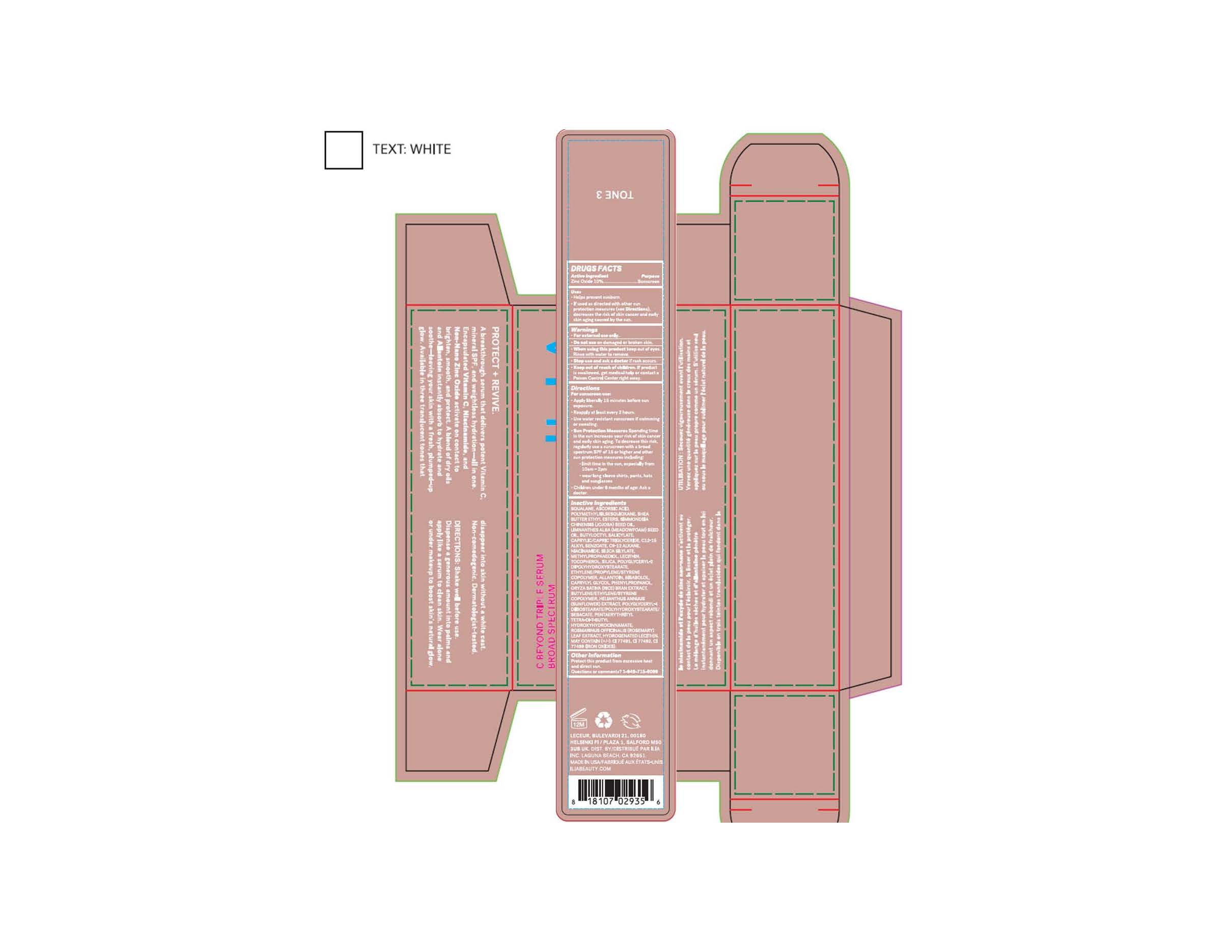
Label: C BEYOND TRIPLE SERUM (TONE 3)- zinc oxide lotion
- NDC Code(s): 81110-072-01, 81110-072-02, 81110-072-03, 81110-072-04
- Packager: Ilia, Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 7, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- C BEYOND TRIPLE SERUM (TONE 3)
-
INGREDIENTS AND APPEARANCE
C BEYOND TRIPLE SERUM (TONE 3)
zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81110-072 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 107 mg in 1 mL Inactive Ingredients Ingredient Name Strength LECITHIN, SOYBEAN (UNII: 1DI56QDM62) ALLANTOIN (UNII: 344S277G0Z) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SQUALANE (UNII: GW89575KF9) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) .ALPHA.-BISABOLOL, (+)- (UNII: 105S6I733Z) WATER (UNII: 059QF0KO0R) NIACINAMIDE (UNII: 25X51I8RD4) POLYMETHYLSILSESQUIOXANE (4.5 MICRONS) (UNII: 59Z907ZB69) PROPANEDIOL (UNII: 5965N8W85T) HELIANTHUS ANNUUS FLOWERING TOP (UNII: BKJ0J3D1BP) CAPRYLYL GLYCOL (UNII: 00YIU5438U) METHYLPROPANEDIOL (UNII: N8F53B3R4R) PHENYLPROPANOL (UNII: 0F897O3O4M) SHEA BUTTER (UNII: K49155WL9Y) TOCOPHEROL (UNII: R0ZB2556P8) SIMMONDSIA CHINENSIS SEED (UNII: D24K2Q1F6H) POLYGLYCERYL-2 DIPOLYHYDROXYSTEARATE (UNII: 9229XJ4V12) LIMNANTHES ALBA WHOLE (UNII: DKY81513ER) ROSMARINUS OFFICINALIS WHOLE (UNII: EA3289138M) ORYZA SATIVA WHOLE (UNII: 84IVV0906Z) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) POLYGLYCERYL-4 DIISOSTEARATE/POLYHYDROXYSTEARATE/SEBACATE (UNII: 687U3PEB2Y) HYDROGENATED SOYBEAN LECITHIN (UNII: H1109Z9J4N) FERRIC OXIDE RED (UNII: 1K09F3G675) Product Characteristics Color brown Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81110-072-02 1 in 1 CARTON 03/16/2022 1 NDC:81110-072-01 30 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 2 NDC:81110-072-04 1 in 1 CARTON 03/16/2022 2 NDC:81110-072-03 8 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 03/16/2022 Labeler - Ilia, Inc. (078503090) Registrant - Nanophase Technologies Corporation (623502044) Establishment Name Address ID/FEI Business Operations Nanophase Technologies Corporation 050383046 api manufacture(81110-072) Establishment Name Address ID/FEI Business Operations Nanophase Technologies Corporation 623502044 api manufacture(81110-072) , manufacture(81110-072) Establishment Name Address ID/FEI Business Operations Nanophase Technologies Corporation 118812921 manufacture(81110-072) , pack(81110-072)