Label: PRENATAL- vitamin a, ascorbic acid, cholecalciferol, .alpha.-tocopherol, thiamine, riboflavin, niacin, pyridoxine, folic acid, cyanocobalamin, calcium, ferrous fumarate, and zinc oxide tablet
- NHRIC Code(s): 76413-315-01
- Packager: Central Texas Community Health Centers
- Category: DIETARY SUPPLEMENT
Drug Label Information
Updated September 15, 2017
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
STATEMENT OF IDENTITY
Supplement Facts Serving Size: 1 Tablet Amount Per Serving % Daily Value* - *
- % Daily Value for Pregnant and Lactating Women
Vitamin A (as Acetate and Beta Carotene) 4000 I.U. 50% Vitamin C (as Ascorbic Acid) 100 mg. 167% Vitamin D-3 (as Cholecalciferol) 400 I.U. 100% Vitamin E (as dl-Alpha Tocopheryl Acetate) 11 I.U. 37% Vitamin B-1 (as Thiamine Mononitrate) 1.5 mg. 88% Vitamin B-2 (Riboflavin) 1.7 mg. 85% Vitamin B-3 (as Niacinamide) 18 mg. 90% Vitamin B-6 (as Pyridoxine HCl) 2.6 mg. 104% Folic Acid 0.8 mg. 100% Vitamin B-12 (as Cyanocobalamin) 4 mcg. 50% Calcium (as Calcium Carbonate and Calcium Sulfate) 263 mg. 26% Iron (as Ferrous Fumarate) 27 mg. 150% Zinc (as Zinc Oxide) 25 mg. 167% OTHER INGREDIENTS: Microcrystalline Cellulose, Stearic Acid, Hypromellose, Croscarmellose Sodium, Gelatin, Acacia, Maltodextrin, Titanium Dioxide, Talc, Triacetin, Starch, Magnesium Stearate, Dicalcium Phosphate, Mineral Oil, Sucrose, Silica, Sodium Ascorbate, Triglycerides, FD&C Red #40 Lake, FD&C Yellow #6 Lake, Tocopherols, Sodium Benzoate, Sorbic Acid, Tricalcium Phosphate, BHT and Ascorbyl Palmitate.
- DIRECTIONS
-
WARNING
If you are pregnant, nursing, taking any medications or planning any medical procedure, consult your doctor before use. Discontinue use and consult your doctor if any adverse reactions occur.
WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under six. KEEP THIS PRODUCT OUT OF THE REACH OF CHILDREN. In case of accidental overdose, call a doctor or Poison Control Center immediately. - FREE OF
- HEALTH CLAIM
- HEALTH CLAIM
-
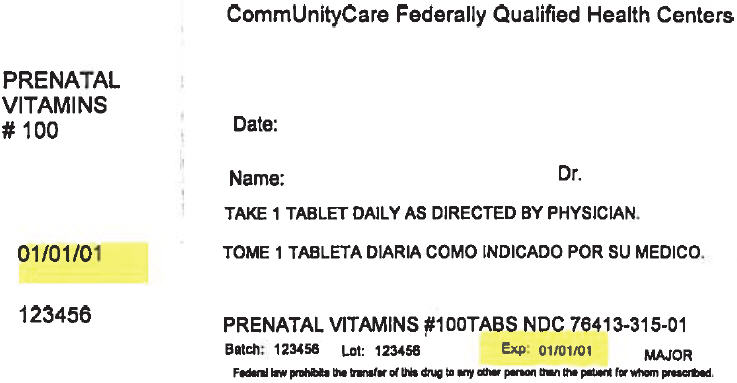
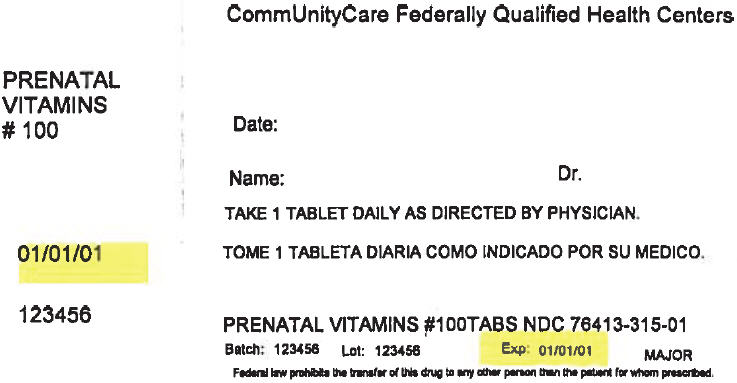
PRINCIPAL DISPLAY PANEL - 100 Tablet Bottle Label
CommUnityCare Federally Qualified Health Centers
PRENATAL
VITAMINS
# 100Date:
Name:
Dr.TAKE 1 TABLET DAILY AS DIRECTED BY PHYSICIAN.
