Label: TRIHEXYPHENIDYL HYDROCHLORIDE tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 53808-0633-1, 53808-0634-1 - Packager: State of Florida DOH Central Pharmacy
- This is a repackaged label.
- Source NDC Code(s): 0603-6240, 0603-6241
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated October 1, 2013
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Trihexyphenidyl HCl is a synthetic antispasmodic. Each tablet for oral administration, contains 2 mg or 5 mg trihexyphenidyl HCl, each strength also containing as inactive ingredients: magnesium stearate, microcrystalline cellulose, and sodium starch glycolate.
Trihexyphenidyl HCl is a white or slightly off white, crystalline powder, having not more than a very faint odor.
Trihexyphenidyl HCl is the substituted piperidine salt, 1-piperidinepropanol,α-cyclohexyl-α-phenyl-,hydrochloride,(±)-. The structural formula is:
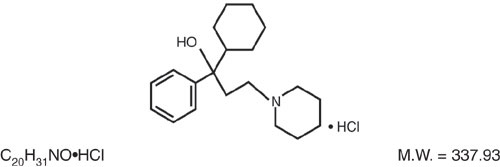
-
CLINICAL PHARMACOLOGY
Trihexyphenidyl exerts a direct inhibitory effect upon the parasympathetic nervous system. It also has a relaxing effect on smooth musculature; exerted both directly upon the muscle tissue itself and indirectly through an inhibitory effect upon the parasympathetic nervous system. Its therapeutic properties are similar to those of atropine, although undesirable side effects are ordinarily less frequent and severe than with the latter.
-
INDICATIONS AND USAGE
Trihexyphenidyl HCl tablets are indicated as an adjunct in the treatment of all forms of parkinsonism (postencephalitic, arteriosclerotic, and idiopathic). It is often useful as adjuvant therapy when treating these forms of parkinsonism with levodopa. Additionally, it is indicated for the control of extrapyramidal disorders caused by central nervous system drugs such as the dibenzoxazepines, phenothiazines, thioxanthenes, and butyrophenones.
- WARNINGS
-
PRECAUTIONS
Although trihexyphenidyl HCl is not contraindicated for patients with cardiac, liver, or kidney disorders, or with hypertension, such patients should be maintained under close observation.
Since the use of trihexyphenidyl HCl may, in some cases, continue indefinitely and since it has atropine-like properties, patients should be subjected to constant and careful long-term observation to avoid allergic and other untoward reactions. Inasmuch as trihexyphenidyl HCl possesses some parasympatholytic activity, it should be used with caution in patients with glaucoma, obstructive disease of the gastrointestinal or genitourinary tracts, and in elderly males with possible prostatic hypertrophy. Geriatric patients, particularly over the age of 60, frequently develop increased sensitivity to the actions of drugs of this type, and hence, require strict dosage regulation. Incipient glaucoma may be precipitated by parasympatholytic drugs such as trihexyphenidyl HCl.
Tardive dyskinesia may appear in some patients on long-term therapy with antipsychotic drugs or may occur after therapy with these drugs has been discontinued. Antiparkinsonism agents do not alleviate the symptoms of tardive dyskinesia and, in some instances, may aggravate them. However, parkinsonism and tardive dyskinesia often coexist in patients receiving chronic neuroleptic treatment, and anticholinergic therapy with trihexyphenidyl HCl may relieve some of these parkinsonism symptoms.
-
ADVERSE REACTIONS
Minor side effects, such as dryness of the mouth, blurring of vision, dizziness, mild nausea or nervousness, will be experienced by 30 to 50 percent of all patients. These sensations, however, are much less troublesome with trihexyphenidyl HCl than with belladonna alkaloids and are usually less disturbing than unalleviated parkinsonism. Such reactions tend to become less pronounced, and even to disappear, as treatment continues. Even before these reactions have remitted spontaneously, they may often be controlled by careful adjustment of dosage form, amount of drug, or interval between doses.
Isolated instances of suppurative parotitis secondary to excessive dryness at the mouth, skin rashes, dilatation of the colon, paralytic ileus, and certain psychiatric manifestations such as delusions and hallucinations, plus one doubtful case of paranoia all of which may occur with any of the atropine-like drugs, have been reported rarely with trihexyphenidyl hydrochloride.
Patients with arteriosclerosis or with a history of idiosyncrasy to other drugs may exhibit reactions of mental confusion, agitation, disturbed behavior, or nausea and vomiting. Such patients should be allowed to develop a tolerance through the initial administration of a small dose and gradual increase in dose until an effective level is reached. If a severe reaction should occur, administration of the drug should be discontinued for a few days and then resumed at a lower dosage. Psychiatric disturbances can result from indiscriminate use (leading to overdosage) to sustain continued euphoria.
Potential side effects associated with the use of any atropine-like drugs include constipation, drowsiness, urinary hesitancy or retention, tachycardia, dilation of the pupil, increased intraocular tension, weakness, vomiting, and headache.
The occurrence of angle-closure glaucoma due to long-term treatment with trihexyphenidyl has been reported.
