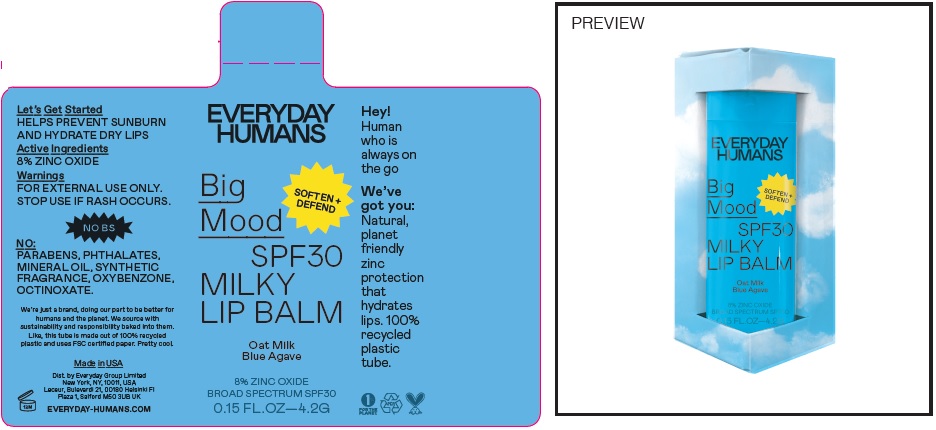
Label: BIG MOOD SPF 30 MILKY LIP BALM- zinc oxide stick
- NDC Code(s): 72098-004-01
- Packager: Everyday Group Limited
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 9, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients
- Uses
- Warnings
-
Directions
- Apply liberally 15 minutes before sun exposure
- Use a water resistant sunscreen if swimming or sweating
- Reapply at least every 2 hours
- Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease the risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including: Sun Protection Measures:
- Limit time in the sun, especially from 10 a.m to 2 p.m.
- Wear long-sleeved shirts, pants, hats, and sunglasses
- Children under 6 months of age: Ask a doctor
-
Inactive Ingredients
Prunus Amygdalus Dulcis (Sweet Almond) Oil, Cera Alba (Beeswax), Simmondsia Chinensis (Jojoba) Seed Oil, Butyrospermum Parkii (Shea) Butter, Euphorbia Cerifera (Candelilla) Wax, Flavor, Argania Spinosa Kernal (Argan) Oil, Caprylic/capric Triglyceride, Tocopheryl Acetate, Copernicia Cerifera (Carnauba) Wax, Avena Sativa Kernel (Oat) Oil, Agave Tequilana Stem Extract, Bisabolol, Helianthus Annuus (Sunflower) Seed Oil, Tocopherol, Stevia Rebaudiana Extract, Glycine Soja (Soybean) Oil, Glyceryl Caprylate, Polyhydroxystearic Acid, Polyglyceryl-3-Polyricinoleate, Isostearic Acid, Lecithin, Glyceryl Undecylenta
- Other Information
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
BIG MOOD SPF 30 MILKY LIP BALM
zinc oxide stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72098-004 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 80 mg in 1 g Inactive Ingredients Ingredient Name Strength TRICAPRYLIN (UNII: 6P92858988) ARGAN OIL (UNII: 4V59G5UW9X) YELLOW WAX (UNII: 2ZA36H0S2V) ALMOND OIL (UNII: 66YXD4DKO9) JOJOBA OIL (UNII: 724GKU717M) SHEA BUTTER (UNII: K49155WL9Y) CANDELILLA WAX (UNII: WL0328HX19) METHYL SALICYLATE (UNII: LAV5U5022Y) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) CARNAUBA WAX (UNII: R12CBM0EIZ) OAT (UNII: Z6J799EAJK) LEVOMENOL (UNII: 24WE03BX2T) SUNFLOWER OIL (UNII: 3W1JG795YI) TOCOPHEROL (UNII: R0ZB2556P8) SOYBEAN OIL (UNII: 241ATL177A) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) POLYGLYCERYL-3 PENTARICINOLEATE (UNII: 7Q0OK5DOT4) ISOSTEARIC ACID (UNII: X33R8U0062) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72098-004-01 1 in 1 CARTON 12/01/2021 1 4.2 g in 1 APPLICATOR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 12/01/2021 Labeler - Everyday Group Limited (664624682)