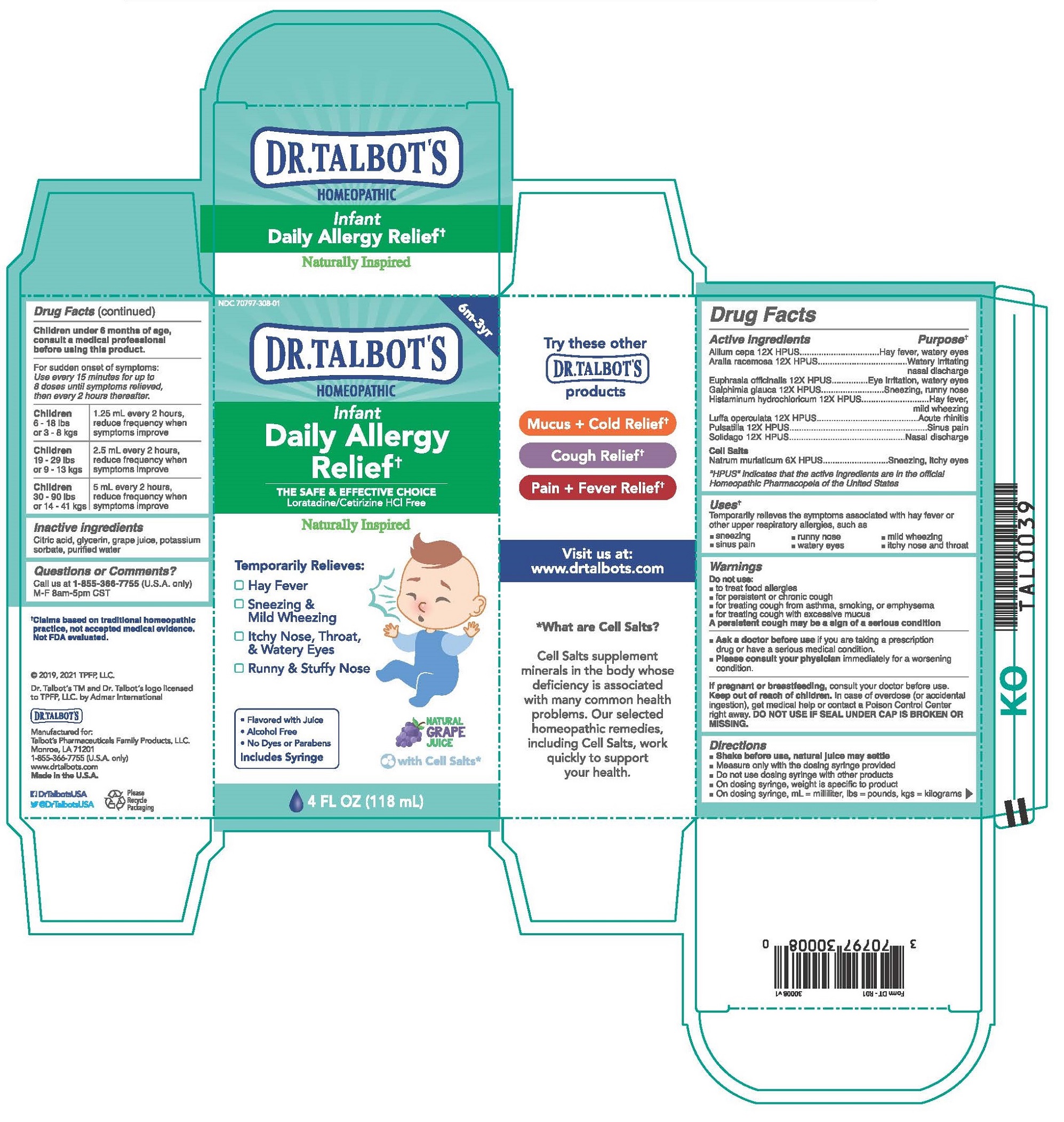
Label: DR. TALBOTS INFANT DAILY ALLERGY RELIEF- allium cepa, aralia racemosa, euphrasia officinalis, galphimia glauca, histaminum hydrochloricum, luffa operculata, pulsatilla, solidago, natrum muriaticum liquid
- NDC Code(s): 70797-308-01, 70797-308-04
- Packager: Talbot's Pharmaceuticals Family Products, LLC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated April 22, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
- Shake before use, natural juice may settle
- Measure only with the dosing syringe provided
- Do not use dosing syringe with other products
- On dosing syringe, weight is specific to product
- On dosing syringe, ml = milliliter, lbs = pounds, kgs = kilograms
Children under 6 months of age, consult a medical professional before using this product.
