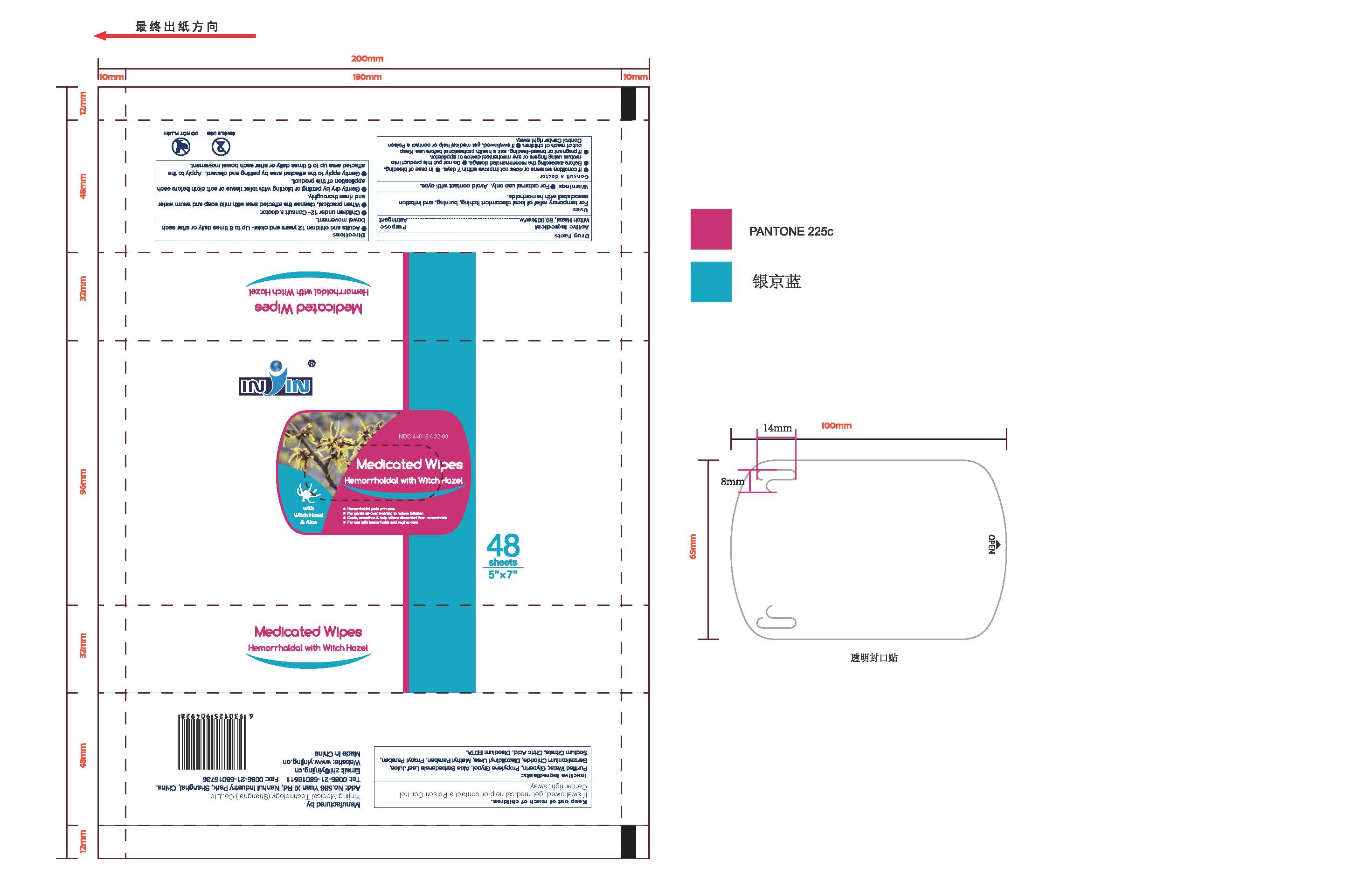
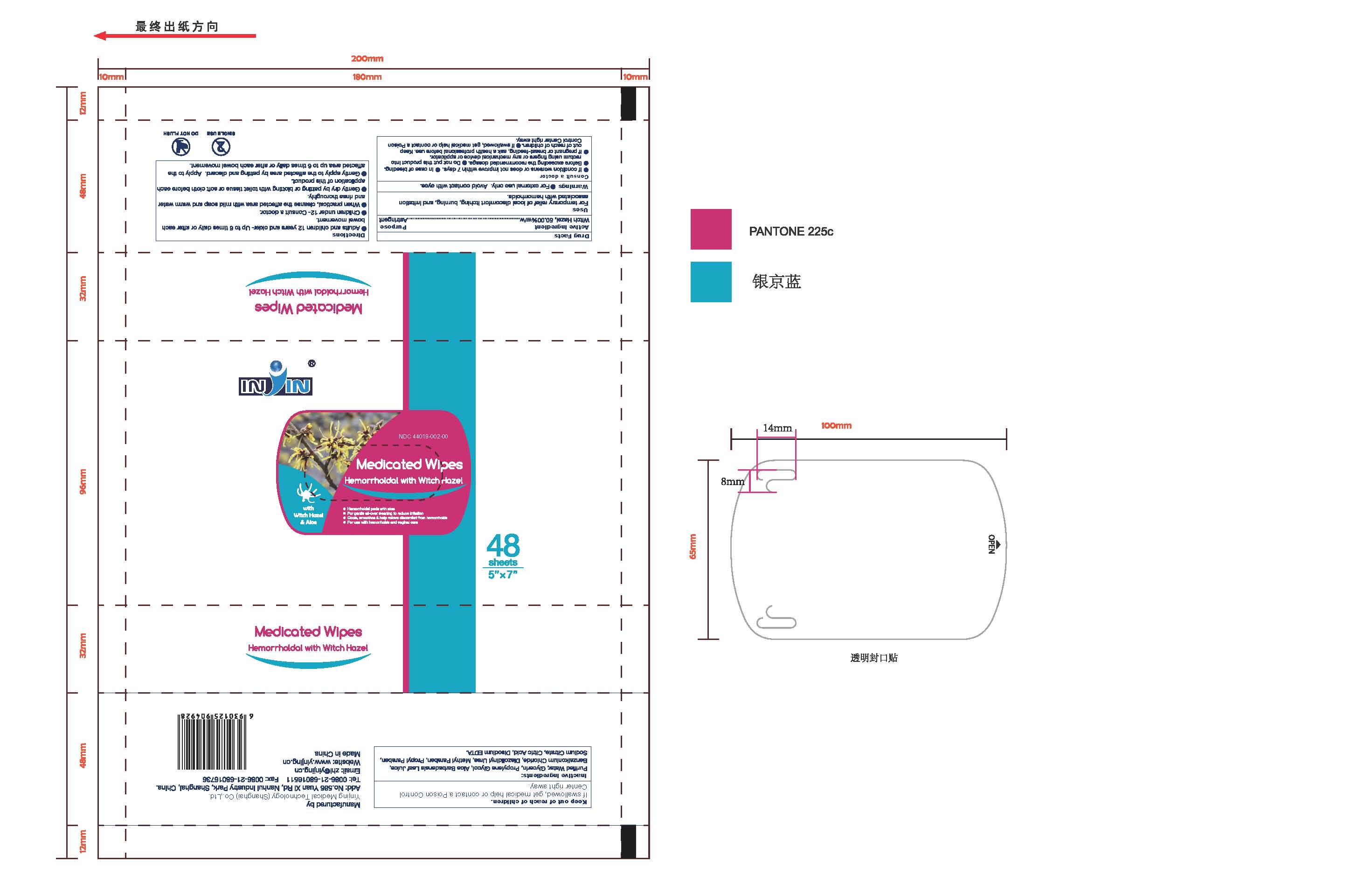
Label: FLUSHABLE MEDICATED WIPES HEMORRHOIDAL PADS WITH WITCH HAZEL AND ALOE- witch hazel liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 44019-002-00 - Packager: Yinjing Medical Technology (Shanghai) Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 27, 2017
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Flushable Medicated Wipes Hemorrhoidal Pads with Witch Hazel and Aloe
- Active ingredient
- Uses
-
Warnings
For external use only. Avoid contact with eyes.
-
Directions
- Adults and children 12 years and older- Up to 6 times daily or after each bowel movement ? Children under 12- Consult a doctor ? When practical, cleanse the affected area with mild soap and warm water and rinse thoroughly. ? Gently dry by patting or blotting with toilet tissue or soft cloth before each application of this product. ? Gently apply to the affected area by patting and discard. Apply to the affected area up to 6 times daily or after each bowel movement
- Other information
- Inactive Ingredient
- PRINCIPAL DISPLAY PANEL
- Flushable Medicated Wipes Hemorrhoidal Pads with Witch Hazel and Aloe 48 Wipes (44019-002-00)
-
INGREDIENTS AND APPEARANCE
FLUSHABLE MEDICATED WIPES HEMORRHOIDAL PADS WITH WITCH HAZEL AND ALOE
witch hazel liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:44019-002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WITCH HAZEL (UNII: 101I4J0U34) (WITCH HAZEL - UNII:101I4J0U34) WITCH HAZEL 50 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) ALOE VERA LEAF (UNII: ZY81Z83H0X) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) SODIUM CITRATE (UNII: 1Q73Q2JULR) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) EDETATE DISODIUM (UNII: 7FLD91C86K) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:44019-002-00 48 in 1 BAG 08/03/2016 1 2.9 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part346 07/08/2013 Labeler - Yinjing Medical Technology (Shanghai) Co., Ltd. (530501535) Registrant - Yinjing Medical Technology (Shanghai) Co., Ltd. (530501535)