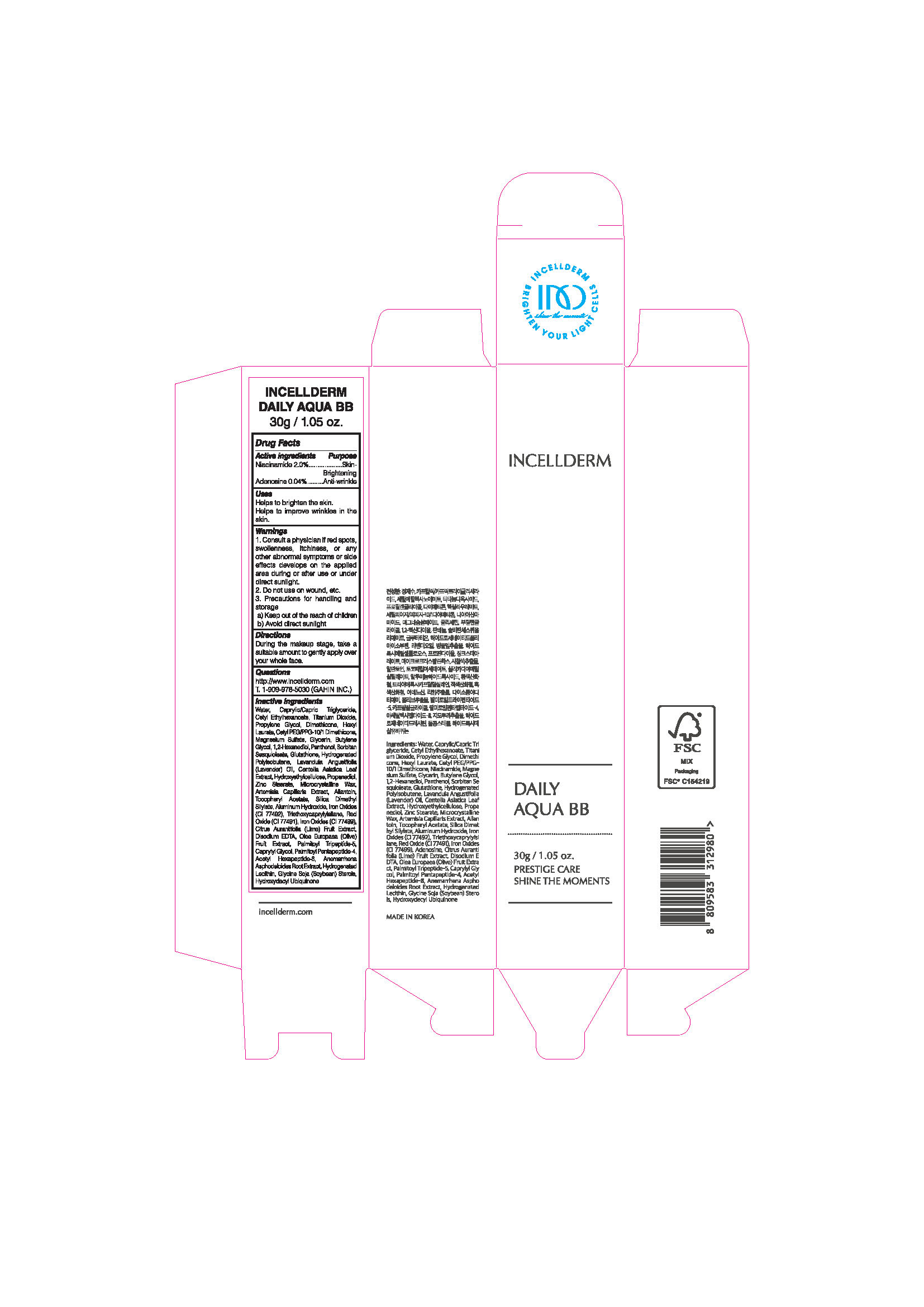
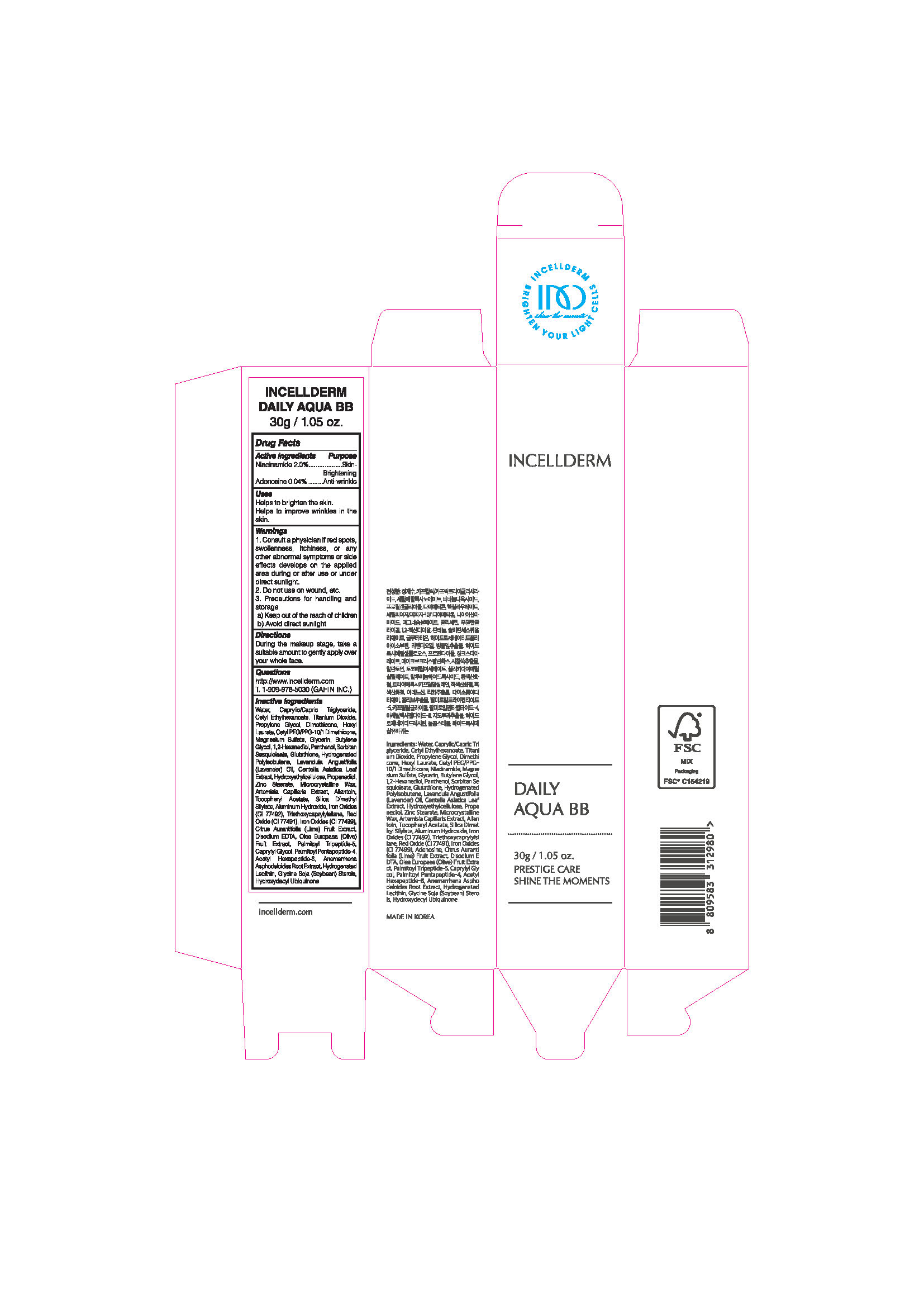
Label: INCELLDERM DAILY AQUA BB- niacinamide, adenosine cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 72650-100-01 - Packager: Riman Korea Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated April 1, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS
-
INACTIVE INGREDIENTS
Water, Caprylic/Capric Triglyceride, CetylEthylhexanoate, Titanium Dioxide, Propylene Glycol, Dimethicone, Hexyl Laurate, Cetyl PEG/PPG-10/1 Dimethicone, Magnesium Sulfate, Glycerin, Butylene Glycol, 1,2-Hexanediol, Panthenol, SorbitanSesquioleate, Glutathione, Hydrogenated Polyisobutene, Lavandula Angustifolia (Lavender) Oil, Centella Asiatica Leaf Extract , Hydroxyethylcellulose, Propanediol, Zinc Stearate, Microcrystalline Wax, Artemisia Capillaris Extract, Allantoin, Tocopheryl Acetate, Silica Dimethyl Silylate, Aluminum Hydroxide, Iron Oxides (CI 77492), Triethoxycaprylylsilane, Red Oxide (CI 77491), Iron Oxides (CI 77499), Citrus Aurantifolia (Lime) Fruit Extract, Disodium EDTA, Olea Europaea (Olive) Fruit Extract, Palmitoyl Tripeptide-5, Caprylyl Glycol, Palmitoyl Pentapeptide-4, Acetyl Hexapeptide-8, AnemarrhenaAsphodeloides Root Extract, Hydrogenated Lecithin, Glycine Soja (Soybean) Sterols, Hydroxydecyl Ubiquinone
- PURPOSE
-
WARNINGS
1. Consult a physician if red spots, swollenness, itchiness, or any other abnormal symptoms or side effects develops on the applied area during or after use or under direct sunlight.
2. Do not use on wound, etc.
3. Precautions for handling and storage
a) Keep out of the reach of children
b) Avoid direct sunlight
- KEEP OUT OF REACH OF CHILDREN
- Uses
- Directions
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
INCELLDERM DAILY AQUA BB
niacinamide, adenosine creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72650-100 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 2 g in 100 g ADENOSINE (UNII: K72T3FS567) (ADENOSINE - UNII:K72T3FS567) ADENOSINE 0.04 g in 100 g Inactive Ingredients Ingredient Name Strength DIMETHICONE (UNII: 92RU3N3Y1O) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72650-100-01 30 g in 1 CONTAINER; Type 0: Not a Combination Product 03/27/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 03/27/2021 Labeler - Riman Korea Co., Ltd. (695094804) Registrant - Riman Korea Co., Ltd. (695094804) Establishment Name Address ID/FEI Business Operations HANSOLBIOTECH Co., Ltd. 694455165 manufacture(72650-100) Establishment Name Address ID/FEI Business Operations Riman Korea Co Ltd 695094804 label(72650-100)