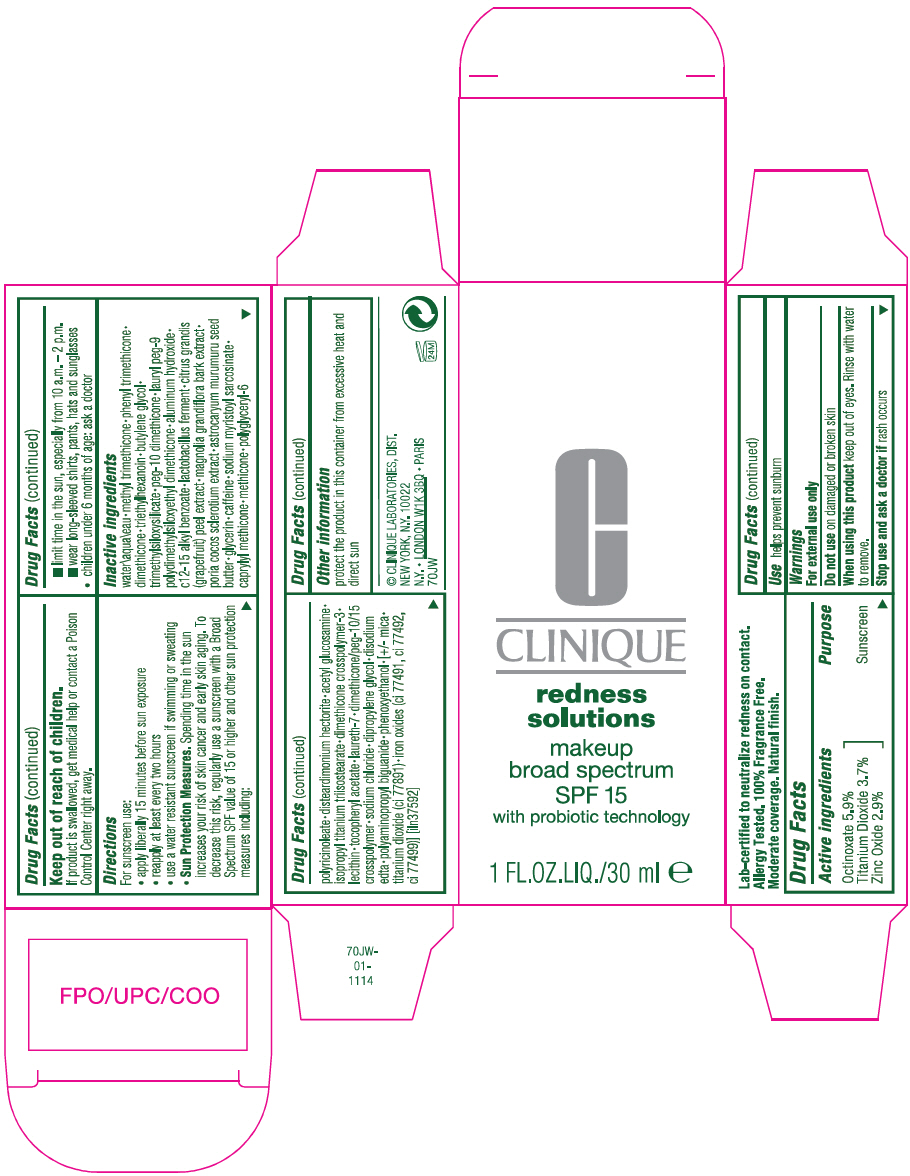
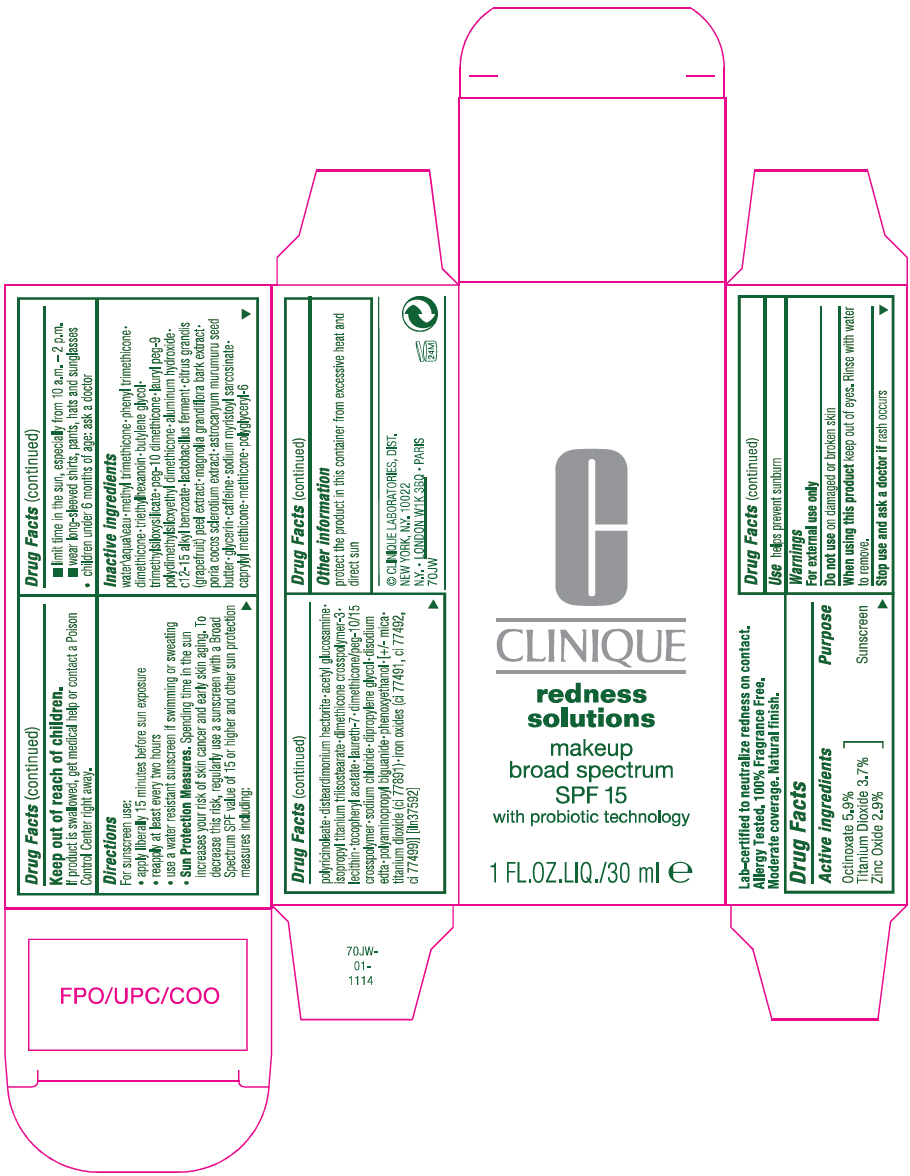
Label: REDNESS SOLUTIONS MAKEUP BROAD SPECTRUM SPF 15 WITH PROBIOTIC TECHNOLOGY- octinoxate, titanium dioxide, and zinc oxide lotion
- NDC Code(s): 49527-070-01
- Packager: CLINIQUE LABORATORIES LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 13, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Purpose
- Use
- Warnings
-
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- reapply at least every two hours
- use a water resistant sunscreen if swimming or sweating
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: ask a doctor
-
Inactive ingredients
water\aqua\eau • methyl trimethicone • phenyl trimethicone • dimethicone • triethylhexanoin • butylene glycol • trimethylsiloxysilicate • peg-10 dimethicone • lauryl peg-9 polydimethylsiloxyethyl dimethicone • aluminum hydroxide • c12-15 alkyl benzoate • lactobacillus ferment • citrus grandis (grapefruit) peel extract • magnolia grandiflora bark extract • poria cocos sclerotium extract • astrocaryum murumuru seed butter • glycerin • caffeine • sodium myristoyl sarcosinate • caprylyl methicone • methicone • polyglyceryl-6 polyricinoleate • disteardimonium hectorite • acetyl glucosamine • isopropyl titanium triisostearate • dimethicone crosspolymer-3 • lecithin • tocopheryl acetate • laureth-7 • dimethicone/peg-10/15 crosspolymer • sodium chloride • dipropylene glycol • disodium edta • polyaminopropyl biguanide • phenoxyethanol • [+/- mica • titanium dioxide (ci 77891) • iron oxides (ci 77491, ci 77492, ci 77499)] [iln37592]
- Other information
- PRINCIPAL DISPLAY PANEL - 30 ml Bottle Carton
-
INGREDIENTS AND APPEARANCE
REDNESS SOLUTIONS MAKEUP BROAD SPECTRUM SPF 15 WITH PROBIOTIC TECHNOLOGY
