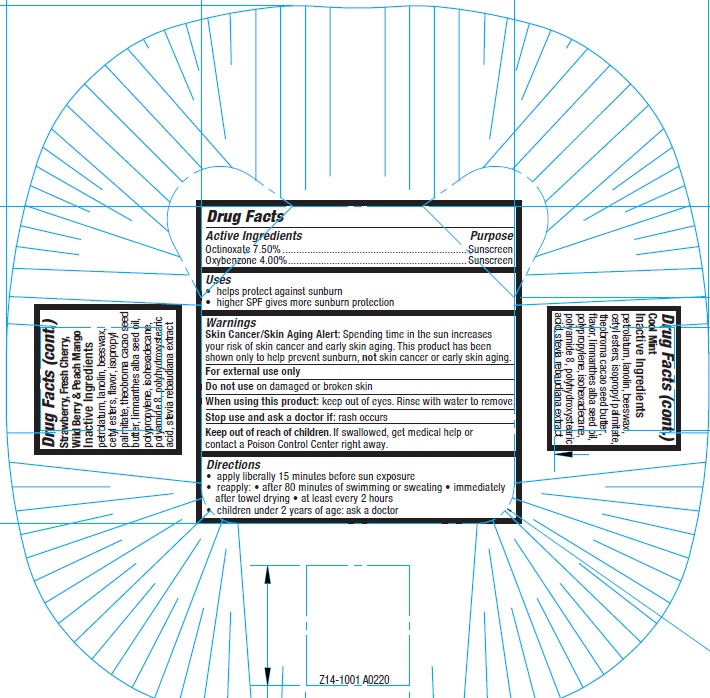
Label: CARMEX DAILY CARE MOISTURIZING LIP BALM SPF 15 MINIS- octinoxate, oxybenzone kit
-
NDC Code(s):
10210-0029-0,
10210-0032-0,
10210-0034-0,
10210-0035-0, view more10210-0036-0, 10210-0040-1
- Packager: Carma Laboratories, Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 25, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients
- Uses
- Warnings
- Directions:
- Other Information
- Inactive Ingredients:
- Package Labeling:
- Package Labeling:10210-0029-0
- Package Labeling:10210-0032-0
- Package Labeling:10210-0034-0
- Package Labeling:10210-0035-0
- Package Labeling:10210-0036-0
-
INGREDIENTS AND APPEARANCE
CARMEX DAILY CARE MOISTURIZING LIP BALM SPF 15 MINIS
octinoxate, oxybenzone kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10210-0040 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10210-0040-1 1 in 1 KIT 11/05/2020 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 5 g Part 2 1 TUBE 5 g Part 3 1 TUBE 5 g Part 4 1 TUBE 5 g Part 5 1 TUBE 5 g Part 1 of 5 CARMEX DAILY CARE MOISTURIZING LIP BALM FRESH CHERRY SPF 15
octinoxate, oxybenzone salveProduct Information Item Code (Source) NDC:10210-0029 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 40 mg in 1 g Inactive Ingredients Ingredient Name Strength YELLOW WAX (UNII: 2ZA36H0S2V) PETROLATUM (UNII: 4T6H12BN9U) LANOLIN (UNII: 7EV65EAW6H) CETYL ESTERS WAX (UNII: D072FFP9GU) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) ISOHEXADECANE (UNII: 918X1OUF1E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10210-0029-0 5 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 05/20/2019 Part 2 of 5 CARMEX DAILY CARE MOISTURIZING LIP BALM STRAWBERRY SPF 15
octinoxate, oxybenzone salveProduct Information Item Code (Source) NDC:10210-0032 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 40 mg in 1 g Inactive Ingredients Ingredient Name Strength YELLOW WAX (UNII: 2ZA36H0S2V) PETROLATUM (UNII: 4T6H12BN9U) LANOLIN (UNII: 7EV65EAW6H) CETYL ESTERS WAX (UNII: D072FFP9GU) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) ISOHEXADECANE (UNII: 918X1OUF1E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10210-0032-0 5 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 07/12/2017 Part 3 of 5 CARMEX DAILY CARE MOISTURIZING LIP BALM WILD BERRY SPF 15
octinoxate, oxybenzone salveProduct Information Item Code (Source) NDC:10210-0034 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 40 mg in 1 g Inactive Ingredients Ingredient Name Strength YELLOW WAX (UNII: 2ZA36H0S2V) PETROLATUM (UNII: 4T6H12BN9U) LANOLIN (UNII: 7EV65EAW6H) CETYL ESTERS WAX (UNII: D072FFP9GU) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) ISOHEXADECANE (UNII: 918X1OUF1E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10210-0034-0 5 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 11/05/2020 Part 4 of 5 CARMEX DAILY CARE MOISTURIZING LIP BALM COOL MINT SPF 15
octinoxate, oxybenzone salveProduct Information Item Code (Source) NDC:10210-0035 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 40 mg in 1 g Inactive Ingredients Ingredient Name Strength YELLOW WAX (UNII: 2ZA36H0S2V) PETROLATUM (UNII: 4T6H12BN9U) LANOLIN (UNII: 7EV65EAW6H) CETYL ESTERS WAX (UNII: D072FFP9GU) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) ISOHEXADECANE (UNII: 918X1OUF1E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10210-0035-0 5 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 11/05/2020 Part 5 of 5 CARMEX DAILY CARE MOISTURIZING LIP BALM PEACH MANGO SPF 15
octinoxate, oxybenzone salveProduct Information Item Code (Source) NDC:10210-0036 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 40 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) LANOLIN (UNII: 7EV65EAW6H) CETYL ESTERS WAX (UNII: D072FFP9GU) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) ISOHEXADECANE (UNII: 918X1OUF1E) YELLOW WAX (UNII: 2ZA36H0S2V) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10210-0036-0 5 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 11/05/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 11/05/2020 Labeler - Carma Laboratories, Inc. (006090153)