For sudden onset of symptoms: Use every 15 minutes for up to 8 doses until symptoms relieved, then every 2 hours thereafter.
Children 6 - 18 lbs or 3 - 8 kgs
1.25 ml every 2 hours, reduce frequency when symptoms improve
Children 19 - 29 lbs or 9 - 13 kgs
2.5 ml every 2 hours, reduce frequency when symptoms improve
Children 30 - 90 lbs or 14 - 41 kgs
5 ml every 2 hours, reduce frequency when symptoms improve
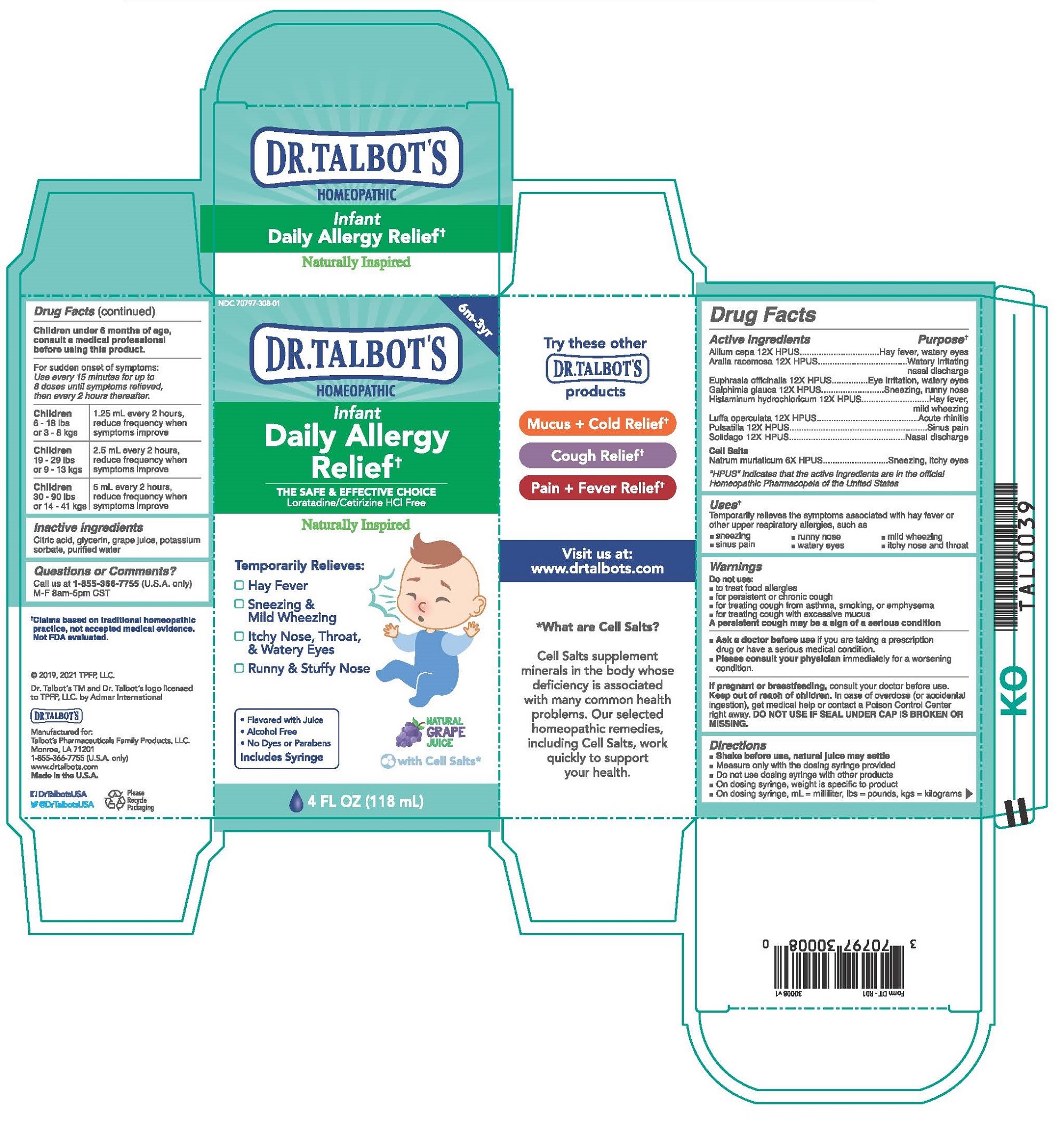
- INACTIVE INGREDIENT
- QUESTIONS
- INDICATIONS & USAGE
- WARNINGS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DR. TALBOTS INFANT DAILY ALLERGY RELIEF
allium cepa, aralia racemosa, euphrasia officinalis, galphimia glauca, histaminum hydrochloricum, luffa operculata, pulsatilla, solidago, natrum muriaticum liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70797-308 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LUFFA OPERCULATA FRUIT (UNII: C4MO6809HU) (LUFFA OPERCULATA FRUIT - UNII:C4MO6809HU) LUFFA OPERCULATA FRUIT 12 [hp_X] in 118 mL EUPHRASIA STRICTA (UNII: C9642I91WL) (EUPHRASIA STRICTA - UNII:C9642I91WL) EUPHRASIA STRICTA 12 [hp_X] in 118 mL GALPHIMIA GLAUCA