Label: KROGER ULTRA STRENGTH ASSORTED FRUIT- calcium carbonate tablet, chewable
- NDC Code(s): 30142-307-72
- Packager: KROGER COMPANY
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated July 29, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
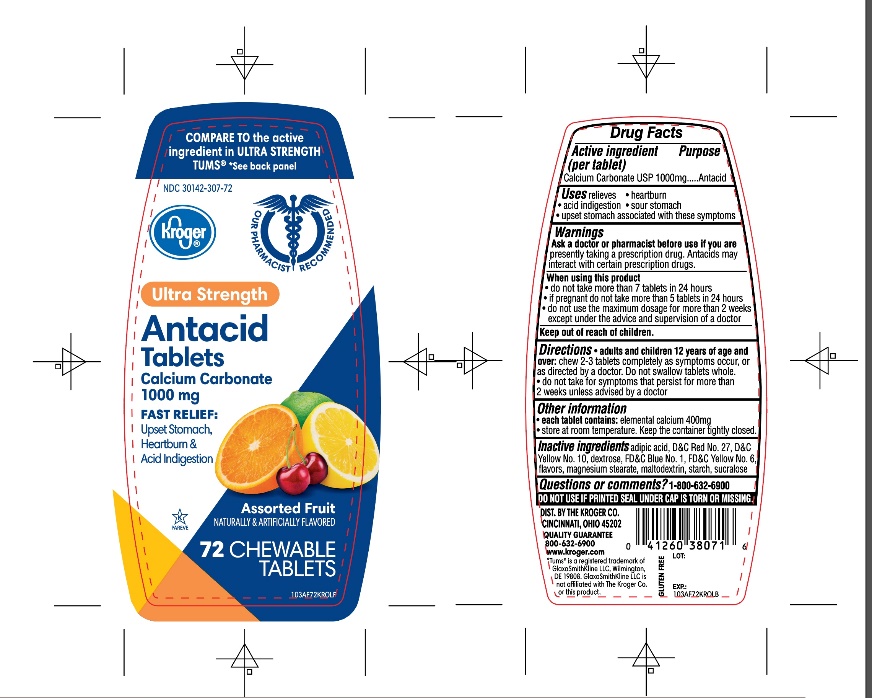
- Active ingredient (per tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor or pharmacist before use if you arepresently taking a prescription drug. Antacids may interact with certain prescription drugs.
When using this product
- do not take more than 7 tablets in 24 hours
- if pregnant do not take more than 5 tablets in 24 hours
- do not use the maximum dosage for more than 2 weeks except under the advice and supervision of a doctor.
- Keep out of reach of children.
- Directions
- Other information
- Inactive ingredients
-
Package/Label Principal Display Panel
NDC 30142-307-72
Kroger ®
FAST RELIEF OF UPSET STOMACH, HEARTBURN AND ACID INDIGESTION
COMPARE TO the active ingredient in TUMS ®ULTRA STRENGTH
See back panel
ULTRA STRENGTH
Antacid Tablets
Antacid/Calcium Supplement
CALCIUM CARBONATE 1000 mg
Assorted Fruit Flavor
Naturally and Artificially Flavored
72 CHEWABLE TABLETS
K PAREVE
Our Pharmacists Recommend
GLUTEN- FREE
DISTRIBUTED BY:
THE KROGER CO. CINCINNATI, OHIO 45202
1800-632-6900
www.kroger.com
*Tums® is registered trademark of GlaxoSmithKline LLC, GlaxoSmithKline LLC is not affiliated with The Kroger Co. or this product.
-
INGREDIENTS AND APPEARANCE
KROGER ULTRA STRENGTH ASSORTED FRUIT
calcium carbonate tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:30142-307 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CARBONATE (UNII: H0G9379FGK) (CARBONATE ION - UNII:7UJQ5OPE7D, CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM CARBONATE 1000 mg Inactive Ingredients Ingredient Name Strength ADIPIC ACID (UNII: 76A0JE0FKJ) D&C RED NO. 27 (UNII: 2LRS185U6K) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) DEXTROSE, UNSPECIFIED FORM (UNII: IY9XDZ35W2) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) STARCH, CORN (UNII: O8232NY3SJ) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color red, green, yellow, orange Score no score Shape ROUND Size 19mm Flavor FRUIT (assorted fruit) Imprint Code RP103 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:30142-307-72 72 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 05/21/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M001 05/21/2019 Labeler - KROGER COMPANY (006999528)