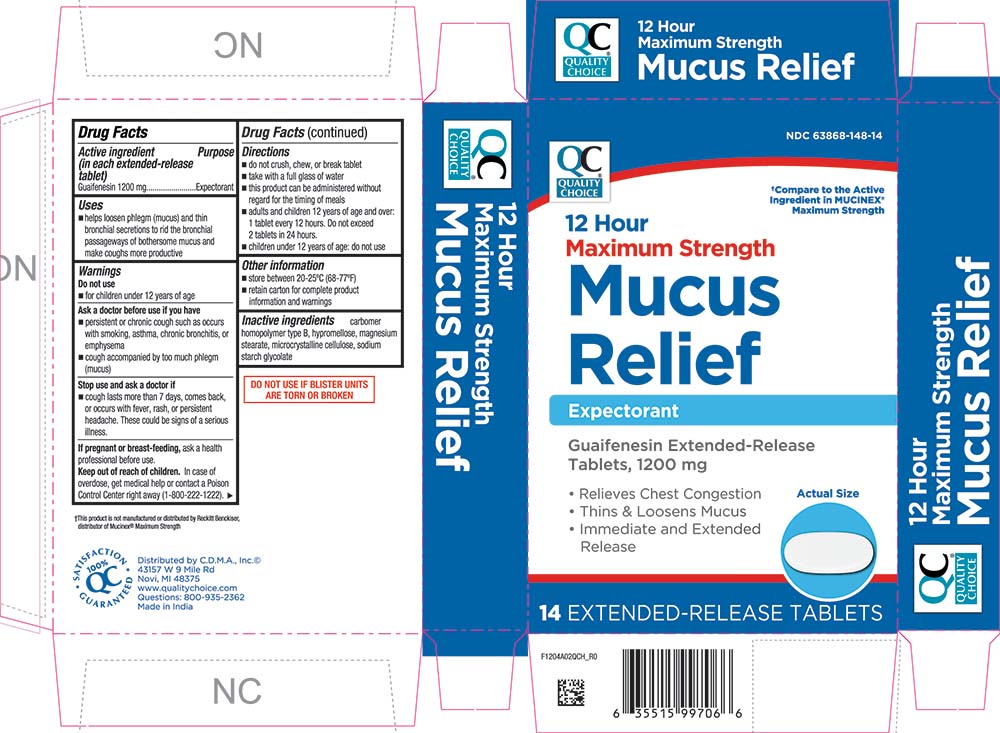
Label: MAXIMUM STRENGTH MUCUS RELIEF- guaifenesin tablet, extended release
- NDC Code(s): 63868-148-14
- Packager: CHAIN DRUG MARKETING ASSOCIATION INC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 10, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each extended-release tablet)
- Purpose
- Uses
-
WARNINGS
Warnings
Ask a doctor before use if you have
■ persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
■ cough accompanied by too much phlegm (mucus)
-
Directions
■ do not crush, chew, or break tablet
■ take with a full glass of water
■ this product can be administered without regard for the timing of meals
■ adults and children 12 years of age and over: 1 tablet every 12 hours. Do not exceed 2 tablets in 24 hours.
■ children under 12 years of age: do not use
- Other information
- Inactive ingredients
-
PRINCIPAL DISPLAY PANEL
QUALITY CHOICE®
NDC 63868-148-14
†Compare to the Active Ingredient in MUCINEX® Maximum Strength
12 Hour
Maximum Strength
Mucus Relief
Expectorant
Guaifenesin Extended-Release Tablets, 1200 mg
• Relieves Chest Congestion
• Thin & Loosens Mucus
• Immediate and Extended Release
Actual Size
14 EXTENDED-RELEASE TABLETS
-
INGREDIENTS AND APPEARANCE
MAXIMUM STRENGTH MUCUS RELIEF
guaifenesin tablet, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63868-148 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 1200 mg Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER TYPE B (ALLYL SUCROSE CROSSLINKED) (UNII: Z135WT9208) HYPROMELLOSE 2910 (15 MPA.S) (UNII: 36SFW2JZ0W) SODIUM STARCH GLYCOLATE TYPE A (UNII: H8AV0SQX4D) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) Product Characteristics Color white Score no score Shape OVAL (Elliptical) Size 22mm Flavor Imprint Code G;1200 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63868-148-14 2 in 1 CARTON 09/17/2021 1 7 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA213420 09/17/2021 Labeler - CHAIN DRUG MARKETING ASSOCIATION INC (011920774)