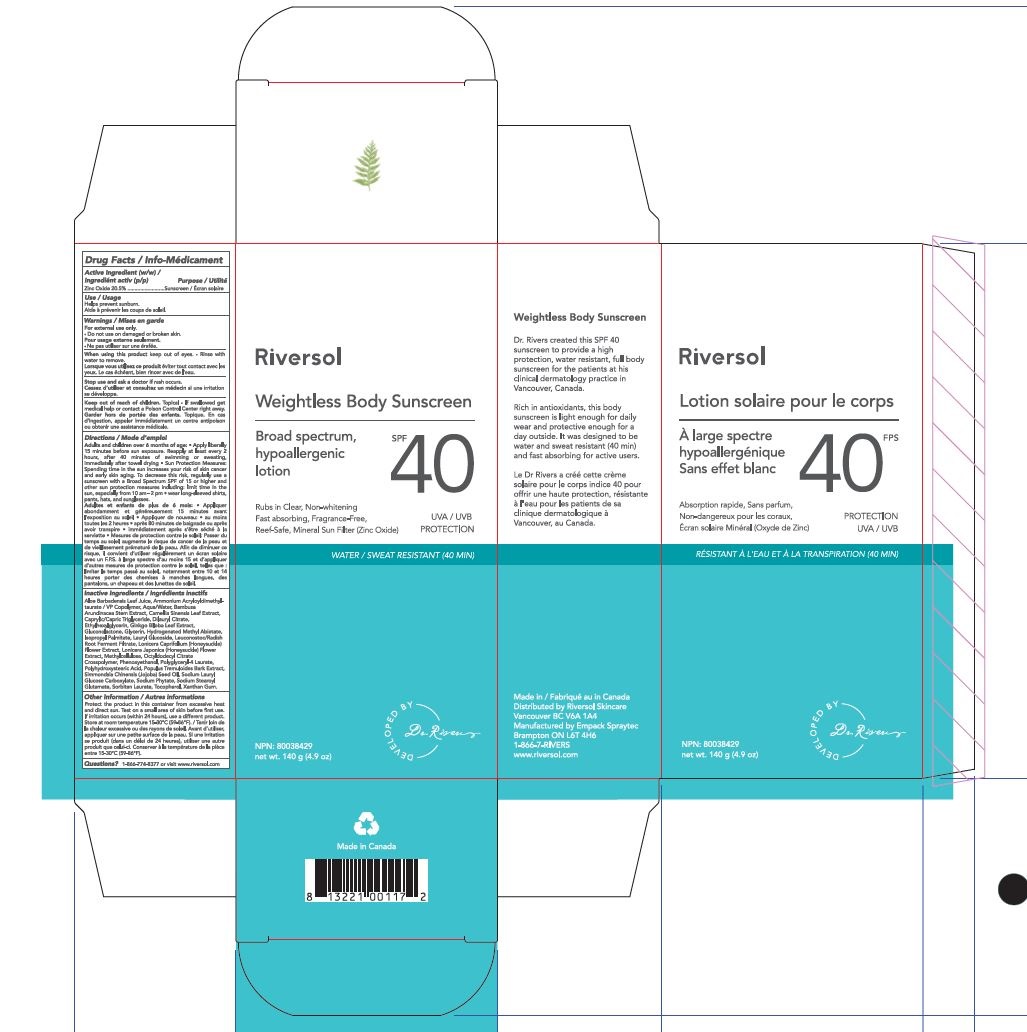
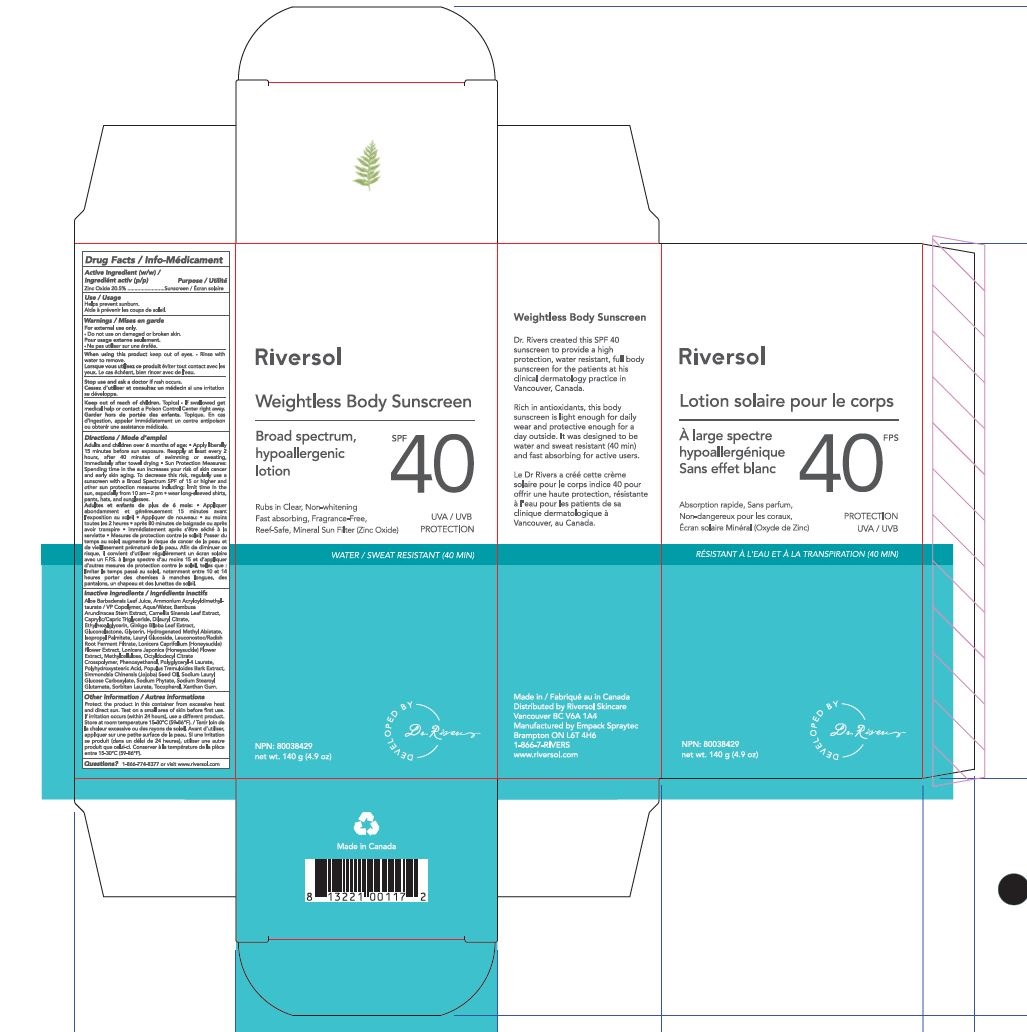
Label: RIVERSOL SPF 40 WEIGHTLESS BODY SUNSCREEN- weightless body sunscreen cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 72019-0004-1 - Packager: Riversol Skincare Solutions Inc
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 30, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
-
WHEN USING
Apply liberally to face and body 15 minutes before sun exposure.Reapply after 40 minutes of swimming or sweatingimmediately after towel dryingat least every 2 hours.Children under 6 months: ask a doctor.Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF 15 value or higher along with other sun protection measures including: limit time in the sun, especially from 10 a.m. – 2 p.m.wear long sleeved shirts, pants, hats and sunglasses.
- PRECAUTIONS
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
RIVERSOL SPF 40 WEIGHTLESS BODY SUNSCREEN
weightless body sunscreen creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72019-0004 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 20.5 g in 100 g Inactive Ingredients Ingredient Name Strength AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) METHYLCELLULOSE (100 CPS) (UNII: 4GFU244C4J) XANTHAN GUM (UNII: