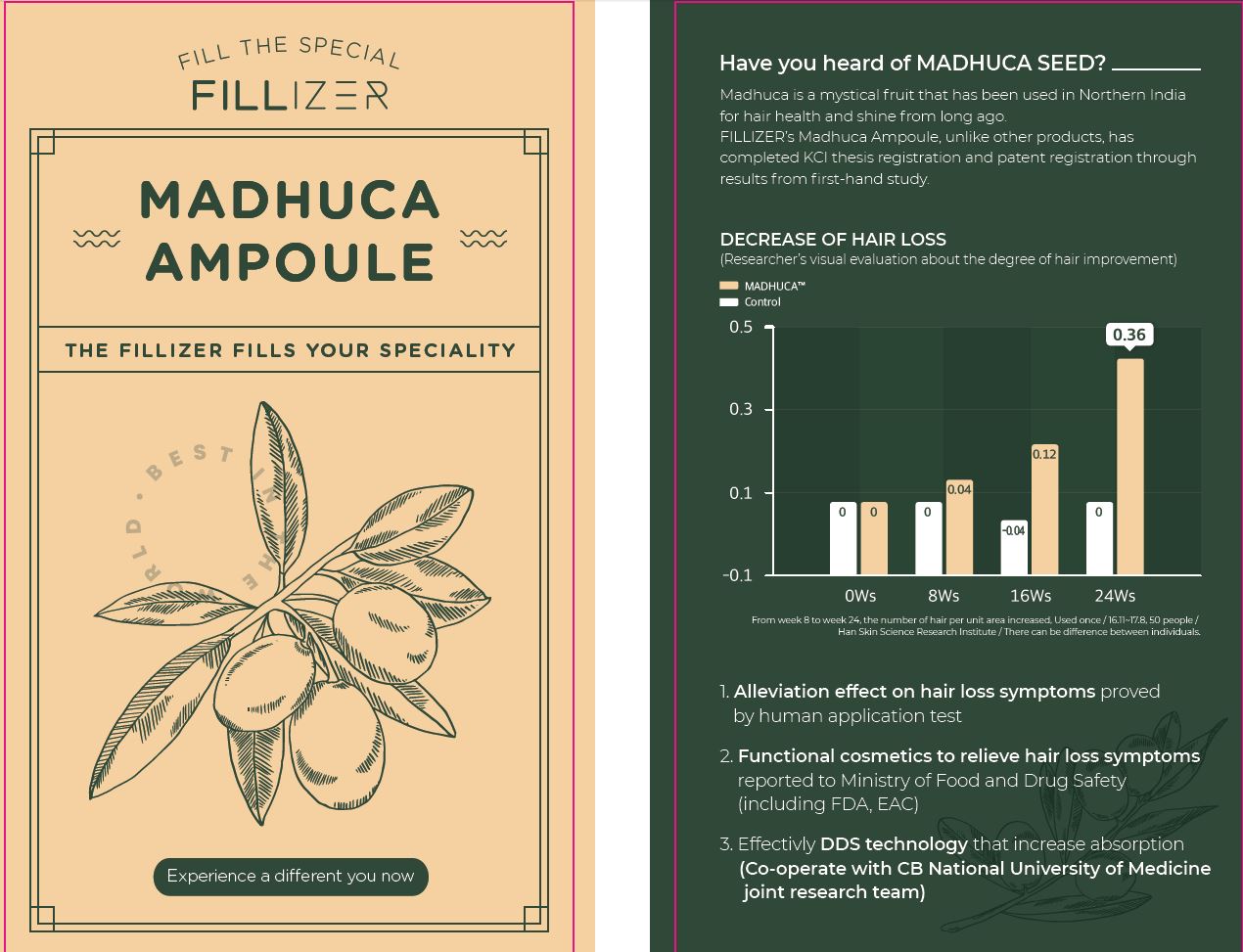
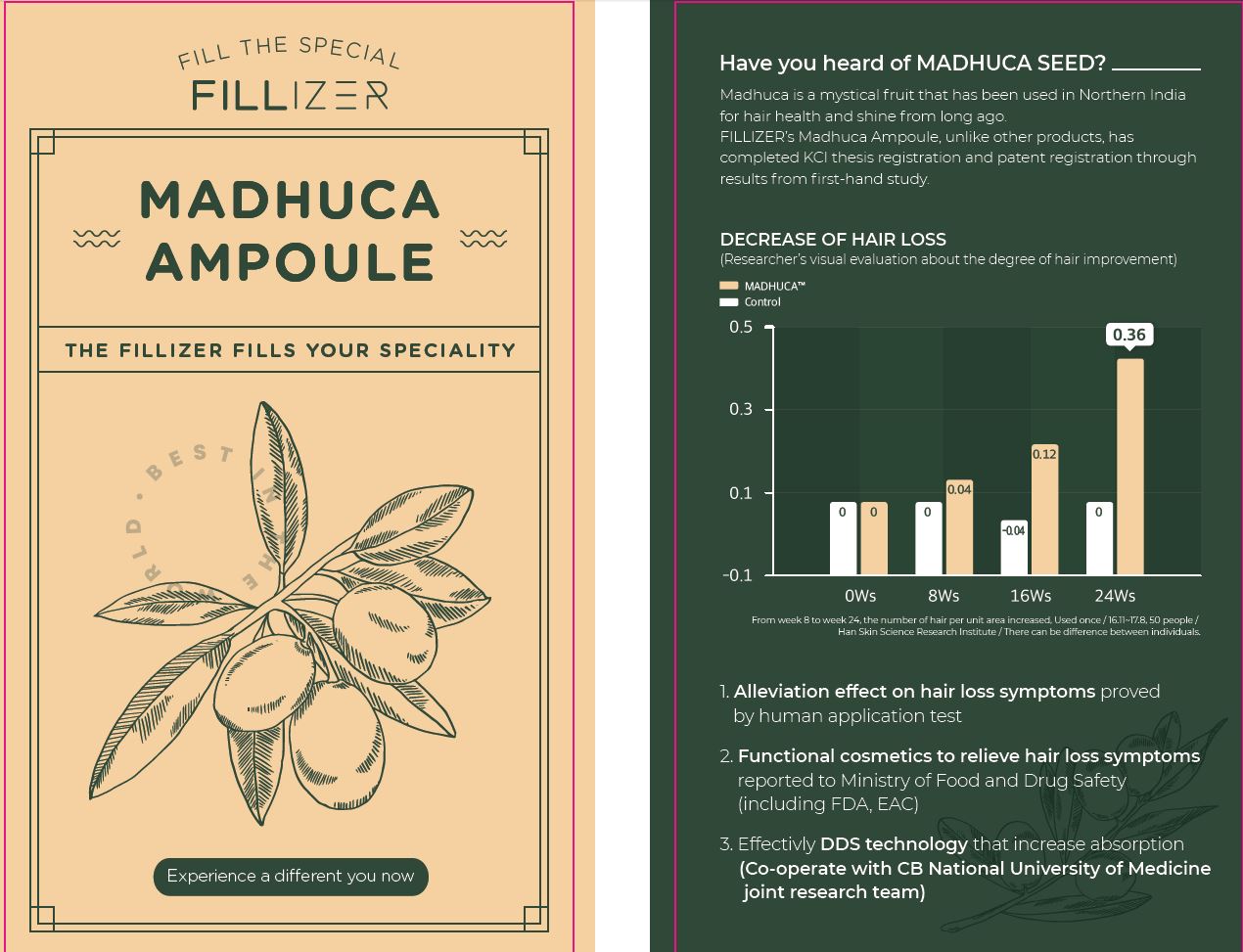
Label: FILLIZER MADHUCA AMPOULE- salicylic acid, tocopheryl acetate, niacinamide liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 81069-0001-1 - Packager: CJ ENM Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated November 13, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
- DOSAGE & ADMINISTRATION
-
INACTIVE INGREDIENT
Water
Alcohol
Polysorbate 80
Benzyl Glycol
Ammonium Acryloyldimethyltaurate/VP Copolymer
Avena Sativa (Oat) Leaf Extract
Trideceth-10
Salicylic Acid
Butylene Glycol
Madhuca Longifolia Sophorolipids(0.8%)
Fragrance
Hydrolyzed Collagen
Citrus Unshiu Peel Extract
Dipotassium Glycyrrhizate
Sodium Citrate
Ethylhexylglycerin
Pyridoxine HCl
1,2-Hexanediol
Raspberry Ketone
Menthol
Althaea Rosea Flower Extract
Coffea Arabica (Coffee) Seed Extract
Camellia Sinensis Leaf Extract
Morus Alba Bark Extract
Perilla Frutescens Leaf Extract
Houttuynia Cordata Extract
Polygonum Multiflorum Root Extract
Limonene
Geraniol
Citronellol
Butylphenyl Methylpropional
Linalool
Alpha-Isomethyl Ionone
Hydroxycitronellal
Amyl Cinnamal
Benzyl Benzoate
Citral - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FILLIZER MADHUCA AMPOULE
salicylic acid, tocopheryl acetate, niacinamide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81069-0001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) (.ALPHA.-TOCOPHEROL - UNII:H4N855PNZ1) .ALPHA.-TOCOPHEROL ACETATE 2 g in 100 mL SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 0.495 g in 100 mL NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 0.3 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81069-0001-1 7.5 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/14/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 11/14/2020 Labeler - CJ ENM Co., Ltd. (687982736) Registrant - CJ ENM Co., Ltd. (687982736) Establishment Name Address ID/FEI Business Operations CJ ENM Co., Ltd. 687982736 manufacture(81069-0001) , label(81069-0001)