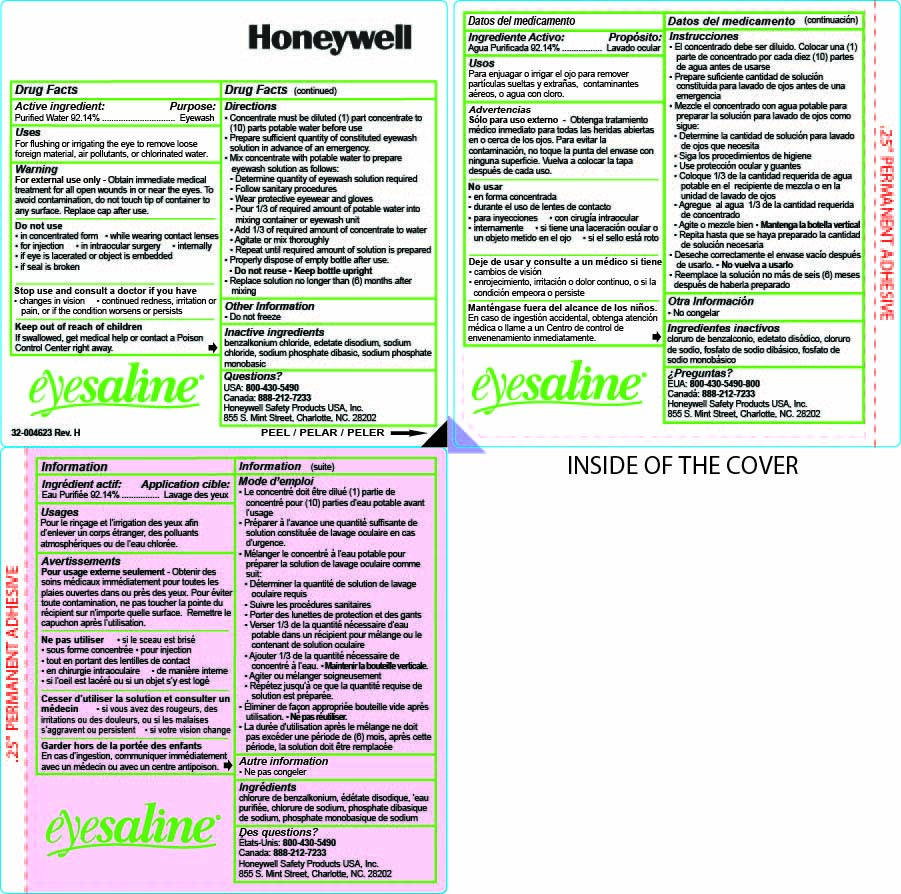
Label: EYESALINE CONCENTRATE- water liquid
- NDC Code(s): 0498-0601-18, 0498-0601-19
- Packager: Honeywell Safety Products USA, Inc
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated May 27, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
-
Warnings
For external use only- Obtain immidiate medical treatment for all open wounds in or near the eyes.
Do not use
- in concentrated form
- while wearing contact lenses
- if solution changes color or becomes cloudy
- for injection
- in intraocular surgery
- internally
- if eye is lacerated or object is embedded
- if seal is broken
-
Directions
- Concentrate must be diluted (1) part concentrate to (10) parts potable water before use
- Prepare sufficient quantity of constituted eyewash solution in advance of an emergency
- Mix concentrate with potable water to prepare eyewash solution as follows:
- Determine quantity of eyewash solution required
- Follow sanitary procedures
- Wear protective eyewear and gloves
- Pour 1/3 of required amount of potable water into mixing container or eyewash unit
- Add 1/3 of required amount of concentrate into water
- Agitate or mix thoroughly
- Repeat until required amount of solution is prepared
- Properly dispose of empty bottle after use
Do not reuse.
- Replace solution no longer than (6) months after mixing
- Other information
- Inactive ingredients
- Questions?
- Package Label
-
INGREDIENTS AND APPEARANCE
EYESALINE CONCENTRATE
water liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0498-0601 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 92.2 L in 100 L Inactive Ingredients Ingredient Name Strength SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) SODIUM CHLORIDE (UNII: 451W47IQ8X) EDETATE DISODIUM (UNII: 7FLD91C86K) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0601-18 2.07 L in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/01/2020 2 NDC:0498-0601-19 5.32 L in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 09/01/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M018 09/01/2020 Labeler - Honeywell Safety Products USA, Inc (118768815) Establishment Name Address ID/FEI Business Operations Keystone Industries 014769301 manufacture(0498-0601)