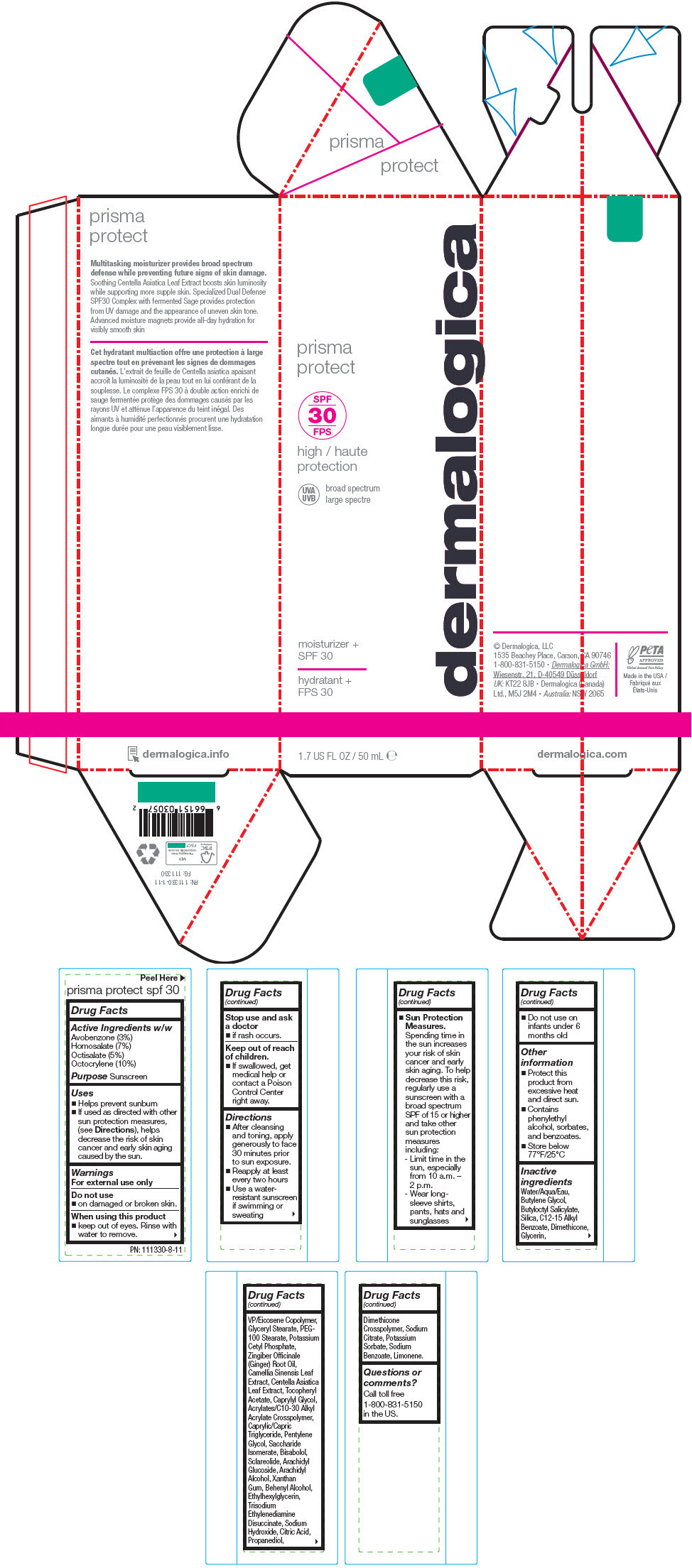
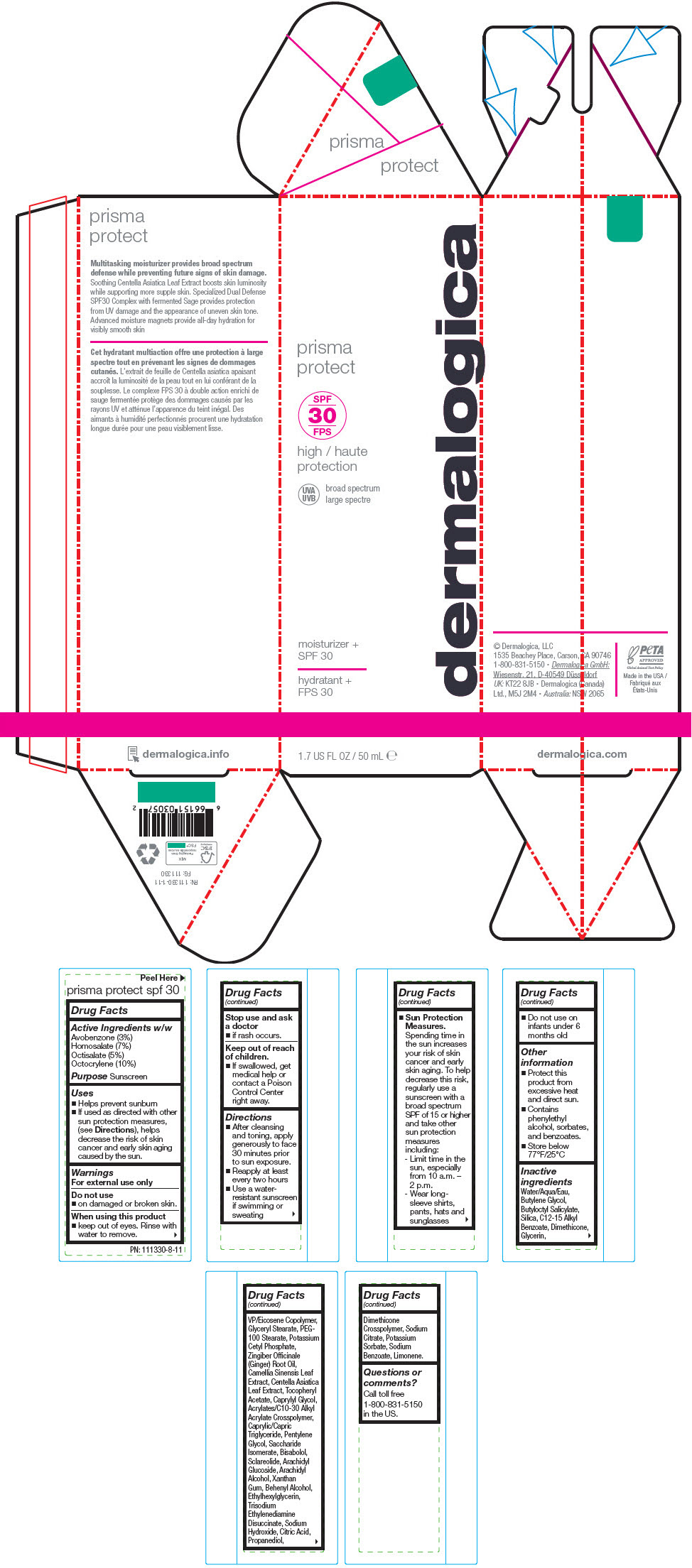
Label: PRISMA PROTECT SPF30- octocrylene, homosalate, octisalate, and avobenzone lotion
- NDC Code(s): 68479-852-02
- Packager: Dermalogica, LLC.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated April 25, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredients w/w
- Purpose
-
Uses
- Helps prevent sunburn
- If used as directed with other sun protection measures, (see Directions), helps decrease the risk of skin cancer and early skin aging caused by the sun.
- Warnings
-
Directions
- After cleansing and toning, apply generously to face 30 minutes prior to sun exposure.
- Reapply at least every two hours
- Use a water-resistant sunscreen if swimming or sweating
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To help decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and take other sun protection measures including:
- -
- Limit time in the sun, especially from 10 a.m. – 2 p.m.