TTV12P4NEE) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) PHYTATE SODIUM (UNII: 88496G1ERL) LONICERA CAPRIFOLIUM FLOWER (UNII: 5N1WD9784U) GINKGO BILOBA WHOLE (UNII: 660486U6OI) CAMELLIA SINENSIS WHOLE (UNII: C5M4585ZBZ) BAMBUSA ARUNDINACEA STEM (UNII: NRA4497HC5) POLYGLYCERYL-4 OLEATE (UNII: 15B05TY4GX) GLUCONOLACTONE (UNII: WQ29KQ9POT) HYDROGENATED METHYL ABIETATE (UNII: A23O709X8O) SORBITAN MONOLAURATE (UNII: 6W9PS8B71J) JOJOBA OIL (UNII: 724GKU717M) LAURYL GLUCOSIDE (UNII: 76LN7P7UCU) LONICERA JAPONICA FLOWER (UNII: 4465L2WS4Y) POPULUS TREMULOIDES BARK (UNII: 5543O0CEID) TOCOPHEROL (UNII: R0ZB2556P8) OCTYLDODECYL LACTATE (UNII: 8JKW8M5W35) SODIUM STEAROYL GLUTAMATE (UNII: 65A9F4P024) PHENOXYETHANOL (UNII: HIE492ZZ3T) SODIUM LAURYL GLYCOL CARBOXYLATE (UNII: 8L0472VMYL) SOYBEAN OIL (UNII: 241ATL177A) WATER (UNII: 059QF0KO0R) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) LEUCONOSTOC/RADISH ROOT FERMENT FILTRATE (UNII: D2QHA03458) GLYCERIN (UNII: PDC6A3C0OX) DILAURYL CITRATE (UNII: 5OYS3G306B) ALOE VERA LEAF (UNII: ZY81Z83H0X) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72019-0004-1 1 in 1 BOX 06/01/2020 1 140 g in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 06/01/2020 Labeler - Riversol Skincare Solutions Inc (203514773)