-
DOSAGE AND ADMINISTRATION
Dosage should be individualized. The initial dose should be low and then increased gradually, especially in patients over 60 years of age. Whether trihexyphenidyl HCl may best be given before or after meals should be determined by the way the patient reacts. Postencephalitic patients, who are usually more prone to excessive salivation, may prefer to take it after meals and may, in addition, require small amounts of atropine which, under such circumstances, is sometimes an effective adjuvant. If trihexyphenidyl HCl tends to dry the mouth excessively, it may be better to take it before meals, unless it causes nausea. If taken after meals, the thirst sometimes induced can be allayed by mint candies, chewing gum or water.
Trihexyphenidyl HCl in Idiopathic Parkinsonism
As initial therapy for parkinsonism, 1 mg of trihexyphenidyl in tablet form may be administered the first day. The dose may then be increased by 2 mg increments at intervals of three to five days, until a total of 6 to 10 mg is given daily. The total daily dose will depend upon what is found to be the optimal level. Many patients derive maximum benefit from this daily total of 6 to 10 mg, but some patients, chiefly those in the postencephalitic group, may require a total daily dose of 12 to 15 mg.
Trihexyphenidyl HCl in Drug-Induced Parkinsonism
The size and frequency of dose of trihexyphenidyl HCl needed to control extrapyramidal reactions to commonly employed tranquilizers, notably the phenothiazines, thioxanthenes, and butyrophenones, must be determined empirically. The total daily dosage usually ranges between 5 and 15 mg, although, in some cases, these reactions have been satisfactorily controlled on as little as 1 mg daily. It may be advisable to commence therapy with a single 1 mg dose. If the extrapyramidal manifestations are not controlled in a few hours, the subsequent doses may be progressively increased until satisfactory control is achieved. Satisfactory control may sometimes be more rapidly achieved by temporarily reducing the dosage of the tranquilizer on instituting trihexyphenidyl HCl therapy and then adjusting dosage of both drugs until the desired ataractic effect is retained without onset of extrapyramidal reactions.
It is sometimes possible to maintain the patient on a reduced trihexyphenidyl HCl dosage after the reactions have remained under control for several days. Instances have been reported in which these reactions have remained in remission for long periods after trihexyphenidyl HCl therapy was discontinued.
Concomitant Use of Trihexyphenidyl HCl with Levodopa
When trihexyphenidyl HCl is used concomitantly with levodopa, the usual dose of each may need to be reduced. Careful adjustment is necessary, depending on side effects and degree of symptom control. Trihexyphenidyl HCl dosage of 3 to 6 mg daily, in divided doses, is usually adequate.
Concomitant Use of Trihexyphenidyl HCl with Other Parasympathetic Inhibitors
Trihexyphenidyl HCl may be substituted, in whole or in part, for other parasympathetic inhibitors. The usual technique is partial substitution initially, with progressive reduction in the other medication as the dose of trihexyphenidyl HCl is increased.
The total daily intake of trihexyphenidyl HCl tablets is tolerated best if divided into 3 doses and taken at mealtimes. High doses (>10 mg daily) may be divided into 4 parts, with 3 doses administered at mealtimes and the fourth at bedtime.
-
HOW SUPPLIED
Trihexyphenidyl HCl tablets are available as follows:
2 mg – round, flat, scored, white tablets; debossed "5971" above the score and "V" below the score.
5 mg – round, flat, scored, white tablets; debossed "5972" above the score and "V" below the score.
They are supplied by State of Florida DOH Central Pharmacy as follows:
NDC Strength Quantity/Form Color Source Prod. Code 53808-0633-1 2 mg 30 Tablets in a Blister Pack WHITE 0603-6240 53808-0634-1 5 mg 30 Tablets in a Blister Pack WHITE 0603-6241 - STORAGE AND HANDLING
- SPL UNCLASSIFIED SECTION
- PACKAGE LABEL
- PACKAGE LABEL
-
INGREDIENTS AND APPEARANCE
TRIHEXYPHENIDYL HYDROCHLORIDE
trihexyphenidyl hydrochloride tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:53808-0633(NDC:0603-6240) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRIHEXYPHENIDYL HYDROCHLORIDE (UNII: AO61G82577) (TRIHEXYPHENIDYL - UNII:6RC5V8B7PO) TRIHEXYPHENIDYL HYDROCHLORIDE 2 mg Inactive Ingredients Ingredient Name Strength MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE Score 2 pieces Shape ROUND Size 8mm Flavor Imprint Code 5971;V Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53808-0633-1 30 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040254 01/01/2013 TRIHEXYPHENIDYL HYDROCHLORIDE
trihexyphenidyl hydrochloride tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:53808-0634(NDC:0603-6241) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRIHEXYPHENIDYL HYDROCHLORIDE (UNII: AO61G82577) (TRIHEXYPHENIDYL - UNII:6RC5V8B7PO) TRIHEXYPHENIDYL HYDROCHLORIDE 5 mg Inactive Ingredients Ingredient Name Strength MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE Score 2 pieces Shape ROUND Size 10mm Flavor Imprint Code 5972;V Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53808-0634-1 30 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040254 01/01/2013 Labeler - State of Florida DOH Central Pharmacy (829348114) Establishment Name Address ID/FEI Business Operations State of Florida DOH Central Pharmacy 829348114 repack(53808-0633, 53808-0634)