octinoxate, titanium dioxide, and zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49527-070 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 59 mg in 1 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 37 mg in 1 mL ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 29 mg in 1 mL Inactive Ingredients Ingredient Name Strength METHYL TRIMETHICONE (UNII: S73ZQI0GXM) WATER (UNII: 059QF0KO0R) PHENYL TRIMETHICONE (UNII: DR0K5NOJ4R) DIMETHICONE (UNII: 92RU3N3Y1O) TRIETHYLHEXANOIN (UNII: 7K3W1BIU6K) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) PEG-10 DIMETHICONE (600 CST) (UNII: 8PR7V1SVM0) LAURYL PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE (UNII: 25G622K2RA) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) LIMOSILACTOBACILLUS REUTERI (UNII: 9913I24QEE) GRAPEFRUIT (UNII: O82C39RR8C) ASTROCARYUM MURUMURU SEED BUTTER (UNII: 12V64UPU6R) GLYCERIN (UNII: PDC6A3C0OX) CAFFEINE (UNII: 3G6A5W338E) SODIUM MYRISTOYL SARCOSINATE (UNII: J07237209D) CAPRYLYL TRISILOXANE (UNII: Q95M2P1KJL) METHICONE (20 CST) (UNII: 6777U11MKT) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) N-ACETYLGLUCOSAMINE (UNII: V956696549) ISOPROPYL TITANIUM TRIISOSTEARATE (UNII: 949E3KBJ1I) LAURETH-7 (UNII: Z95S6G8201) SODIUM CHLORIDE (UNII: 451W47IQ8X) DIPROPYLENE GLYCOL (UNII: E107L85C40) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) PHENOXYETHANOL (UNII: HIE492ZZ3T) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49527-070-01 1 in 1 CARTON 12/01/2011 1 30 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 12/01/2011 Labeler - CLINIQUE LABORATORIES LLC (044475127) Registrant - Estee Lauder Companies Inc. (790802086) Establishment Name Address ID/FEI Business Operations Estee Lauder N.V. 370151326 manufacture(49527-070) , pack(49527-070) , label(49527-070)