- -
- Wear long-sleeve shirts, pants, hats and sunglasses
- Do not use on infants under 6 months old
- Other information
-
Inactive ingredients
Water/Aqua/Eau, Butylene Glycol, Butyloctyl Salicylate, Silica, C12-15 Alkyl Benzoate, Dimethicone, Glycerin, VP/Eicosene Copolymer, Glyceryl Stearate, PEG-100 Stearate, Potassium Cetyl Phosphate, Zingiber Officinale (Ginger) Root Oil, Camellia Sinensis Leaf Extract, Centella Asiatica Leaf Extract, Tocopheryl Acetate, Caprylyl Glycol, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Caprylic/Capric Triglyceride, Pentylene Glycol, Saccharide Isomerate, Bisabolol, Sclareolide, Arachidyl Glucoside, Arachidyl Alcohol, Xanthan Gum, Behenyl Alcohol, Ethylhexylglycerin, Trisodium Ethylenediamine Disuccinate, Sodium Hydroxide, Citric Acid, Propanediol, Dimethicone Crosspolymer, Sodium Citrate, Potassium Sorbate, Sodium Benzoate, Limonene.
- Questions or comments?
- PRINCIPAL DISPLAY PANEL - 50 mL Tube Carton
-
INGREDIENTS AND APPEARANCE
PRISMA PROTECT SPF30
octocrylene, homosalate, octisalate, and avobenzone lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68479-852 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octocrylene (UNII: 5A68WGF6WM) (Octocrylene - UNII:5A68WGF6WM) Octocrylene 10 g in 100 mL Homosalate (UNII: V06SV4M95S) (Homosalate - UNII:V06SV4M95S) Homosalate 7 g in 100 mL Octisalate (UNII: 4X49Y0596W) (Octisalate - UNII:4X49Y0596W) Octisalate 5 g in 100 mL Avobenzone (UNII: G63QQF2NOX) (Avobenzone - UNII:G63QQF2NOX) Avobenzone 3 g in 100 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Butylene Glycol (UNII: 3XUS85K0RA) Butyloctyl Salicylate (UNII: 2EH13UN8D3) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) Dimethicone (UNII: 92RU3N3Y1O) Glycerin (UNII: PDC6A3C0OX) VINYLPYRROLIDONE/EICOSENE COPOLYMER (UNII: 035MV9S1C3) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PEG-100 Stearate (UNII: YD01N1999R) Potassium Cetyl Phosphate (UNII: 03KCY6P7UT) Pentylene Glycol (UNII: 50C1307PZG) Propanediol (UNII: 5965N8W85T) Arachidyl Alcohol (UNII: 1QR1QRA9BU) Saccharide Isomerate (UNII: W8K377W98I) Caprylyl Glycol (UNII: 00YIU5438U) DOCOSANOL (UNII: 9G1OE216XY) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) LEVOMENOL (UNII: 24WE03BX2T) Sclareolide (UNII: 37W4O0O6E6) Xanthan Gum (UNII: TTV12P4NEE) Arachidyl Glucoside (UNII: 6JVW35JOOJ) Ethylhexylglycerin (UNII: 147D247K3P) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Trisodium Ethylenediamine Disuccinate (UNII: YA22H34H9Q) GINGER OIL (UNII: SAS9Z1SVUK) CENTELLA ASIATICA LEAF (UNII: 6810070TYD) Sodium Hydroxide (UNII: 55X04QC32I) GREEN TEA LEAF (UNII: W2ZU1RY8B0) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) Potassium Sorbate (UNII: 1VPU26JZZ4) Sodium Benzoate (UNII: OJ245FE5EU) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68479-852-02 1 in 1 CARTON 02/09/2024 1 50 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph drug M020 02/09/2024 Labeler - Dermalogica, LLC. (177698560) Establishment Name Address ID/FEI Business Operations Cosway 052400223 MANUFACTURE(68479-852) Establishment Name Address ID/FEI Business Operations McKenna 090631412 MANUFACTURE(68479-852)