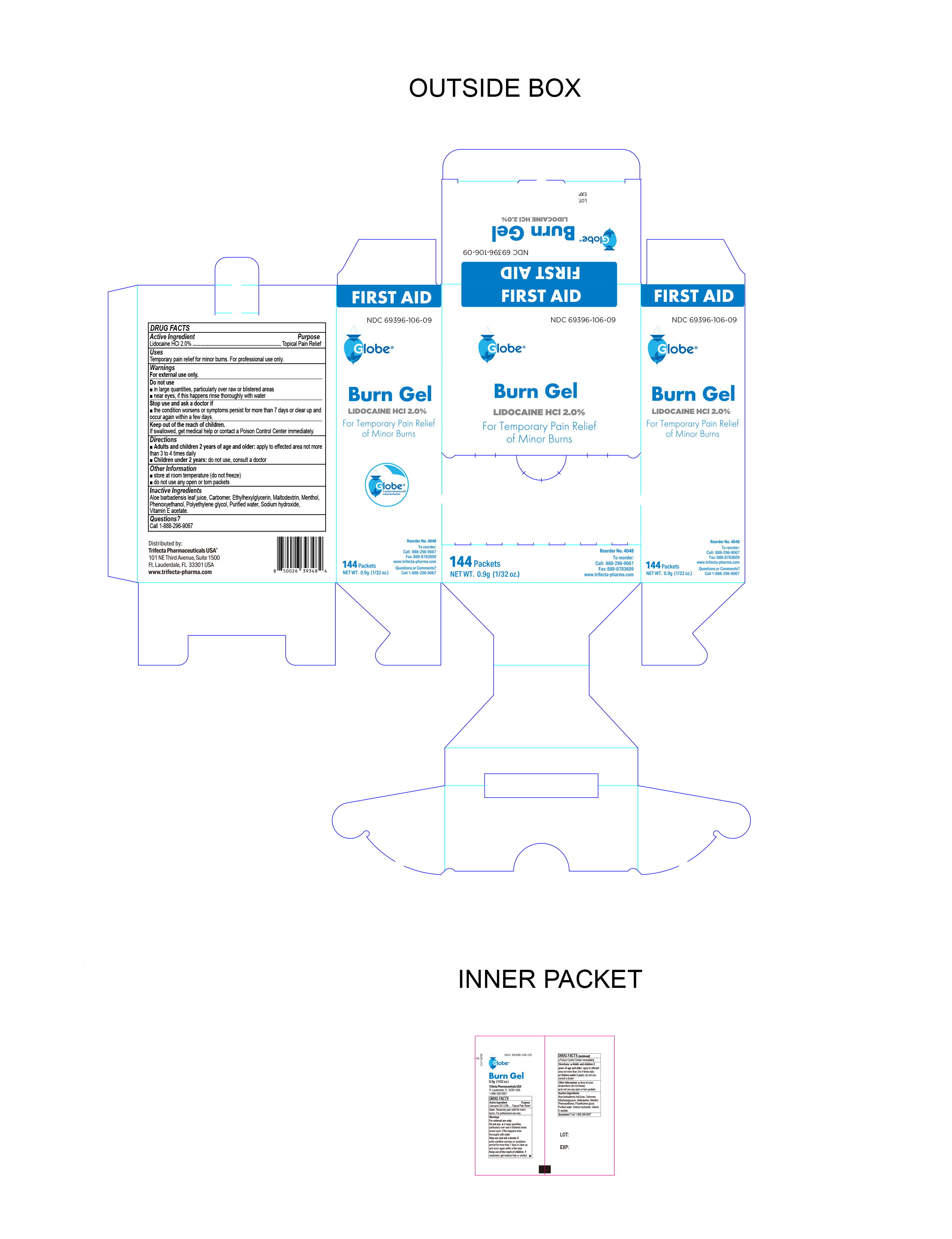
Label: BURN GEL- lidocaine hci gel
- NDC Code(s): 69396-106-09
- Packager: Trifecta Pharmaceuticals USA LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated July 12, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
- Warnings
- Directions
- Inactive Ingredients
- Questions?
- Stop Use and Ask a doctor if
- Storage Information
- Keep out of reach of children
- Other Information
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BURN GEL
lidocaine hci gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69396-106 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 0.02 g in 1 g Inactive Ingredients Ingredient Name Strength .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) MENTHOL (UNII: L7T10EIP3A) WATER (UNII: 059QF0KO0R) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) ALOE VERA LEAF (UNII: ZY81Z83H0X) SODIUM HYDROXIDE (UNII: 55X04QC32I) MALTODEXTRIN (UNII: 7CVR7L4A2D) PHENOXYETHANOL (UNII: HIE492ZZ3T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69396-106-09 144 in 1 CARTON 09/27/2022 1 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 09/27/2022 Labeler - Trifecta Pharmaceuticals USA LLC (079424163)