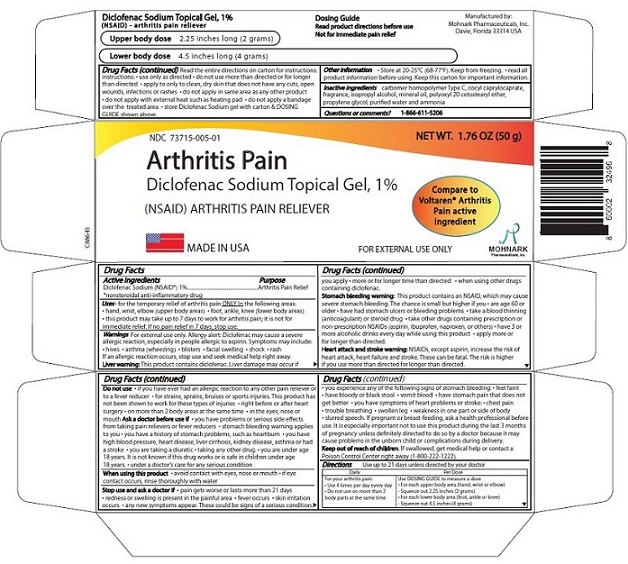
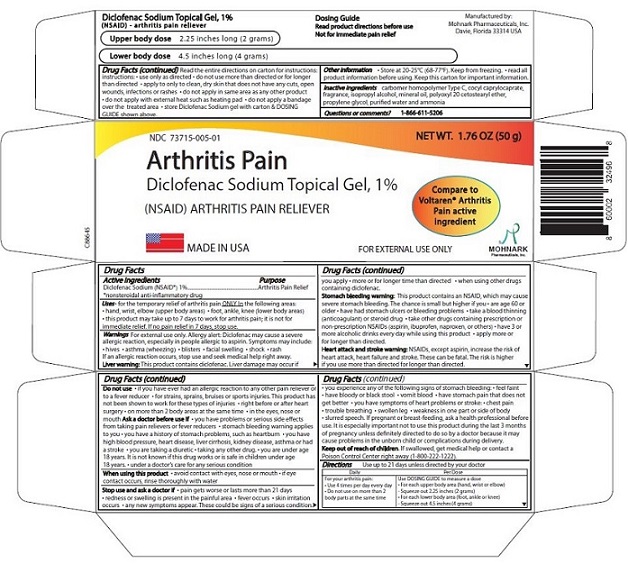
Label: DICLOFENAC SODIUM TOPICAL GEL, 1% (diclofenac sodium topical gel, 1%- nsaid gel
- NDC Code(s): 73715-005-01
- Packager: Mohnark Pharmaceuticals Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated July 7, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INACTIVE INGREDIENTS
- PURPOSE
- USES
-
WARNINGS
For external use only
Allergy alert: Diclofenac may cause a severe allergic reaction, especially in people allergic to aspirin.
Symptoms may include:
• hives • asthma (wheezing) • skin reddening • blisters • facial swelling • shock • rash
If an allergic reaction occurs, stop use and seek medical help right away.
Liver warning: This product contains diclofenac. Liver damage may occur if you apply
- more or for a longer time than directed
- when using other drugs containing diclofenac
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is small but higher if you
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or non-prescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- have 3 or more alcoholic drinks every day while using this product
- apply more or for longer than directed
Heart attack and stroke warning: NSAIDs, except aspirin, increase the risk of heart attack, heart failure, and stroke. These can be fatal. The risk is higher if you use more than directed or for longer than directed.
-
DO NOT USE
- if you have ever had an allergic reaction to any other pain reliever or to a fever reducer
- for strains, sprains, bruises or sports injuries. This product has not been shown to work for these types of injuries.
- right before or after heart surgery
- on more than 2 body areas at the same time
- in the eyes, nose or mouth
-
ASK A DOCTOR BEFORE USE IF
- you have problems or serious side effects from taking pain relievers or fever reducers
- stomach bleeding warning applies to you you have a history of stomach problems, such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis, kidney disease, asthma, or had a stroke
- you are taking a diuretic
- you are under age 18 years. It is not known if this drug works or is safe in children under age 18 years.
- ASK A DOCTOR OR PHARMACIST BEFORE USE IF YOU ARE
- WHEN USING THIS PRODUCT
-
STOP USE AND ASK A DOCTOR IF
- pain gets worse or lasts more than 21 days
- redness or swelling is present in the painful area
- fever occurs
- skin irritation occurs
- any new symptoms appear. These could be signs of a serious condition.
- you experience any of the following signs of stomach bleeding
- feel faint
- have bloody or black stools
- vomit blood
- have stomach pain that does not get better
- you have symptoms of heart problems or stroke
- chest pain
- trouble breathing
- leg swelling
- weakness in one part or side of body
- slurred speech
- IF PREGNANT OR BREAST-FEEDING
- KEEP OUT OF REACH OF CHILDREN
-
DIRECTIONS
Use up to 21 days unless directed by your doctor
Not for strains, sprains, bruises or sports injuries. This product has not been shown to work for these types of injuries.
For your arthritis pain:Daily Per Dose
Use 4 times per day every day
Do not use on more than 2 bodyareas at the same time
Use DOSING GUIDE to measure a dose
For each upper body area (hand, wrist, or elbow)
- Squeeze out 2.25 inches (2 grams)
For each lower body area (foor, ankle or knee) -
Squeeze out 4.5 inches (4 grams)
Read the entire directions on carton for instructions:
use only as directed
do not use more than directed or for longer than directed
apply only to clean, dry skin that does not have any cuts, open wounds, infections or rashes
do not apply in same area as any other product
do not apply with external heat such as heating pad
do not apply a bandage over the treated areastore Diclofenac Sodium Gel with carton and dosing guide as shown.
-
OTHER INFORMATION
- Store at 20-25°C (68°F - 77°F). Keep from freezing.
- read all product information before using. Keep from freezing. read all product information before using. Keep this carton for important information.
Inactive ingredients
Carbomer homopolymer Type C, cocoyl caprylocaprate, ispropyl alcohol, mineral oil, polyoxyl 20 cetostearyl ether, propylene glycol, purified water, strong ammonia solution.
Question and comments: 1-866-611-5206
- INDICATIONS AND USAGE
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DICLOFENAC SODIUM TOPICAL GEL, 1%
diclofenac sodium topical gel, 1% (nsaid) gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73715-005 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DICLOFENAC SODIUM (UNII: QTG126297Q) (DICLOFENAC - UNII:144O8QL0L1) DICLOFENAC SODIUM 1 g in 100 g Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) WATER (UNII: 059QF0KO0R) MINERAL OIL (UNII: T5L8T28FGP) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) COCO-CAPRYLATE/CAPRATE (UNII: 8D9H4QU99H) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73715-005-01 50 g in 1 CARTON; Type 0: Not a Combination Product 08/31/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 08/31/2021 Labeler - Mohnark Pharmaceuticals Inc. (117013830) Registrant - Mohnark Pharmaceuticals Inc. (117013830) Establishment Name Address ID/FEI Business Operations Mohnark Pharmaceuticals Inc 117013830 manufacture(73715-005)