Label: ACNE TREATMENT- benzoyl peroxide lotion
- NDC Code(s): 67226-2050-3
- Packager: Vivier Pharma, Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 27, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
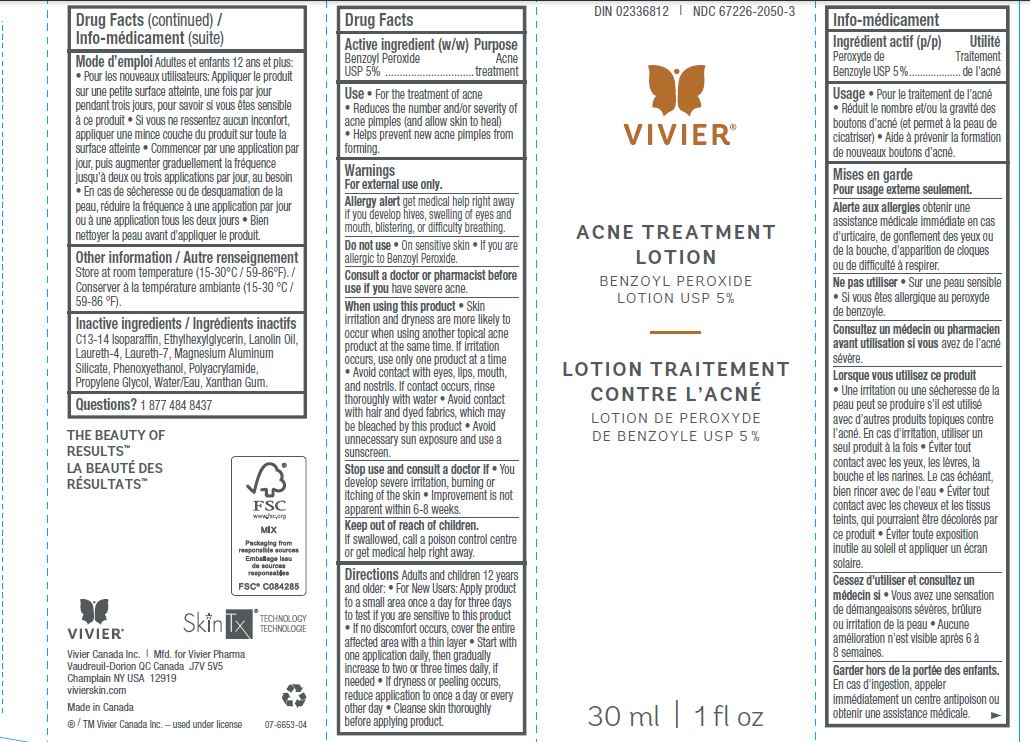
- SkinTx Acne Treatment Lotion Benzoyl Peroxide Lotion USP 5%
- Active Ingredient
- Purpose
- Use
- Warnings
- Ask a doctor before use if you are
- Keep out of reach of children.
- Do not use if you
-
When using this product
- avoid unnecessary sun exposure and use a sunscreen
- avoid contact with eyes, lips, and mouth
- avoid contact with hair and dyed fabrics, which may be bleached by this product
- skin irritation may occur, characterized by redness, burning, itching, peeling, or possibly swelling. Irritation may be reduced by using the product less frequently or in a lower oncentration.
- Stop use and ask a doctor if
-
Directions
- Wash hands with a non-medicated soap (contains no Benzoyl Peroxide).
- Cleanse and dry face thoroughly 5 minutes before use.
- Using fingertips apply evenly to affected areas.
- Use both morning and night or as directed by a physician.
- If dryness or peeling occurs, reduce application to once a day or every other day.
- Fair- skinned individuals should begin with one application.
- For more severe cases, and/or if substantial improvement is not apparent within 3 to 4 weeks, consult a health care practitioner.
- Inactive Ingredients
- Other Information
- SkinTx Acne Treatment Lotion Benzoyl Peroxide Lotion USP 5%
-
INGREDIENTS AND APPEARANCE
ACNE TREATMENT
benzoyl peroxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:67226-2050 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOYL PEROXIDE (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) BENZOYL PEROXIDE 50 mg in 1 mL Inactive Ingredients Ingredient Name Strength C13-14 ISOPARAFFIN (UNII: E4F12ROE70) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) LANOLIN OIL (UNII: OVV5IIJ58F) LAURETH-4 (UNII: 6HQ855798J) LAURETH-7 (UNII: Z95S6G8201) MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) PHENOXYETHANOL (UNII: HIE492ZZ3T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) XANTHAN GUM (UNII: TTV12P4NEE) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67226-2050-3 1 in 1 BOX 11/01/2011 1 30 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 11/01/2011 Labeler - Vivier Pharma, Inc. (250996550) Establishment Name Address ID/FEI Business Operations Dermolab Pharma Ltd 245414743 manufacture(67226-2050)