUNSCREEN SPF 70- avobenzone, homosalate, octisalate, octocrylene aerosol, spray
- NDC Code(s): 69968-0779-7
- Packager: Kenvue Brands LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 12, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Uses
-
Warnings
For external use only
- When using this product keep out of eyes. Rinse with water to remove.
- Keep away from face to avoid breathing it
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away
Warning: FLAMMABLE, Contents under pressure. Do not use near fire, heat or while smoking. Do not puncture or incinerate. Store at temperature below 120°F (48°C). Use only as directed. Intentional misuse by deliberately concentrating and inhaling the contents can be harmful or fatal.
-
Directions
- spray liberally and spread evenly by hand 15 minutes before sun exposure
- hold container 4-6 inches from the skin to apply. Rub in.
- do not spray directly into face. Spray on hands and then apply to face. Rub in.
- do not apply in windy conditions.
- use in well-ventilated area
- reapply:
- after 80 minutes of swimming or sweating
- immediately after towel drying
- at least every 2 hours
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
- children under 6 months of age: Ask a doctor
- Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
- Purpose
- PRINCIPAL DISPLAY PANEL - 184g Can
-
INGREDIENTS AND APPEARANCE
NEUTROGENA BEACH DEFENSE KIDS SUNSCREEN SPF 70
avobenzone, homosalate, octisalate, octocrylene aerosol, sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69968-0779 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 150 mg in 1 g OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 100 mg in 1 g AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 g Inactive Ingredients Ingredient Name Strength DIETHYLHEXYL 2,6-NAPHTHALATE (UNII: I0DQJ7YGXM) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) ASCORBYL PALMITATE (UNII: QN83US2B0N) ISOBUTANE (UNII: BXR49TP611) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) POLYESTER-7 (UNII: 0841698D2F) NEOPENTYL GLYCOL DIHEPTANOATE (UNII: 5LKW3C543X) ALCOHOL (UNII: 3K9958V90M) ACRYLATE/ISOBUTYL METHACRYLATE/N-TERT-OCTYLACRYLAMIDE COPOLYMER (75000 MW) (UNII: JU3XHR8VWK) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69968-0779-7 184 g in 1 CAN; Type 0: Not a Combination Product 12/05/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 12/05/2022 Labeler - Kenvue Brands LLC (118772437)