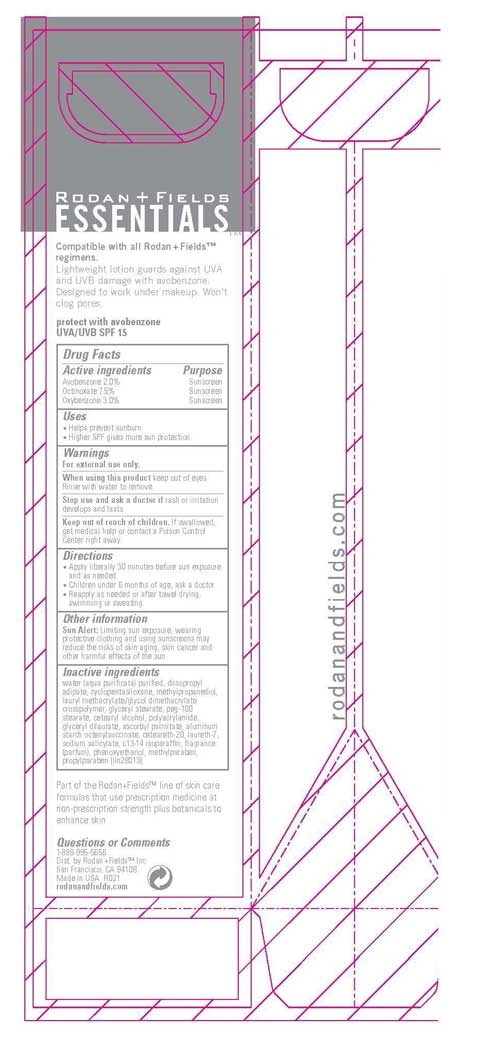
Label: RODAN AND FIELDS ESSENTIALS UVA/UVB- avobenzone, octinoxate, oxybenzone cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 56152-0815-1, 56152-0815-2 - Packager: Cosmetic Enterprises Ltd.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 28, 2011
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active Ingredients Purpose
Avobenzone 2.0% Sunscreen
Octinoxate 7.5% Sunscreen
Oxybenzone 3.0 % Sunscreen
Warnings
For external use only.
When using this product keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if rash or irritation develops and lasts
Keep out of of children. If swallowed, get medical help or contact a Poison Control Center right away.
water (aqua purificata) purified, diisopropyl adipate, cyclopentasiloxane, methylpropanediol, lauryl methacrylate/glycol dimethacrylate crosspolymer, glyceryl stearate, peg-100 stearate, cetearyl alcohol, polyacrylamide, glyceryl dilaurate, ascorbyl palmitate, aluminum starch octenylsuccinate, ceteareth-20, laureth-7, sodium salicylate, c13-14 isoparaffin, fragrance (parfum), phenoxyethanol, methylparaben, propylparaben
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
RODAN AND FIELDS ESSENTIALS UVA/UVB
avobenzone, octinoxate, oxybenzone creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:56152-0815 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 2.0 mL in 100 mL OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 7.5 mL in 100 mL OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 3.0 mL in 100 mL Inactive Ingredients Ingredient Name Strength DIISOPROPYL ADIPATE (UNII: P7E6YFV72X) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) METHYLPROPANEDIOL (UNII: N8F53B3R4R) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PEG-100 STEARATE (UNII: YD01N1999R) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) ASCORBYL PALMITATE (UNII: QN83US2B0N) ALUMINUM STARCH OCTENYLSUCCINATE (UNII: I9PJ0O6294) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) LAURETH-7 (UNII: Z95S6G8201) SODIUM SALICYLATE (UNII: WIQ1H85SYP) C13-14 ISOPARAFFIN (UNII: E4F12ROE70) PHENOXYETHANOL (UNII: HIE492ZZ3T) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:56152-0815-2 1 in 1 BOX 1 NDC:56152-0815-1 30 mL in 1 JAR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 01/01/2009 Labeler - Cosmetic Enterprises Ltd. (017701475) Registrant - Rodan & Fields, LLC. (051659584) Establishment Name Address ID/FEI Business Operations Cosmetic Enterprises Ltd 017701475 manufacture