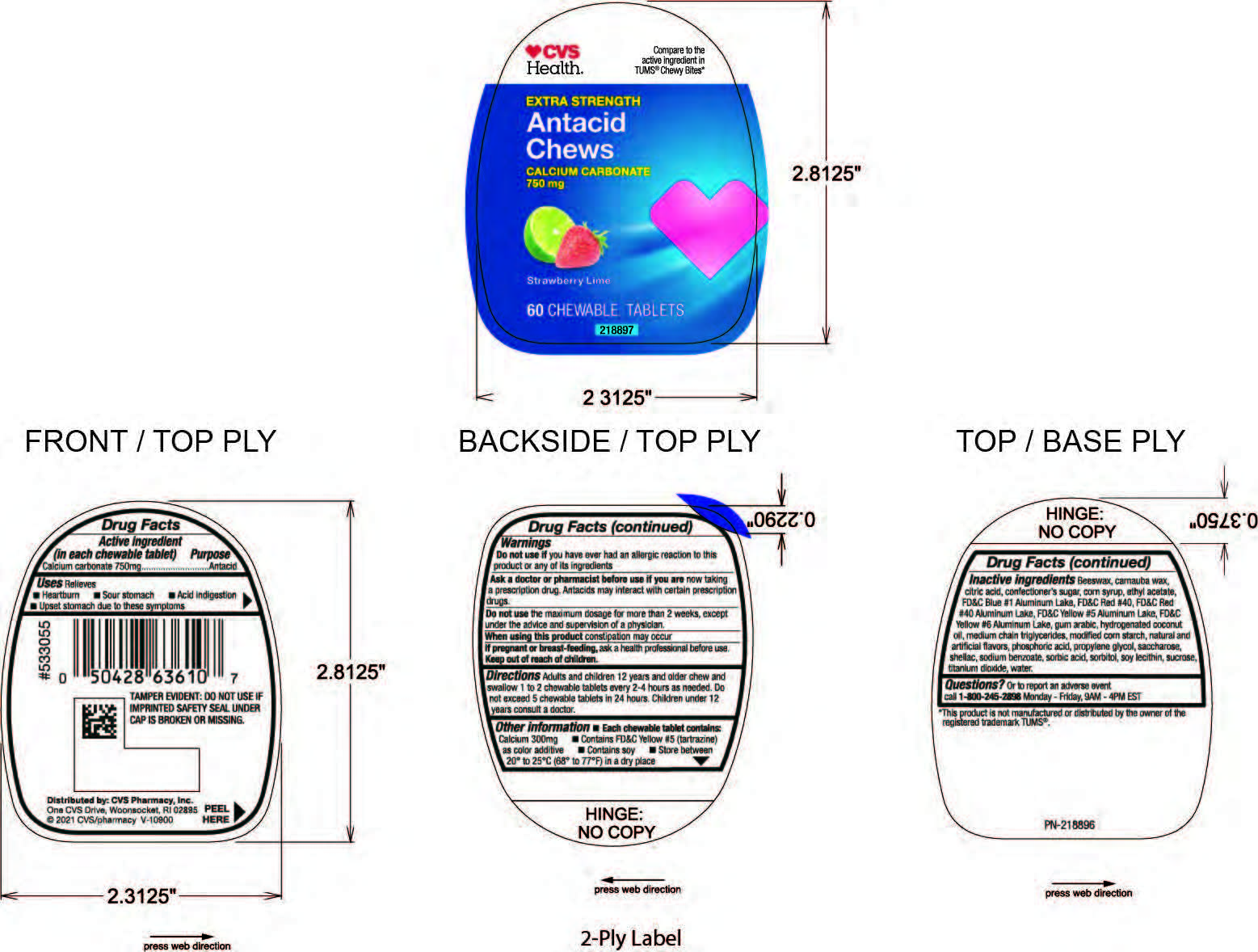
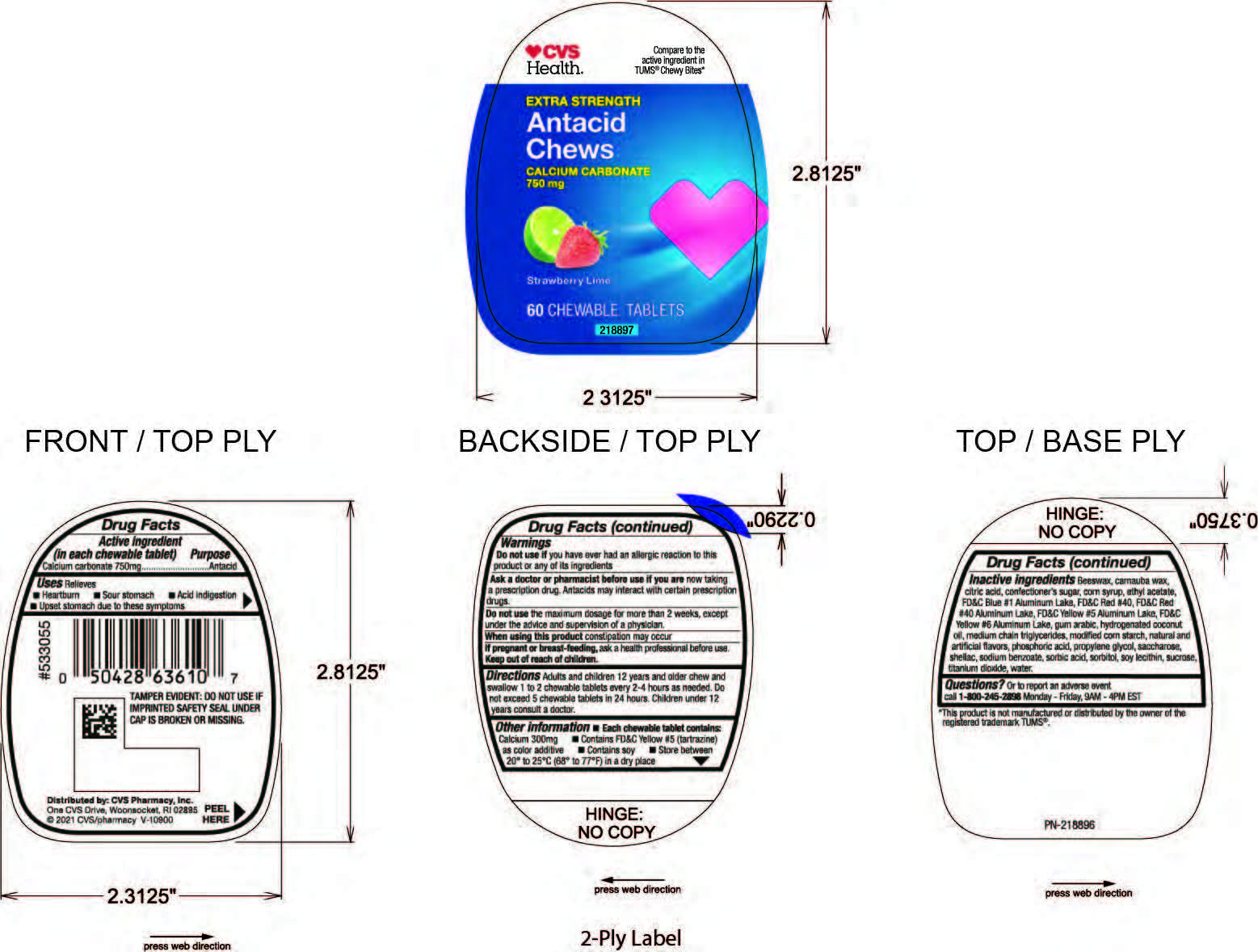
Label: CVS ANTACID LIME-BERRY CHEWS- calcium carbonate tablet, chewable
- NDC Code(s): 69842-436-60
- Packager: CVS Pharmacy
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 20, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- ASK DOCTOR/PHARMACIST
- DO NOT USE
- WHEN USING
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
-
INACTIVE INGREDIENT
Inactive ingredients: Beeswax, carnauba wax, citric acid, confectioner's sugar, corn syrup, ethyl acetate, FD&C Blue #1 Aluminum Lake, FD&C Red #40, FD&C Red #40 Aluminum Lake, FD&C Yellow #5 Aluminum Lake, FD&G Yellow #6 Aluminum Lake, gum arabic, hydrogenated coconut oil, mediul chain triglycerides, modified corn starch, natural and artificial flavors, phosphoric acid, propylene glycol, saccharose, shellac, sodium benzoate, sorbic acid, sorbitol, soy lecithin, sucrose, titanium dioxide, water.
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CVS ANTACID LIME-BERRY CHEWS
calcium carbonate tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-436 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM CARBONATE 750 mg Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Product Characteristics Color green (Green and Red) Score no score Shape ROUND Size 22mm Flavor LIME (Lime and Strawberry) Imprint Code FC Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-436-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 04/08/2021 
Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M001 04/08/2021 Labeler - CVS Pharmacy (062312574) Registrant - Bestco LLC (002149136) Establishment Name Address ID/FEI Business Operations Bestco LLC 002149136 manufacture(69842-436)