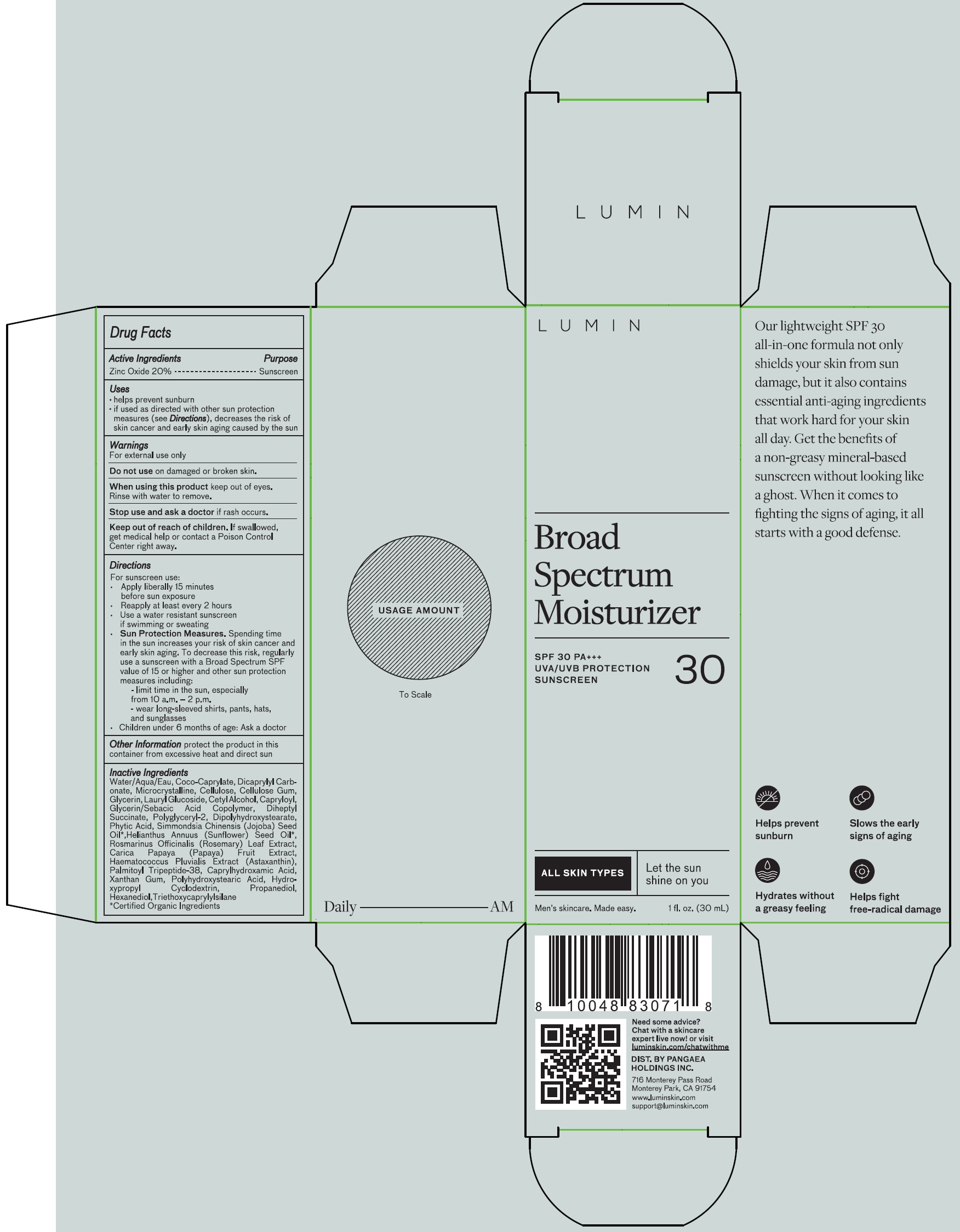
Label: LUMIN BROAD SPECTRUM MOISTURIZER WITH SPF 30 PA UVA/UVB PROTECTION SUNSCREEN- zinc oxide cream
- NDC Code(s): 81234-806-01, 81234-806-02
- Packager: Pangaea Holdings, Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated January 19, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients
- Uses
- Warnings
-
Directions
For sunscreen use:
• Apply liberally 15 minutes before sun exposure
• Reapply at least every 2 hours
• Use a water resistant sunscreen if swimming or sweating
• Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
• Children under 6 months of age: Ask a doctor - Other Information
-
Inactive Ingredients
Water/Aqua, Coco-Caprylate, Dicaprylyl Carbonate, Microcrystalline Cellulose, Cellulose Gum, Glycerin, Lauryl Glucoside, Cetyl Alcohol, Capryloyl, Glycerin/Sebacic Acid Copolymer, Diheptyl Succinate, Polyglyceryl-2 Dipolyhydroxystearate, Phytic Acid, Simmondsia Chinensis (Jojoba) Seed Oil*, Helianthus Annuus (Sunflower) Seed Oil*, Rosmarinus Officinalis (Rosemary) Leaf Extract, Carica Papaya (Papaya) Fruit Extract, Haematococcus Pluvialis Extract (Astaxanthin), Palmitoyl Tripeptide-38, Capryl-O hydroxamic Acid, Xanthan Gum, Polyhydroxystearic Acid, Hydroxypropyl Cyclodextrin, Propanediol, Hexanediol, Triethoxycaprylylsilane *Certified Organic Ingredients
- Package Labeling:30ml
- Package Labeling:40ml
-
INGREDIENTS AND APPEARANCE
LUMIN BROAD SPECTRUM MOISTURIZER WITH SPF 30 PA UVA/UVB PROTECTION SUNSCREEN
zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81234-806 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 200 mg in 1 mL Inactive Ingredients Ingredient Name Strength HYDROXYPROPYL BETADEX (UNII: 1I96OHX6EK) WATER (UNII: 059QF0KO0R) COCO-CAPRYLATE (UNII: 4828G836N6) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED (UNII: K679OBS311) GLYCERIN (UNII: PDC6A3C0OX) LAURYL GLUCOSIDE (UNII: 76LN7P7UCU) CETYL ALCOHOL (UNII: 936JST6JCN) DIHEPTYL SUCCINATE (UNII: 057N7SS26Y) POLYGLYCERYL-2 DIPOLYHYDROXYSTEARATE (UNII: 9229XJ4V12) FYTIC ACID (UNII: 7IGF0S7R8I) JOJOBA OIL (UNII: 724GKU717M) SUNFLOWER OIL (UNII: 3W1JG795YI) ROSEMARY (UNII: IJ67X351P9) PAPAYA (UNII: KU94FIY6JB) HAEMATOCOCCUS PLUVIALIS (UNII: 31T0FF0472) PALMITOYL LYSYLDIOXYMETHIONYLLYSINE (UNII: T7A529FB8O) CAPRYLHYDROXAMIC ACID (UNII: UPY805K99W) XANTHAN GUM (UNII: TTV12P4NEE) PROPANEDIOL (UNII: 5965N8W85T) HEXANEDIOL (UNII: ZIA319275I) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81234-806-01 1 in 1 BOX 01/01/2022 01/05/2025 1 30 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 2 NDC:81234-806-02 1 in 1 BOX 01/01/2022 01/05/2025 2 40 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2022 01/05/2025 Labeler - Pangaea Holdings, Inc. (081181313)