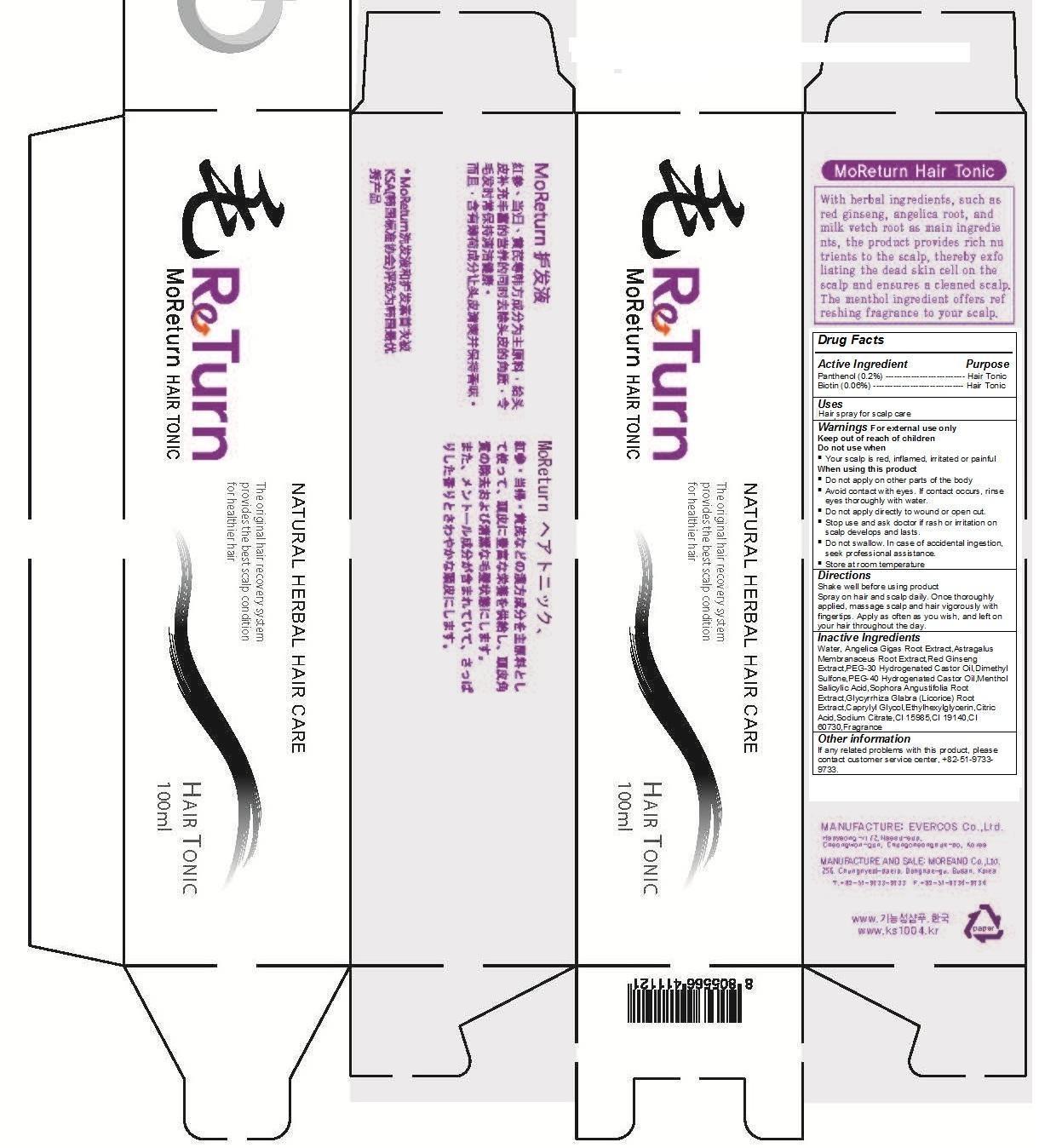
Label: MORETURN HAIR TONIC (panthenol (0.2%), biotin- 0.06% spray
-
Contains inactivated NDC Code(s)
NDC Code(s): 61310-100-01, 61310-100-02 - Packager: Moreand Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated December 3, 2013
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- MoReturn Hair Tonic
- Hair tonic Scalp care
- Keep out of reach of children
- Spray on hair and scalp daily. Once thoroughly applied, massage scalp and hair vigorously with fingertips. Apply as often as you wish, and left on your hair throughout the day.
- For external use only
- Spray on hair and scalp daily.
- Water, Angelica Gigas Root Extract,Astragalus Membranaceus Root Extract,Red Ginseng Extract,PEG-30 Hydrogenated Castor Oil,Dimethyl Sulfone,PEG-40 Hydrogenated Castor Oil,MentholSalicylic Acid,Sophora Angustifolia Root Extract,Glycyrrhiza Glabra (Licorice) Root Extract,Caprylyl Glycol,Ethylhexylglycerin,Citric Acid,Sodium Citrate,CI 15985,CI 19140,CI 60730,Fragrance
- Panthenol (0.2%) Biotin (0.06%)
-
INGREDIENTS AND APPEARANCE
MORETURN HAIR TONIC
panthenol (0.2%), biotin (0.06%) sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:61310-100 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PANTHENOL (UNII: WV9CM0O67Z) (PANTHENOL - UNII:WV9CM0O67Z) PANTHENOL 0.2 g in 100 mL BIOTIN (UNII: 6SO6U10H04) (BIOTIN - UNII:6SO6U10H04) BIOTIN 0.06 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ANGELICA GIGAS ROOT (UNII: 32766B2FHX) ASTRAGALUS PROPINQUUS ROOT (UNII: 922OP8YUPF) ASIAN GINSENG (UNII: CUQ3A77YXI) PEG-30 CASTOR OIL (UNII: GF873K38RZ) DIMETHYL SULFONE (UNII: 9H4PO4Z4FT) PEG-40 CASTOR OIL (UNII: 4ERD2076EF) MENTHOL (UNII: L7T10EIP3A) SALICYLIC ACID (UNII: O414PZ4LPZ) SOPHORA FLAVESCENS ROOT (UNII: IYR6K8KQ5K) GLYCYRRHIZA GLABRA (UNII: 2788Z9758H) CAPRYLYL GLYCOL (UNII: 00YIU5438U) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) CITRIC ACID ACETATE (UNII: DSO12WL7AU) SODIUM CITRATE (UNII: 1Q73Q2JULR) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) EXT. D&C VIOLET NO. 2 (UNII: G5UX3K0728) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61310-100-02 1 in 1 CARTON 1 NDC:61310-100-01 100 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 12/20/2013 Labeler - Moreand Co., Ltd. (688731001) Registrant - Moreand Co., Ltd. (688731001) Establishment Name Address ID/FEI Business Operations Moreand Co., Ltd. 688731001 manufacture(61310-100)