FLOWERING TOP (UNII: 93PH5Q8M7E) (GALPHIMIA GLAUCA FLOWERING TOP - UNII:93PH5Q8M7E) GALPHIMIA GLAUCA FLOWERING TOP 12 [hp_X] in 118 mL ARALIA RACEMOSA ROOT (UNII: T90W4582DU) (ARALIA RACEMOSA ROOT - UNII:T90W4582DU) ARALIA RACEMOSA ROOT 12 [hp_X] in 118 mL SOLIDAGO VIRGAUREA FLOWERING TOP (UNII: 5405K23S50) (SOLIDAGO VIRGAUREA FLOWERING TOP - UNII:5405K23S50) SOLIDAGO VIRGAUREA FLOWERING TOP 12 [hp_X] in 118 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM CHLORIDE 6 [hp_X] in 118 mL ONION (UNII: 492225Q21H) (ONION - UNII:492225Q21H) ONION 12 [hp_X] in 118 mL ANEMONE PULSATILLA (UNII: I76KB35JEV) (ANEMONE PULSATILLA - UNII:I76KB35JEV) ANEMONE PULSATILLA 12 [hp_X] in 118 mL HISTAMINE DIHYDROCHLORIDE (UNII: 3POA0Q644U) (HISTAMINE - UNII:820484N8I3) HISTAMINE DIHYDROCHLORIDE 12 [hp_X] in 118 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) CONCORD GRAPE JUICE (UNII: F7039Q79LP) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70797-308-01 1 in 1 CARTON 06/01/2021 1 NDC:70797-308-04 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 06/01/2021 Labeler - Talbot's Pharmaceuticals Family Products, LLC (078855555)