Label: TOUCHLAND HAND SANITIZER SMILEY VARIETY PACK- alcohol kit
- NDC Code(s): 72033-901-03
- Packager: Touchland LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated February 5, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
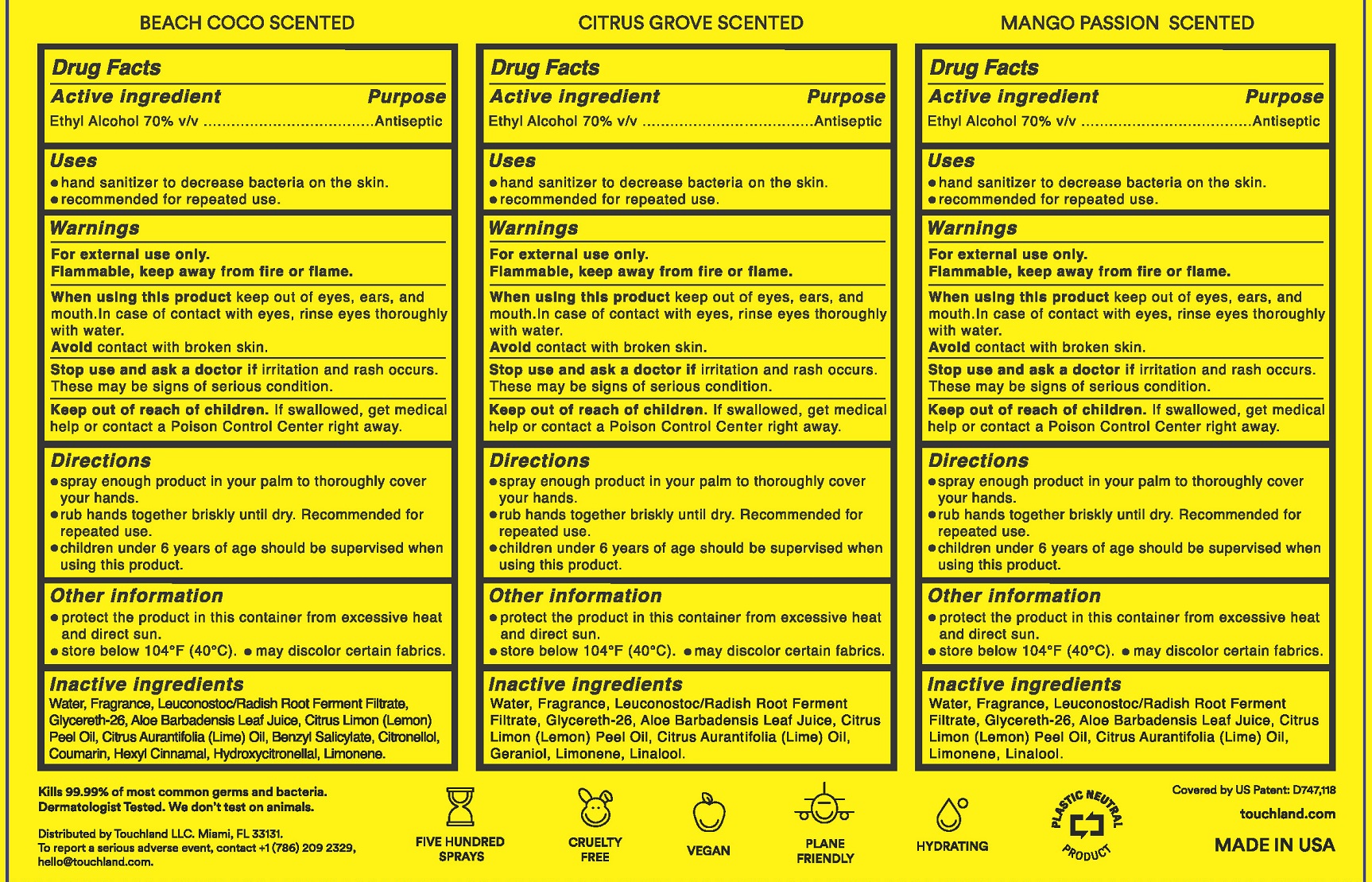
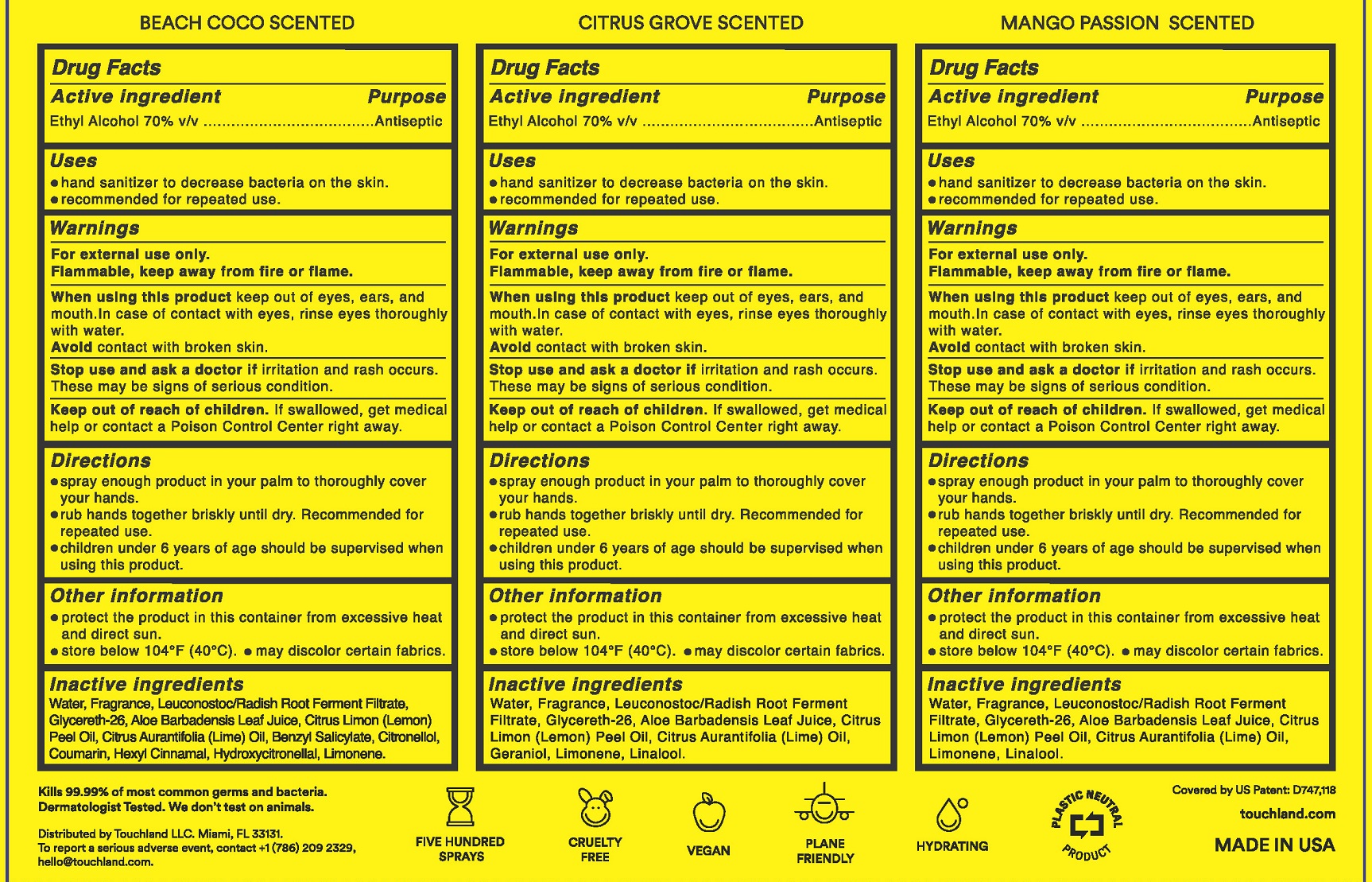
- Active ingredient
- Purpose
- Use
- Warnings
- Directions
- Other Information
- Inactive ingredients (Beach Coco)
- Inactive ingredients (Citrus Grove)
- Inactive Ingredients (Mango Passion)
- Label
-
INGREDIENTS AND APPEARANCE
TOUCHLAND HAND SANITIZER SMILEY VARIETY PACK
alcohol kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72033-901 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72033-901-03 1 in 1 PACKAGE; Type 0: Not a Combination Product 07/22/2022 12/31/2023 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE, SPRAY 30 mL Part 2 1 BOTTLE, SPRAY 30 mL Part 3 1 BOTTLE, SPRAY 30 mL Part 1 of 3 TOUCHLAND POWER MIST HAND SANITIZER BEACH COCO
alcohol sprayProduct Information Item Code (Source) NDC:72033-116 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength LIME OIL (UNII: UZH29XGA8G) LIMONENE, (+)- (UNII: GFD7C86Q1W) .ALPHA.-HEXYLCINNAMALDEHYDE (UNII: 7X6O37OK2I) LEMON OIL (UNII: I9GRO824LL) WATER (UNII: 059QF0KO0R) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) COUMARIN (UNII: A4VZ22K1WT) GLYCERETH-26 (UNII: NNE56F2N14) BENZYL SALICYLATE (UNII: WAO5MNK9TU) ALOE VERA LEAF (UNII: ZY81Z83H0X) LEUCONOSTOC/RADISH ROOT FERMENT FILTRATE (UNII: D2QHA03458) HYDROXYCITRONELLAL (UNII: 8SQ0VA4YUR) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 30 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 07/22/2022 12/31/2023 Part 2 of 3 TOUCHLAND POWER MIST HAND SANITIZER CITRUS GROVE
