Label: JIKIMI PHYTONCIDE- titanium dioxide, hypochlorous acid liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 71544-0020-1 - Packager: MY Corp.,Ltd
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated November 13, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
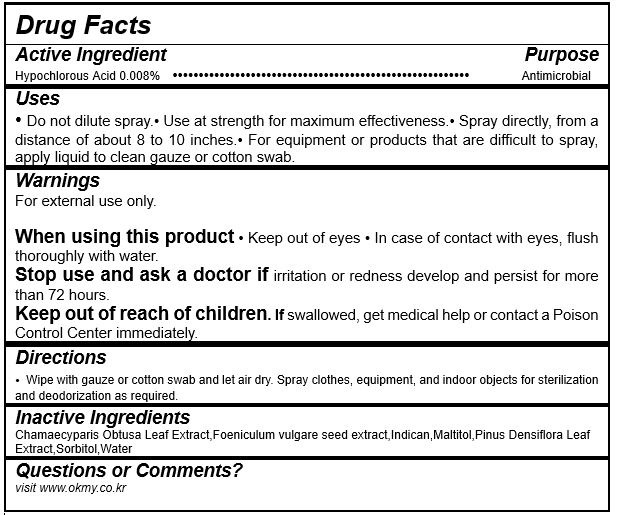
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
WARNINGS
For external use only.
When using this product
- Keep out of eyes
- In case of contact with eyes, flush thoroughly with water.
Stop use and ask a doctor if irritation or redness develop and persist for more than 72 hours.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center immediately.
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
JIKIMI PHYTONCIDE
titanium dioxide, hypochlorous acid liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71544-0020 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYPOCHLOROUS ACID (UNII: 712K4CDC10) (HYPOCHLOROUS ACID - UNII:712K4CDC10) HYPOCHLOROUS ACID 0.00136 g in 17 mL Inactive Ingredients Ingredient Name Strength CHAMAECYPARIS OBTUSA LEAF (UNII: 7OL154J5XB) INDICAN (UNII: N187WK1Y1J) MALTITOL (UNII: D65DG142WK) PINUS DENSIFLORA LEAF (UNII: Q1Q9P50WIY) SORBITOL (UNII: 506T60A25R) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71544-0020-1 17 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 11/09/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 07/15/2020 Labeler - MY Corp.,Ltd (688202781) Registrant - MY Corp.,Ltd (688202781) Establishment Name Address ID/FEI Business Operations MY Corp.,Ltd 688202781 manufacture(71544-0020) , label(71544-0020) , pack(71544-0020)