Label: COLGATE ANTICAVITY- sodium monofluorophosphate paste
- NDC Code(s): 69491-033-01
- Packager: Buzz Export Services Pty., Ltd.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 10, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
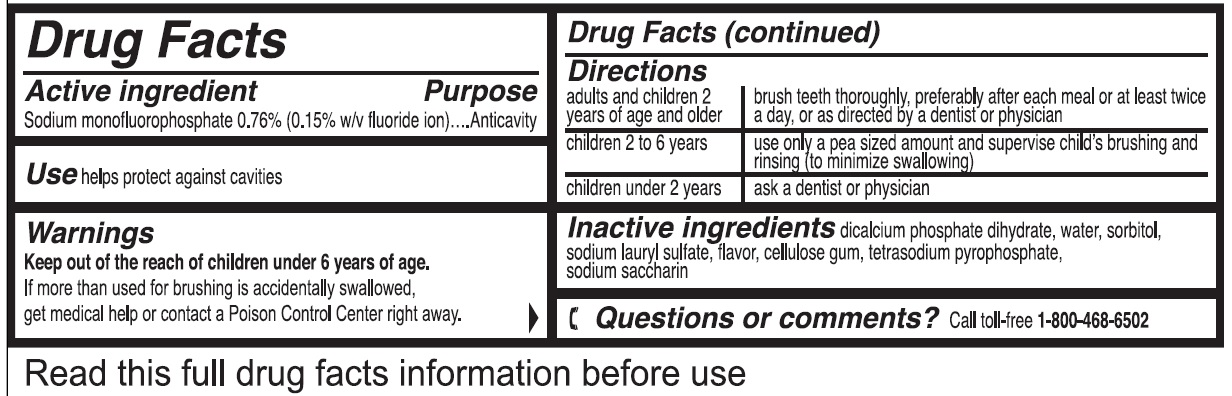
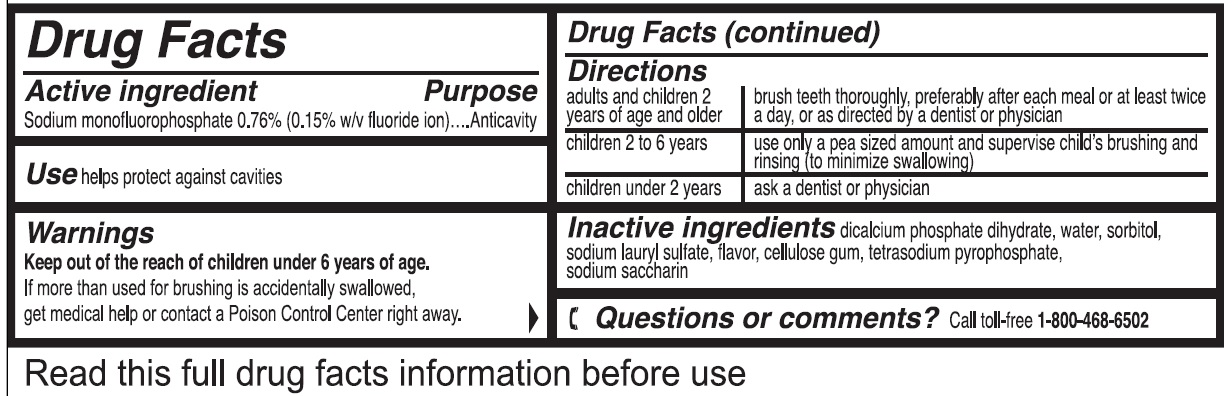
- Drug Facts
- Active ingredient
- Use
- Warnings
-
Directions
adults and children 2 years of age and older brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or physician children 2 to 6 years use only a pea sized amount and supervise child's brushing and rinsing (to minimize swallowing) children under 2 years ask a dentist or physician - Inactive ingredients
- Questions or comments?
- Package Labeling:69491-033-01
-
INGREDIENTS AND APPEARANCE
COLGATE ANTICAVITY
sodium monofluorophosphate pasteProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69491-033 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM MONOFLUOROPHOSPHATE (UNII: C810JCZ56Q) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 7.6 mg in 1 g Inactive Ingredients Ingredient Name Strength DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) SODIUM LAURYL SULFATE (UNII: 368GB5141J) METHYL SALICYLATE (UNII: LAV5U5022Y) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED (UNII: K679OBS311) SODIUM PYROPHOSPHATE (UNII: O352864B8Z) SACCHARIN SODIUM (UNII: SB8ZUX40TY) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69491-033-01 10 g in 1 TUBE; Type 0: Not a Combination Product 10/15/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part355 10/15/2020 Labeler - Buzz Export Services Pty., Ltd. (747317902)