Label: EQUATE PAIN RELIEVING PATCHES- lidocaine 4% patch
- NDC Code(s): 79903-106-06
- Packager: WALMART, INC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated March 15, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS
- PURPOSE
- USES
-
WARNINGS
For external use only
Do not use
- More than one patch on your body at a time
- On cut, irritated or swollen skin
- On puncture wounds
- For more than one week without consulting a doctor
- If you are allergic to any active or inactive ingredients
- If pouch is damaged or opened
When using this product
- Use only as directed
- Read and follow all directions and warnings on this pack
- Do not allow contact with the eyes
- Do not use at the same time as other topical analgesics
- Do not bandage tightly or apply local heat (such as heating pads) to the area of use
- Do not microwave
- Dispose of used patch in manner that always keeps product away from children and pets
- Used patches still contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS
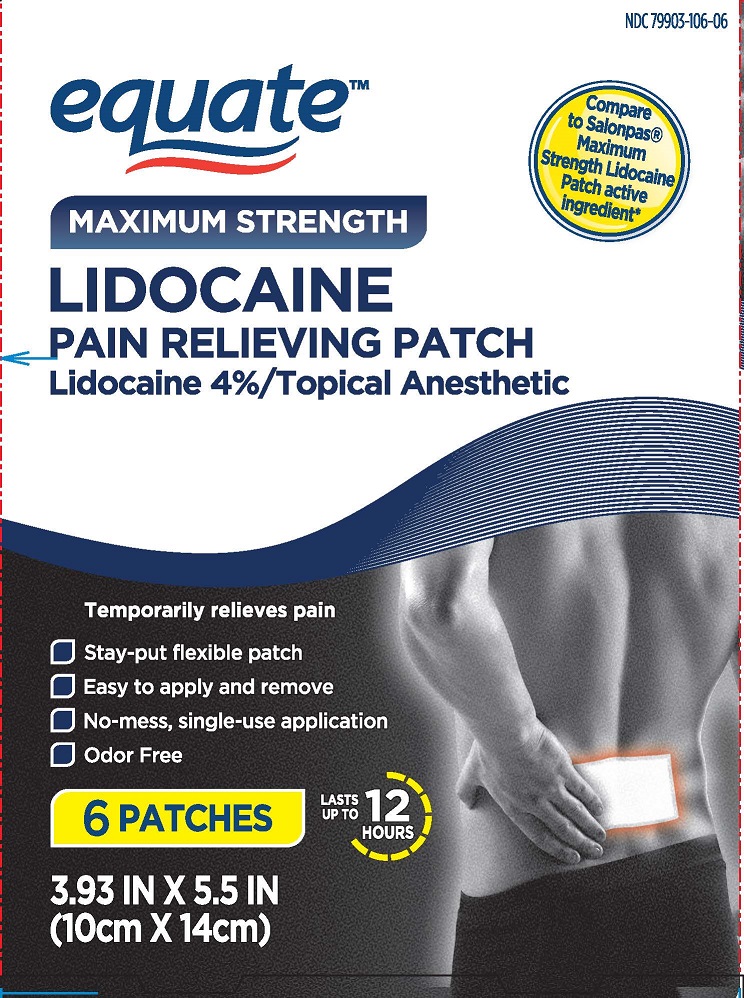
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EQUATE PAIN RELIEVING PATCHES
lidocaine 4% patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:79903-106 Route of Administration TRANSDERMAL, TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 344 mg Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) POVIDONE (UNII: FZ989GH94E) KAOLIN (UNII: 24H4NWX5CO) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WATER (UNII: 059QF0KO0R) PROPYLPARABEN (UNII: Z8IX2SC1OH) SODIUM POLYACRYLATE (8000 MW) (UNII: 285CYO341L) POLYACRYLIC ACID (250000 MW) (UNII: 9G2MAD7J6W) POLYSORBATE 80 (UNII: 6OZP39ZG8H) DIHYDROXYALUMINUM AMINOACETATE (UNII: DO250MG0W6) EDETATE DISODIUM (UNII: 7FLD91C86K) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) METHYLPARABEN (UNII: A2I8C7HI9T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:79903-106-06 6 in 1 BOX 04/01/2022 1 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 04/01/2022 Labeler - WALMART, INC (051957769) Registrant - GURUNANDA, LLC (079671169)