Label: ARCH PACKAGING INSTANT GEL HAND SANITIZER- ethyl alcohol gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 79847-001-00, 79847-001-01 - Packager: Arch Packaging Technologies Llc
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 4, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
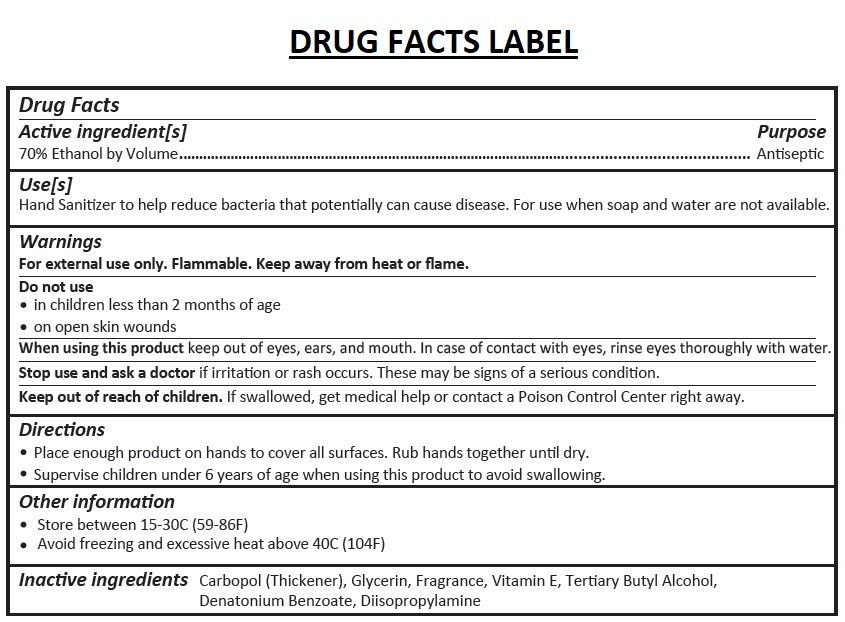
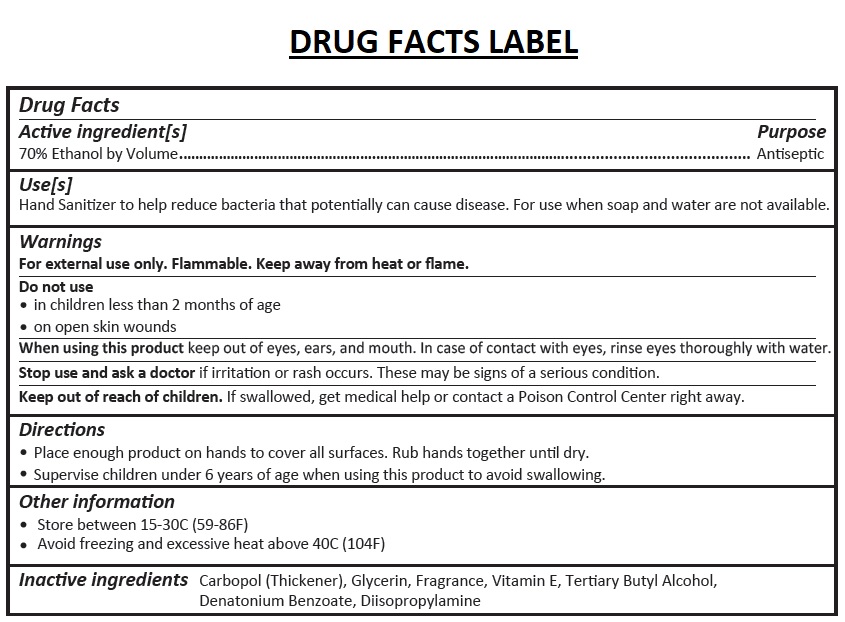
- Drug Facts
- Active ingredient[s]
- Purpose
- Use[s]
-
Warnings
For external use only. Flammable. Keep away from heat or flame.
Do not use
• in children less than 2 months of age
• on open skin wounds
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition. - Directions
- Other information
- Inactive ingredients
-
SPL UNCLASSIFIED SECTION
With Moisturizers and Vitamin E
Gentle Enough for Everyday Use
70% Ethyl Alcohol
DANGER: Highly Flammable liquid and vapor. Causes serious eye irritation. Keep container tightly closed. Keep out of reach of children. Keep away from heat/sparks/open flames/hot surfaces. No smoking. Use only non-sparking tools. Take precautionary measures against static discharge. May discolor certain fabrics or surfaces.
California SCAQMD Rule 443.1 VOC’S:
VOC: 70.50%IN CASE OF FIRE: Use dry sand, dry chemical or alcohol-resistant foam to extinguish. Dispose of contents/container in accordance with local/regional/national/international regulations.
See SDS for Additional InformationManufactured in St. Louis, MO
ARCH PACKAGING TECHNOLOGIES
10077 US HWY 61
Sainte Genevieve, MO 63670
www.archpackagingtechnologies.comMADE IN USA
- Packaging
-
INGREDIENTS AND APPEARANCE
ARCH PACKAGING INSTANT GEL HAND SANITIZER
ethyl alcohol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:79847-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 70 mL in 100 mL Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) GLYCERIN (UNII: PDC6A3C0OX) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) DENATONIUM BENZOATE (UNII: 4YK5Z54AT2) DIISOPROPYLAMINE (UNII: BR9JLI40NO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:79847-001-01 946 mL in 1 BOTTLE; Type 0: Not a Combination Product 09/04/2020 2 NDC:79847-001-00 3785 mL in 1 BOTTLE; Type 0: Not a Combination Product 09/04/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 09/04/2020 Labeler - Arch Packaging Technologies Llc (083005031)