alcohol sprayProduct Information Item Code (Source) NDC:72033-117 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength LIME OIL (UNII: UZH29XGA8G) GERANIOL (UNII: L837108USY) LIMONENE, (+)- (UNII: GFD7C86Q1W) LEMON OIL (UNII: I9GRO824LL) WATER (UNII: 059QF0KO0R) GLYCERETH-26 (UNII: NNE56F2N14) LINALOOL, (+)- (UNII: F4VNO44C09) ALOE VERA LEAF (UNII: ZY81Z83H0X) LEUCONOSTOC/RADISH ROOT FERMENT FILTRATE (UNII: D2QHA03458) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 30 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 07/22/2022 12/31/2023 Part 3 of 3 TOUCHLAND POWER MIST HAND SANITIZER MANGO PASSION
alcohol sprayProduct Information Item Code (Source) NDC:72033-115 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength LIME OIL (UNII: UZH29XGA8G) LIMONENE, (+)- (UNII: GFD7C86Q1W) LINALOOL, (+)- (UNII: F4VNO44C09) WATER (UNII: 059QF0KO0R) GLYCERETH-26 (UNII: NNE56F2N14) ALOE VERA LEAF (UNII: ZY81Z83H0X) LEUCONOSTOC/RADISH ROOT FERMENT FILTRATE (UNII: D2QHA03458) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 30 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 07/22/2022 12/31/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 07/22/2022 Labeler - Touchland LLC (036656461) Registrant - Touchland LLC (036656461) Establishment Name Address ID/FEI Business Operations MICRO CONNECTION ENTERPRISES, INC., THE 144297160 manufacture(72033-901)