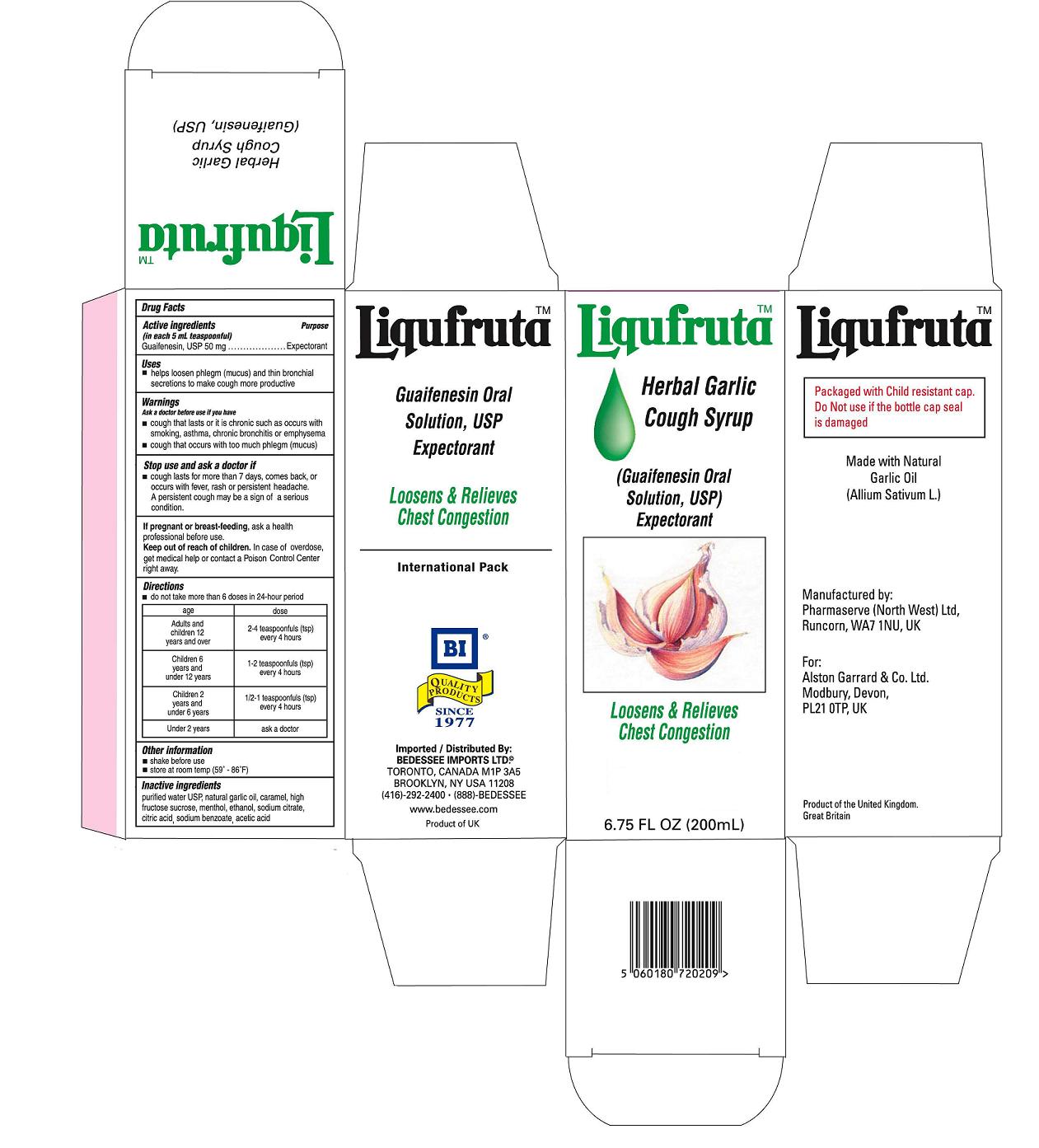
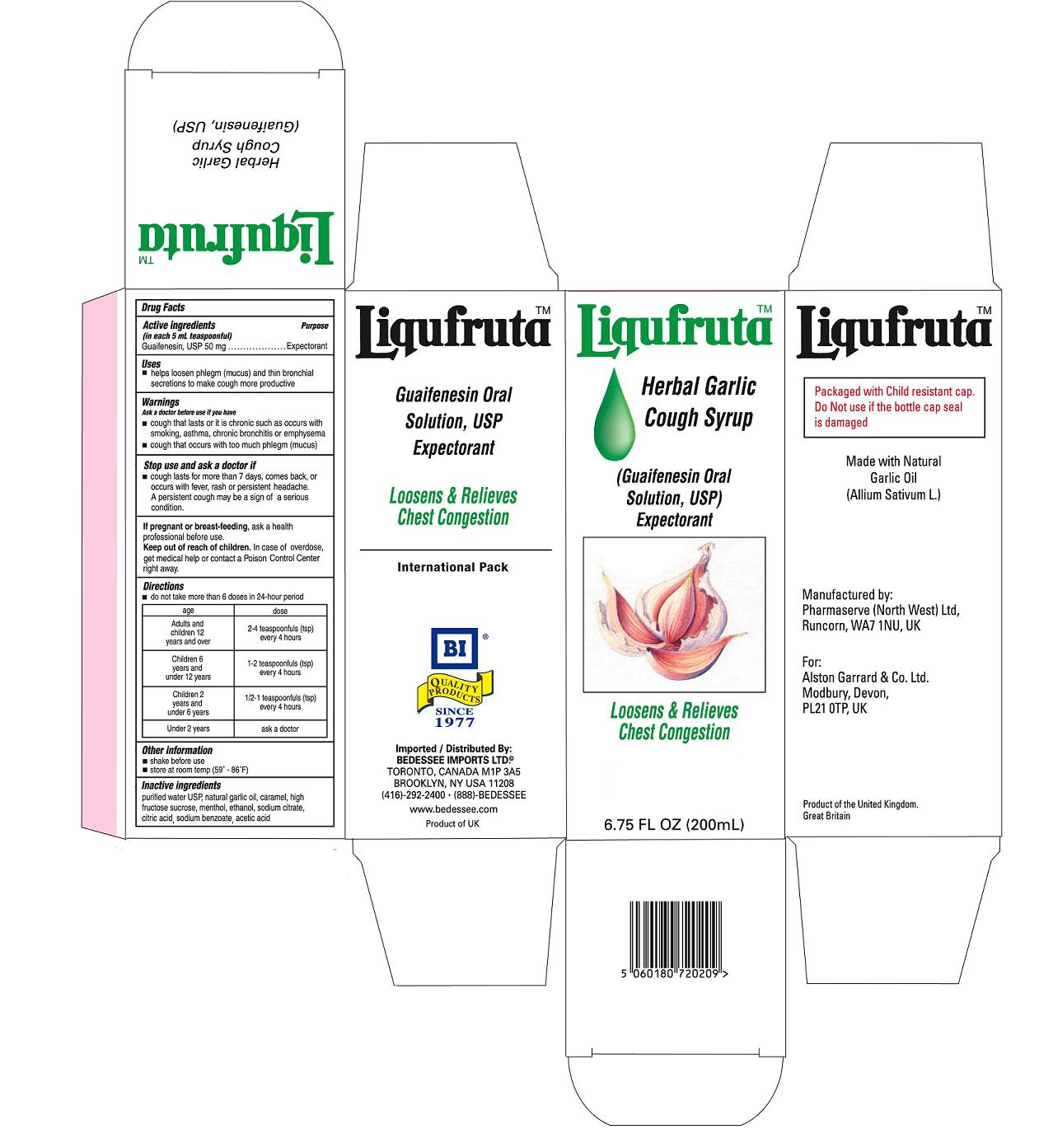
Label: LIQUFRUTA- guaifenesin syrup
-
Contains inactivated NDC Code(s)
NDC Code(s): 75939-9876-2 - Packager: Alston Garrard & Co
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 30, 2012
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
WARNINGS
Ask a doctor before use if you have a cough that last or it is chronic such as occurs with smoking, asthma, chronic Bronchitis or emphysema. Stop use and ask doctor if a cough lasts for more than 7 days comes back or occurs when fever, rash or persistent headache. A persistent cough may be a sign of a serious condition.
If pregnant of breast-feeding, ask a health professional before use.
-
DOSAGE & ADMINISTRATION
do not take more than 6 doses in a 24-hour period
AGE ......................................................DOSE
Adults and Children 12 years and over .......2-4 teaspoonfuls(tsp) every 4 hours
Children 6 years and under 12 years......... 1-2 teaspoonfuls(tsp) every 4 hours
Children 2 years and under 6 years........... 1/2-1 teaspoonfuls(tsp) every 4 hours
Under 2 years ........................................ask a doctor
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LIQUFRUTA
guaifenesin syrupProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:75939-9876 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 50 mg in 5 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) SODIUM CITRATE (UNII: 1Q73Q2JULR) GARLIC OIL (UNII: 4WG8U28833) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) HIGH FRUCTOSE CORN SYRUP (UNII: XY6UN3QB6S) SODIUM BENZOATE (UNII: OJ245FE5EU) CARAMEL (UNII: T9D99G2B1R) MENTHOL (UNII: L7T10EIP3A) ACETIC ACID (UNII: Q40Q9N063P) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:75939-9876-2 200 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 01/01/2011 Labeler - Alston Garrard & Co (735697570) Registrant - Alston Garrard & Co (735697570) Establishment Name Address ID/FEI Business Operations Pharmaserve (North West) Ltd 210141282 manufacture