Label: MAX RELIEF JUNIOR- children acetaminophen suspension
- NDC Code(s): 71399-0038-4, 71399-0038-6, 71399-0038-8
- Packager: Akron Pharma Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated April 28, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
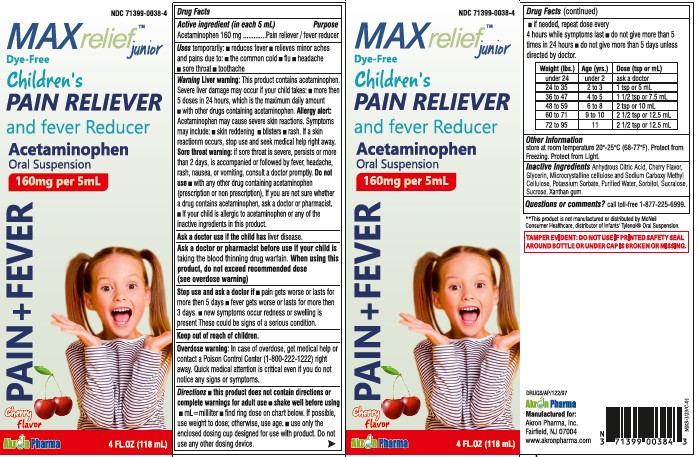
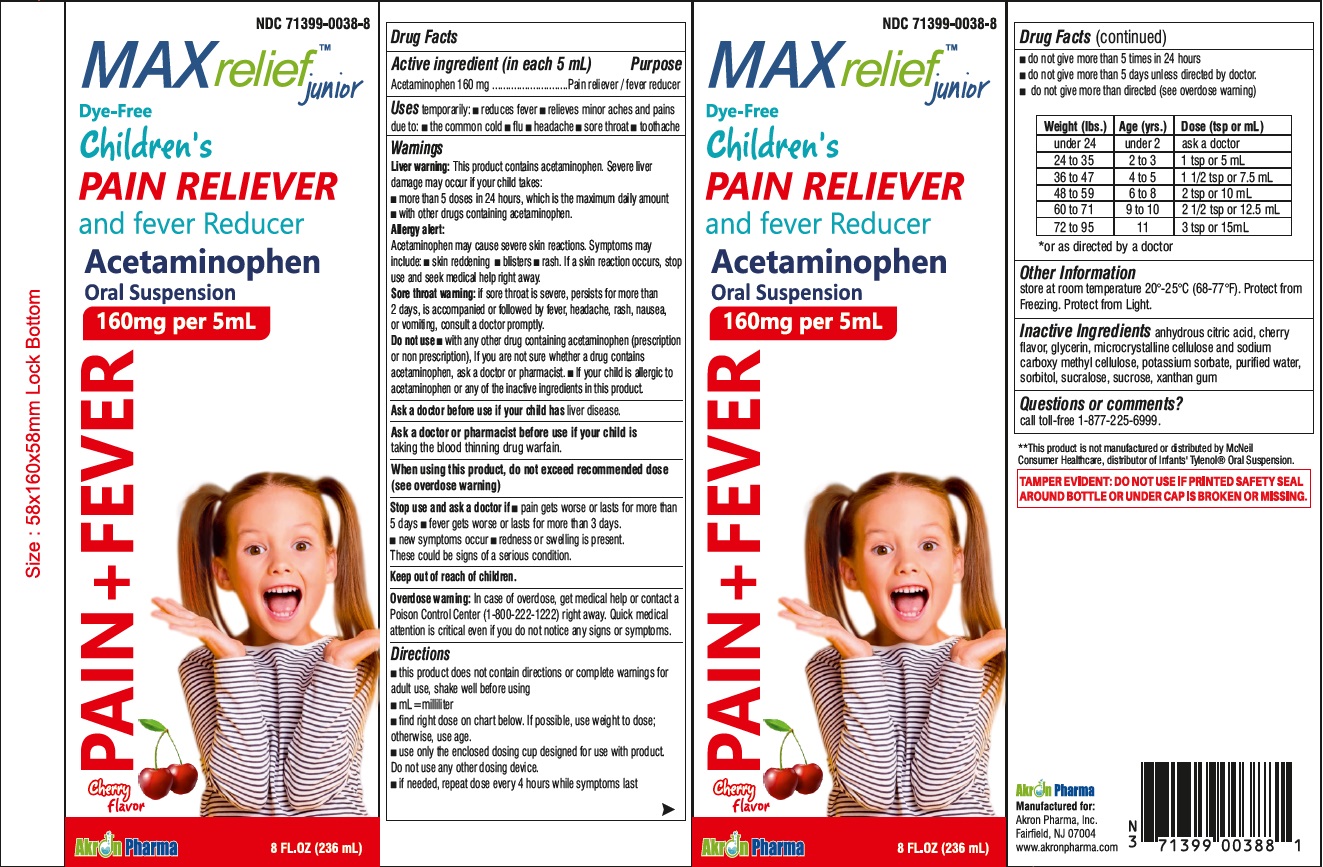
- Active Ingredient
- Uses
-
Warnings
Liver Warning: This product contains acetaminophen. Severe liver damage may occur if your child takes:
- more then 5 doses in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning:
if sore throat is severe, persist or more than 2 days, is accompanied or followed by fever, headache, rash, nausea or vomiting, consult a doctor promptly.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription).
- If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if your child is allergic to acetaminophen or anyof the inactive ingredients in this product.
-
Directions
- this product does not contain directions or complete warnings for adult use
- shake well before using
- mL = milliliter
- find right dose on chart below. if possible, use weight to dose, otherwise use age
- use only the enclosed dosing cup designed for use with product.
Weight (lbs) Age (yrs.) Dose (tsp or mL) Weight (lbs) Age (yrs.) Dose (tsp or mL) under 24 under 2 ask a doctor 48 to 59 6 to 8 2 tsp or 10 mL 25 to 35 2 to 3 1 tsp or 5 mL 60 to 71 9 to 10 2 1/2 tsp or 12.5 mL 36 to 47 4 to 5 1 1/2 tsp or 7.5 mL 72 to 95 11 2 1/2 tsp or 12.5 mL - Other Information
- Inactive Ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MAX RELIEF JUNIOR
children acetaminophen suspensionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71399-0038 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 160 mg in 5 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) GLYCERIN (UNII: PDC6A3C0OX) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) SUCRALOSE (UNII: 96K6UQ3ZD4) SUCROSE (UNII: C151H8M554) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color Score Shape Size Flavor CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71399-0038-4 1 in 1 CARTON 03/18/2024 1 118 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:71399-0038-8 1 in 1 CARTON 04/24/2024 2 236 mL in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:71399-0038-6 473 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/24/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 03/18/2024 Labeler - Akron Pharma Inc. (067878881)