Label: TRIPLE ANTIBIOTIC- first aid antibiotic ointment
- NDC Code(s): 68001-483-45, 68001-483-46
- Packager: BluePoint Laboratories
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated January 31, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Purpose
- Uses
- Warnings
- Do not use
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive ingredients
- Questions?
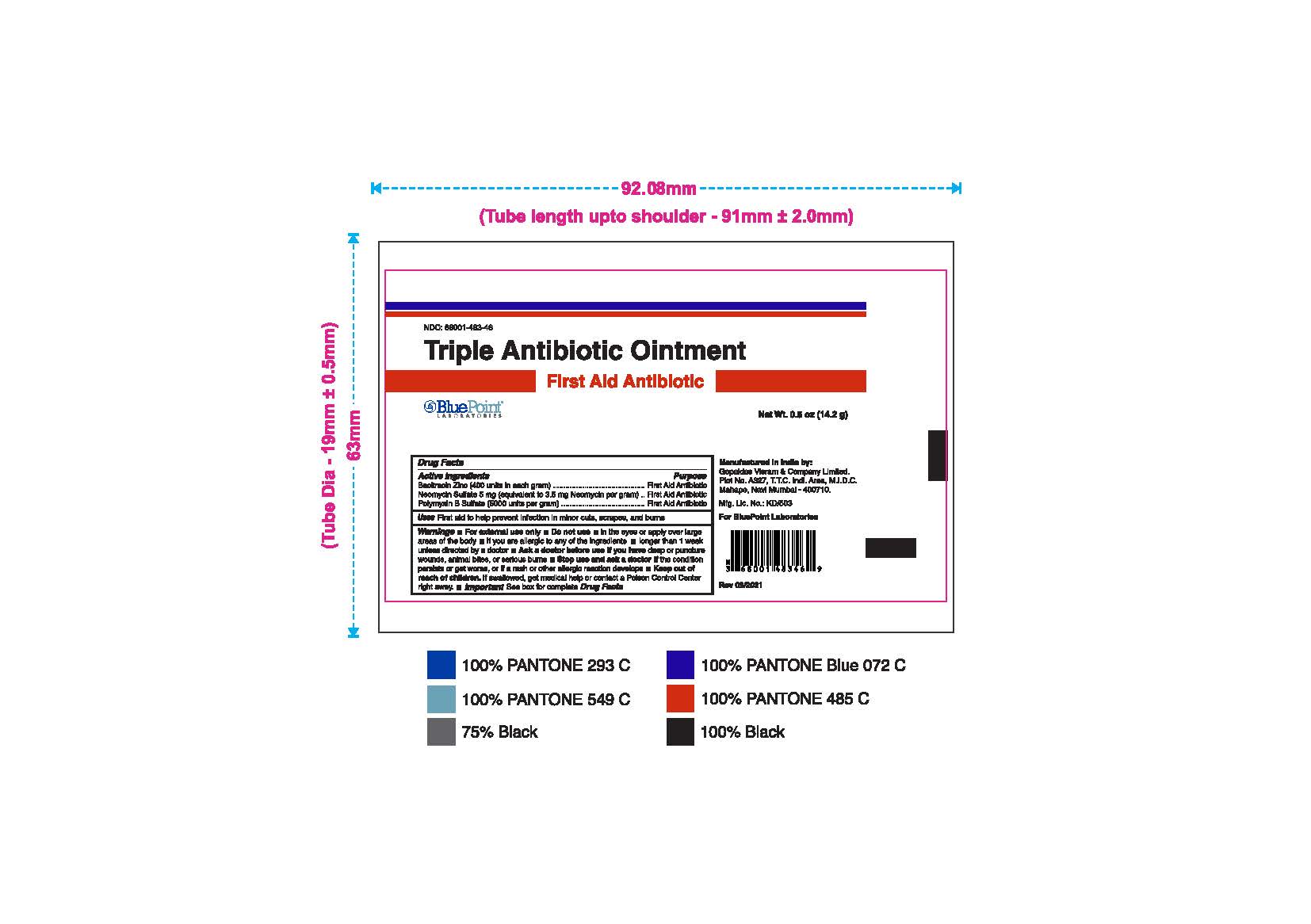
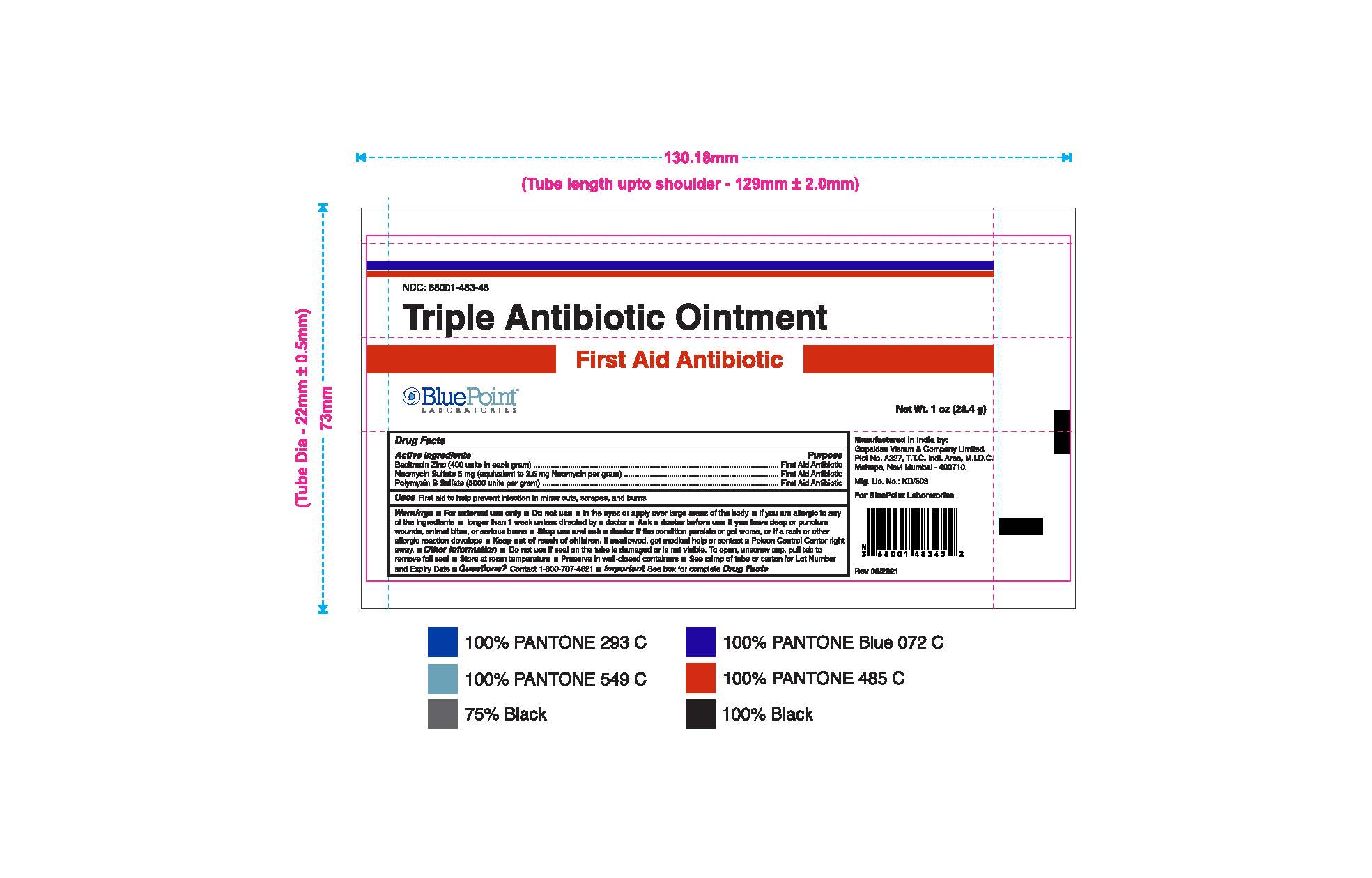
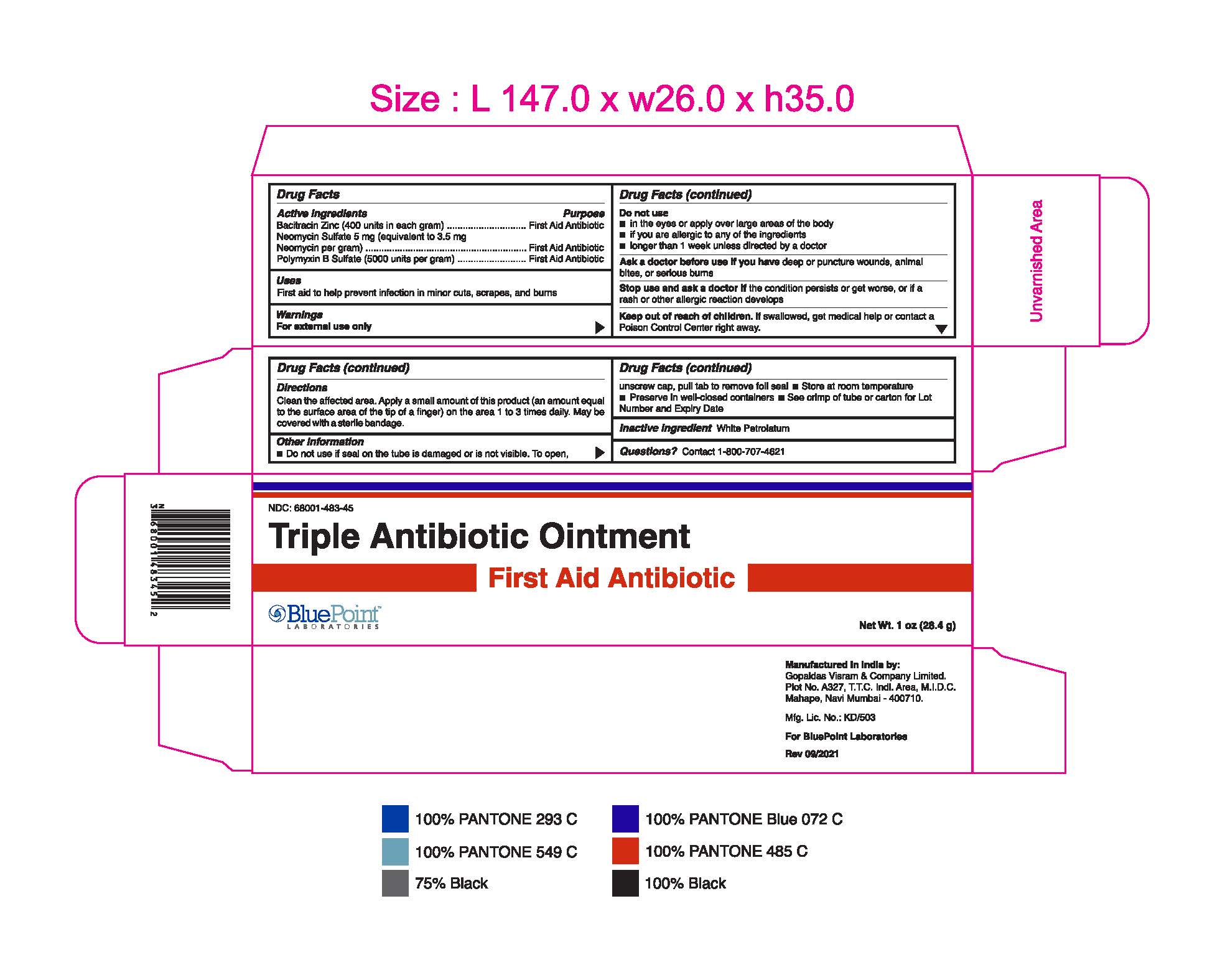
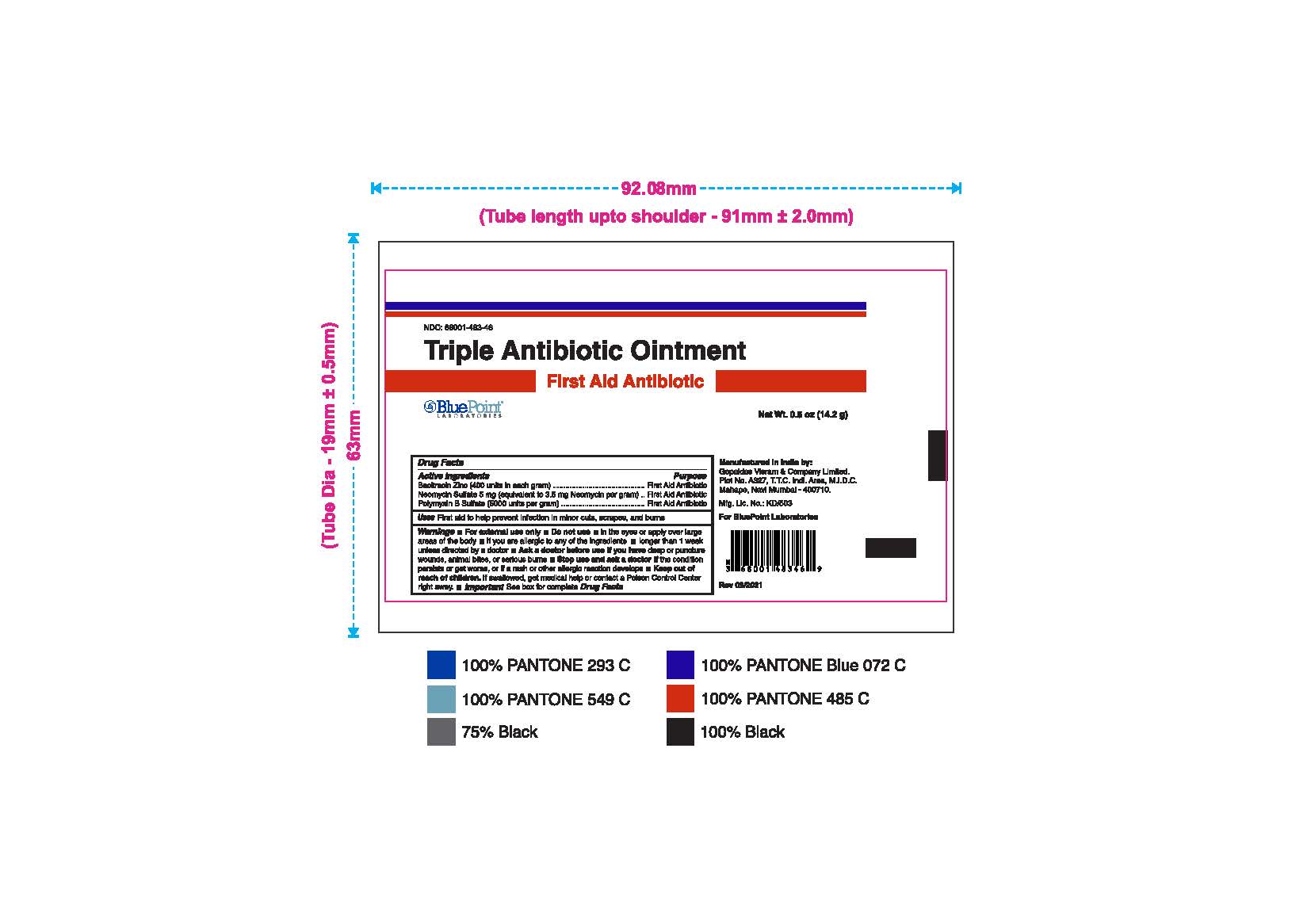
