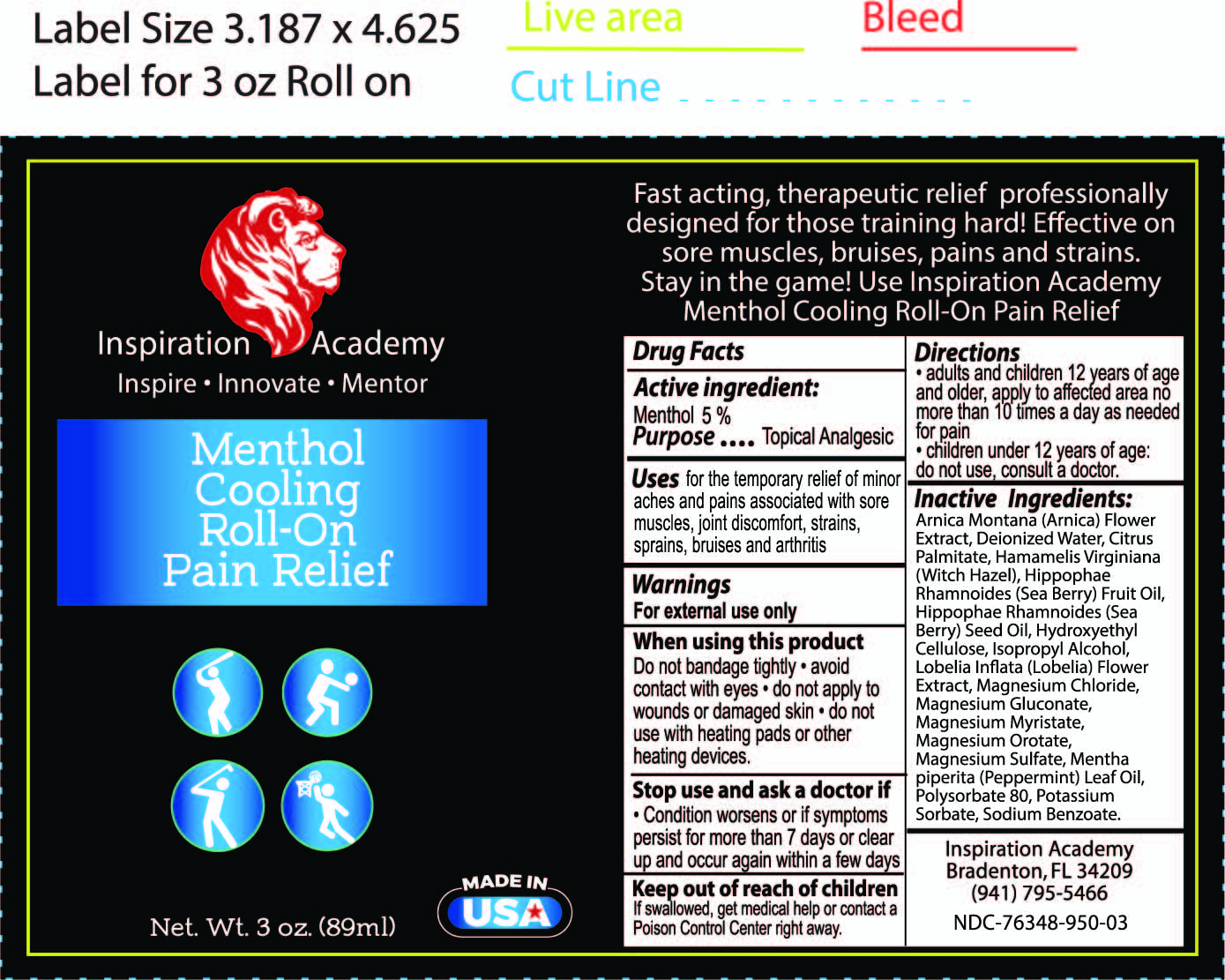
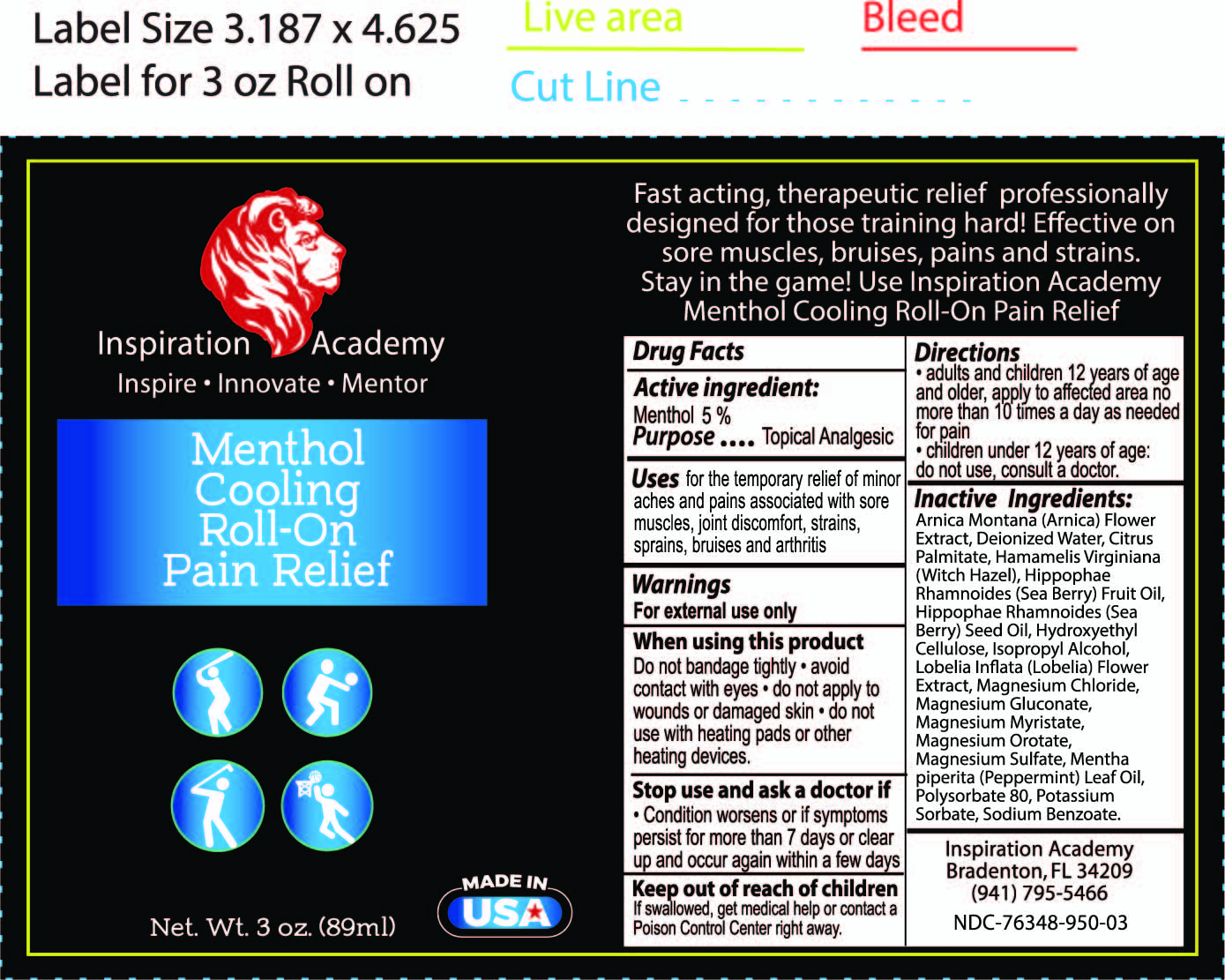
Label: INSPIRATION ACADEMY MENTHOL COOLING ROLL ON- menthol liquid
- NDC Code(s): 76348-950-01, 76348-950-03
- Packager: RENU LABORATORIES, INC.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 14, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- WARNINGS
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
-
INACTIVE INGREDIENT
Arnica Montana (Arnica) Flower Extract, Cannabis Sativa (Hemp) Seed Oil and Hemp Derived Cannabidiol Extract with proprietary terpene blend (Limonene, Myrcene, Caryophyllene, Linalool, Pinene, Terpineol), Deionized Water, Fragrance, Hamamelis Virginiana (Witch Hazel), Hydroxyethyl Cellulose, Isopropyl Alcohol, Magnesium Chloride, Mentha piperita (Peppermint) Leaf Oil, Polysorbate 80, Potassium Sorbate, Sodium Benzoate.
- QUESTIONS
- INDICATIONS & USAGE
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
INSPIRATION ACADEMY MENTHOL COOLING ROLL ON
menthol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76348-950 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 4.2 g in 84 g Inactive Ingredients Ingredient Name Strength POLYSORBATE 80 (UNII: 6OZP39ZG8H) METHYLCELLULOSE (4000 CPS) (UNII: MRJ667KA5E) MAGNESIUM SULFATE, UNSPECIFIED (UNII: DE08037SAB) PEPPERMINT OIL (UNII: AV092KU4JH) WATER (UNII: 059QF0KO0R) WITCH HAZEL (UNII: 101I4J0U34) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) ARNICA MONTANA (UNII: O80TY208ZW) SODIUM BENZOATE (UNII: OJ245FE5EU) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) ISOPROPYL ALCOHOL (UNII: ND2M416302) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76348-950-01 1 in 1 BOX 10/11/2021 1 NDC:76348-950-03 84 g in 1 APPLICATOR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 10/11/2021 Labeler - RENU LABORATORIES, INC. (945739449) Registrant - RENU LABORATORIES, INC. (945739449) Establishment Name Address ID/FEI Business Operations RENU LABORATORIES, INC. 945739449 manufacture(76348-950)