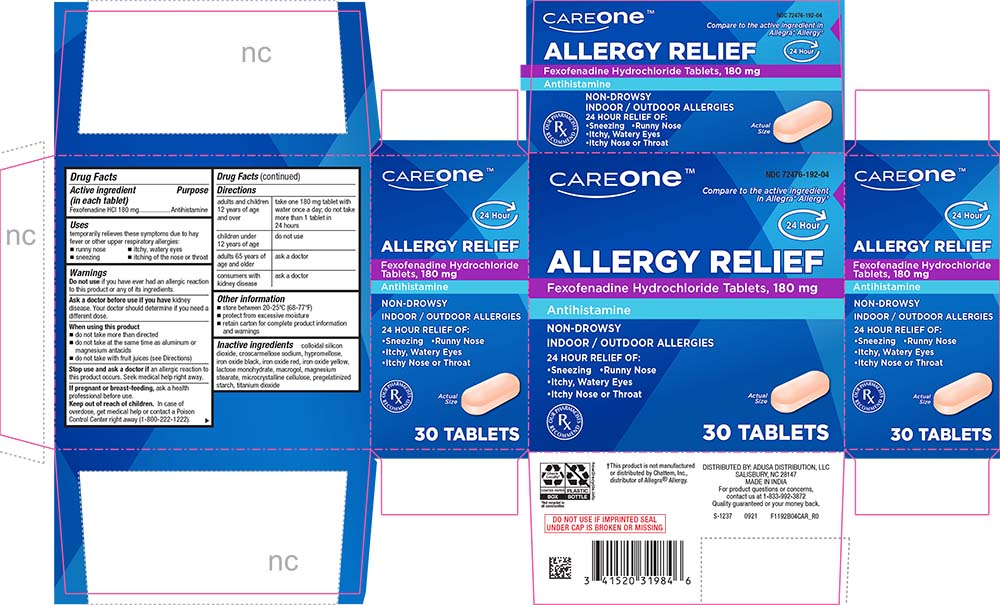
Label: ALLERGY RELIEF- fexofenadine hydrochloride tablet
- NDC Code(s): 72476-192-04, 72476-192-06, 72476-192-09
- Packager: RETAIL BUSINESS SERVICES , LLC.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 24, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each tablet)
- Purpose
- Uses
-
WARNINGS
Warnings
Ask a doctor before use if you have kidney disease. Your doctor should determine if you need a different dose.
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
- Directions
- Other information
- Inactive ingredients
-
PRINCIPAL DISPLAY PANEL
CAREONE™
NDC 72476-192-04
Compare to the active ingredient in Allegra® Allergy†
24 Hour
ALLERGY RELIEF
Fexofenadine Hydrochloride Tablets, 180 mg
Antihistamine
NON-DROWSY
INDOOR / OUTDOOR ALLERGIES
24 HOUR RELIEF OF:
•Sneezing •Runny Nose
•Itchy, Watery Eyes
•Itchy Nose or Throat
Actual Size
30 TABLETS
-
INGREDIENTS AND APPEARANCE
ALLERGY RELIEF
fexofenadine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72476-192 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FEXOFENADINE HYDROCHLORIDE (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) FEXOFENADINE HYDROCHLORIDE 180 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) HYPROMELLOSE 2910 (15 MPA.S) (UNII: 36SFW2JZ0W) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL 6000 (UNII: 30IQX730WE) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color orange ((PEACH)) Score no score Shape OVAL (Capsule-shaped) Size 17mm Flavor Imprint Code G6 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72476-192-04 1 in 1 CARTON 10/04/2021 1 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC:72476-192-06 1 in 1 CARTON 10/04/2021 2 45 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 3 NDC:72476-192-09 1 in 1 CARTON 07/14/2021 3 90 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA211075 07/14/2021 Labeler - RETAIL BUSINESS SERVICES , LLC. (967989935)