- Package Label - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
TRIPLE ANTIBIOTIC
first aid antibiotic ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68001-483 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 g BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 400 [iU] in 1 g POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 5000 [iU] in 1 g Inactive Ingredients Ingredient Name Strength WHITE PETROLATUM (UNII: B6E5W8RQJ4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68001-483-46 1 in 1 CARTON 06/15/2021 1 14.2 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:68001-483-45 1 in 1 CARTON 06/15/2021 2 28.4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M004 06/15/2021 Labeler - BluePoint Laboratories (985523874) Establishment Name Address ID/FEI Business Operations Gopaldas Visram & Co Ltd 858030888 manufacture(68001-483)


 Triple Antibiotic Ointment 0.5 oz NDC: 68001-483-46
Triple Antibiotic Ointment 0.5 oz NDC: 68001-483-46

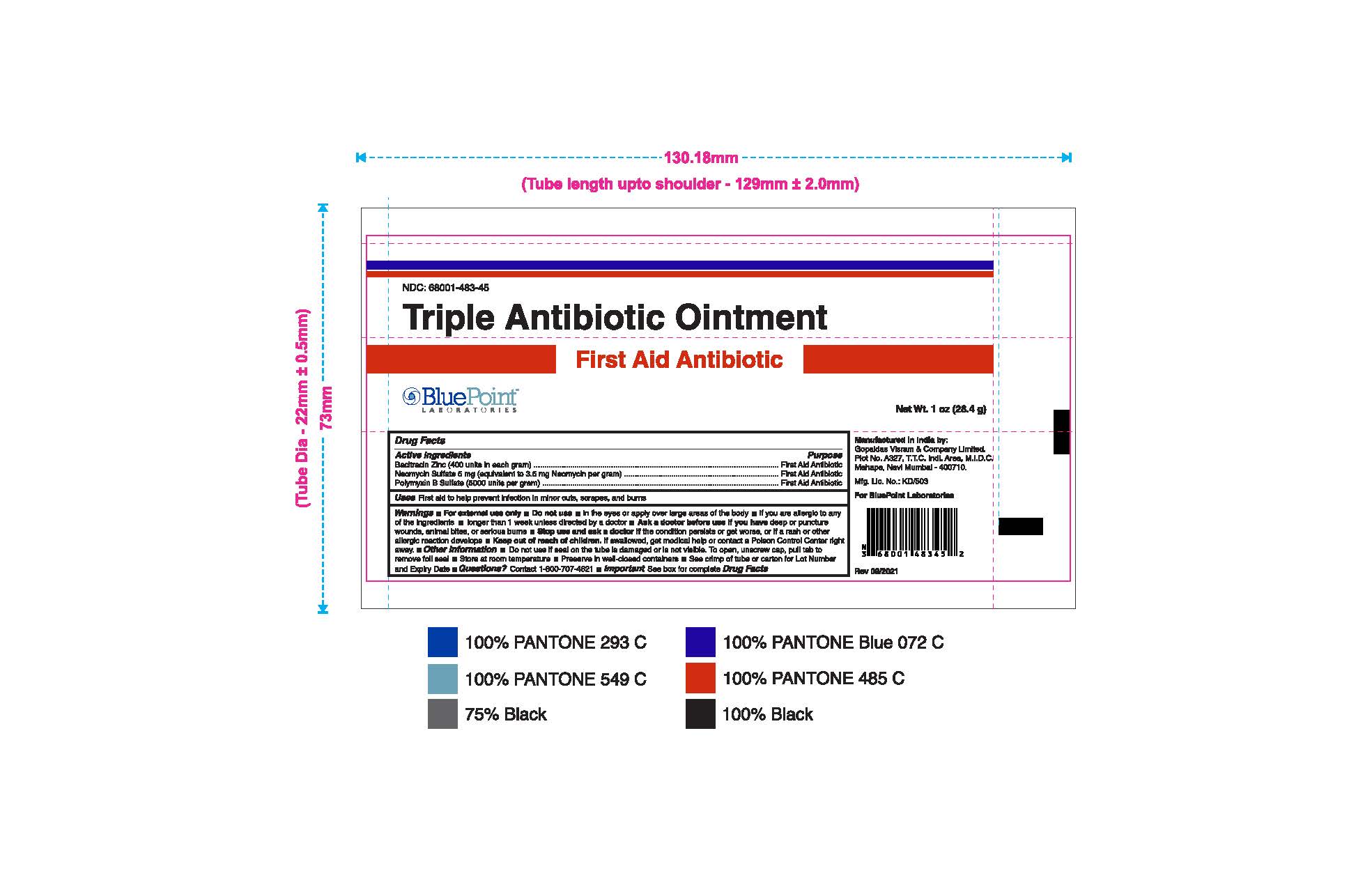
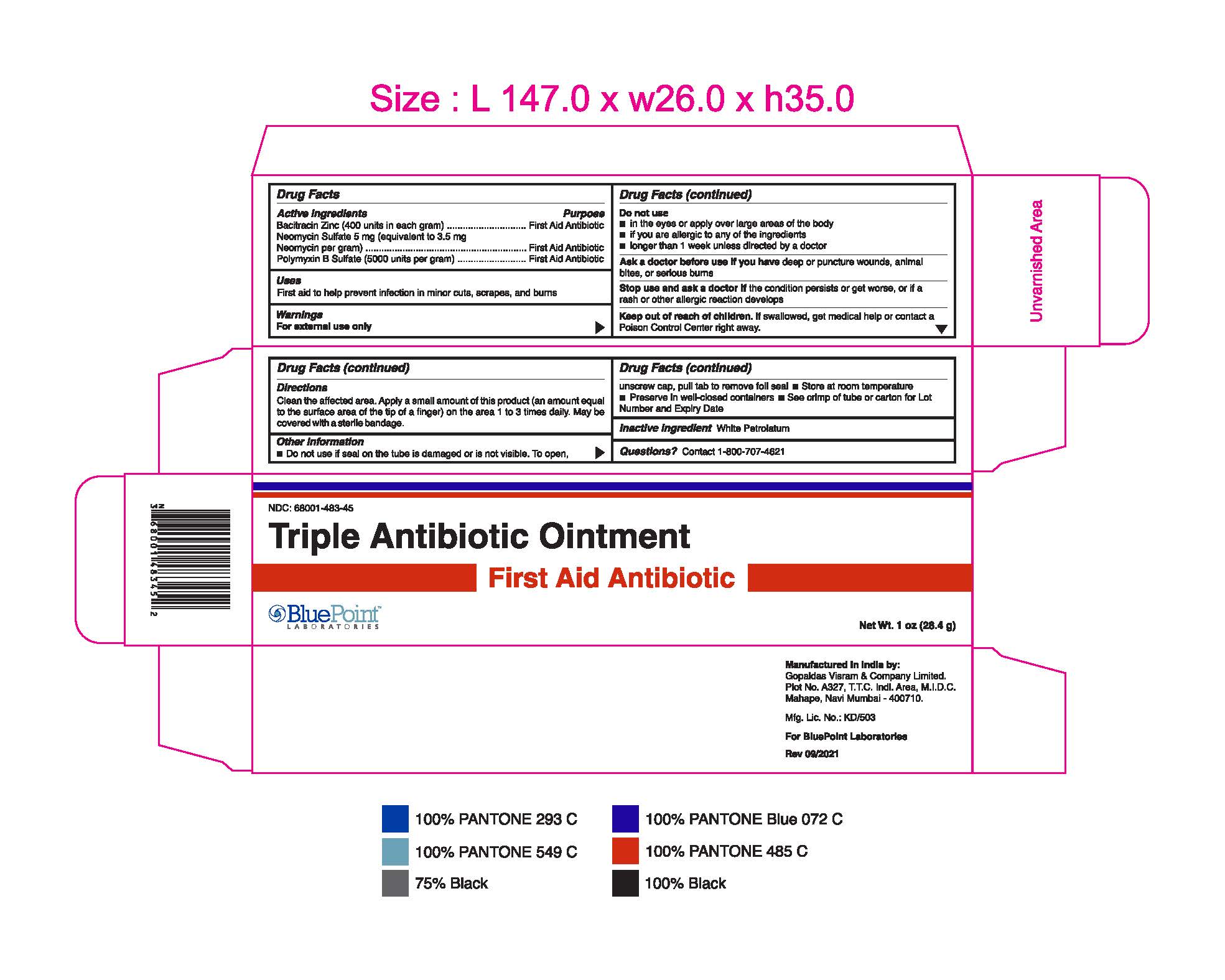 Triple Antibiotic Ointment 1 oz 68001-483-45
Triple Antibiotic Ointment 1 oz 68001-483-45