01/01/01
123456
PRENATAL VITAMINS #100TABS NDC 76413-315-01
Batch:
123456
Lot:
123456
Exp:
01/01/01MAJOR
Federal law prohibits the transfer of this drug to any other person than the patient for whom prescribed.
-
INGREDIENTS AND APPEARANCE
PRENATAL
vitamin a, ascorbic acid, cholecalciferol, .alpha.-tocopherol, thiamine, riboflavin, niacin, pyridoxine, folic acid, cyanocobalamin, calcium, ferrous fumarate, and zinc oxide tabletProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:76413-315 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Vitamin A (UNII: 81G40H8B0T) (Vitamin A - UNII:81G40H8B0T) Vitamin A 4000 [iU] Ascorbic Acid (UNII: PQ6CK8PD0R) (Ascorbic Acid - UNII:PQ6CK8PD0R) Ascorbic Acid 100 mg Cholecalciferol (UNII: 1C6V77QF41) (Cholecalciferol - UNII:1C6V77QF41) Cholecalciferol 400 [iU] .Alpha.-Tocopherol (UNII: H4N855PNZ1) (.Alpha.-Tocopherol - UNII:H4N855PNZ1) .Alpha.-Tocopherol 11 [iU] Thiamine (UNII: X66NSO3N35) (Thiamine Ion - UNII:4ABT0J945J) Thiamine 1.5 mg Riboflavin (UNII: TLM2976OFR) (Riboflavin - UNII:TLM2976OFR) Riboflavin 1.7 mg Niacin (UNII: 2679MF687A) (Niacin - UNII:2679MF687A) Niacin 18 mg Pyridoxine (UNII: KV2JZ1BI6Z) (Pyridoxine - UNII:KV2JZ1BI6Z) Pyridoxine 2.6 mg Folic acid (UNII: 935E97BOY8) (Folic acid - UNII:935E97BOY8) Folic acid 0.8 mg Cyanocobalamin (UNII: P6YC3EG204) (Cyanocobalamin - UNII:P6YC3EG204) Cyanocobalamin 4 ug Calcium (UNII: SY7Q814VUP) (Calcium - UNII:SY7Q814VUP) Calcium 263 mg Ferrous fumarate (UNII: R5L488RY0Q) (Ferrous cation - UNII:GW89581OWR) Ferrous cation 27 mg Zinc oxide (UNII: SOI2LOH54Z) (Zinc oxide - UNII:SOI2LOH54Z) Zinc oxide 25 mg Inactive Ingredients Ingredient Name Strength Microcrystalline cellulose (UNII: OP1R32D61U) stearic acid (UNII: 4ELV7Z65AP) Hypromellose, Unspecified (UNII: 3NXW29V3WO) croscarmellose sodium (UNII: M28OL1HH48) Gelatin, Unspecified (UNII: 2G86QN327L) acacia (UNII: 5C5403N26O) maltodextrin (UNII: 7CVR7L4A2D) titanium dioxide (UNII: 15FIX9V2JP) talc (UNII: 7SEV7J4R1U) triacetin (UNII: XHX3C3X673) magnesium stearate (UNII: 70097M6I30) Anhydrous Dibasic Calcium Phosphate (UNII: L11K75P92J) mineral oil (UNII: T5L8T28FGP) sucrose (UNII: C151H8M554) Silicon Dioxide (UNII: ETJ7Z6XBU4) sodium ascorbate (UNII: S033EH8359) FD&C Red NO. 40 (UNII: WZB9127XOA) FD&C Yellow NO. 6 (UNII: H77VEI93A8) Aluminum Oxide (UNII: LMI26O6933) Tocopherol (UNII: R0ZB2556P8) Sodium Benzoate (UNII: OJ245FE5EU) Sorbic acid (UNII: X045WJ989B) Tricalcium Phosphate (UNII: K4C08XP666) Butylated Hydroxytoluene (UNII: 1P9D0Z171K) Ascorbyl Palmitate (UNII: QN83US2B0N) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:76413-315-01 100 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 03/01/2016 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color scoring 1 shape size (solid drugs) 1 mm Labeler - Central Texas Community Health Centers (079674019) Establishment Name Address ID/FEI Business Operations Central Texas Community Health Centers 079674019 REPACK, RELABEL Establishment Name Address ID/FEI Business Operations Major Pharmaceuticals 383359612 MANUFACTURE