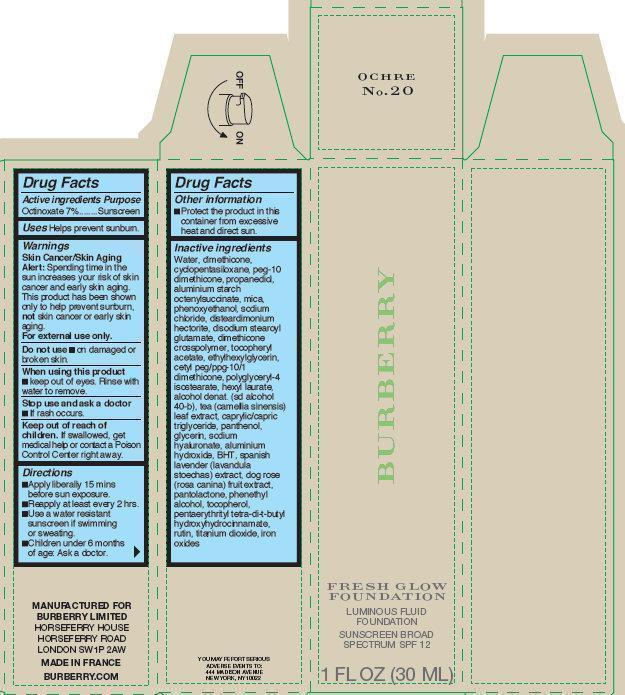
Label: BURBERRY FRESH GLOW FOUNDATION LUMINOUS FLUID OCHRE NO. 20- octinoxate cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 69038-012-00, 69038-012-01 - Packager: Burberry Limited
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 18, 2019
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug facts
- Active Ingredients
- Uses
- Warnings
- Directions
- Other information
-
Inactive ingredients
water, dimethicone, cyclopentasiloxane, peg-10 dimethicone, propanediol, aluminium starch octenylsuccinate, mica, phenoxyethanol, sodium chloride, disteardimonium hectorite, disodium stearoyl glutamate, dimethicone crosspolymer, tocopheryl acetate, ethylhexylglycerin, cetyl peg/ppg-10/1 dimethicone, polyglyceryl-4 isostearate, hexyl laurate, alcohol denat. (sd alcohol 40-b), tea (camellia sinensis) leaf extract, caprylic/capric triglyceride, panthenol, glycerin, sodium hyaluronate, aluminium hydroxide, BHT, spanish lavender (lavandula stoechas) extract, dog rose (rosa canina) fruit extract, pantolactone, phenethyl alcohol, tocopherol, pantaerythrityl tetra-di-t-butyl hydroxyhydrocinnamate, rutin, titanium dioxide, iron oxides
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL
- Product Label
-
INGREDIENTS AND APPEARANCE
BURBERRY FRESH GLOW FOUNDATION LUMINOUS FLUID OCHRE NO. 20
octinoxate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69038-012 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 70 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) DIMETHICONE (UNII: 92RU3N3Y1O) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) PROPANEDIOL (UNII: 5965N8W85T) ALUMINUM STARCH OCTENYLSUCCINATE (UNII: I9PJ0O6294) MICA (UNII: V8A1AW0880) PHENOXYETHANOL (UNII: HIE492ZZ3T) SODIUM CHLORIDE (UNII: 451W47IQ8X) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) DISODIUM STEAROYL GLUTAMATE (UNII: 45ASM2L11M) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) POLYGLYCERYL-4 ISOSTEARATE (UNII: 820DPX33S7) HEXYL LAURATE (UNII: 4CG9F9W01Q) TEA LEAF (UNII: GH42T47V24) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) PANTHENOL (UNII: WV9CM0O67Z) GLYCERIN (UNII: PDC6A3C0OX) HYALURONATE SODIUM (UNII: YSE9PPT4TH) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) ROSA CANINA FRUIT (UNII: 3TNW8D08V3) PANTOLACTONE, (R)- (UNII: J288D7O0JS) PHENYLETHYL ALCOHOL (UNII: ML9LGA7468) TOCOPHEROL (UNII: R0ZB2556P8) PENTAERYTHRITOL TETRAKIS(3-(3,5-DI-TERT-BUTYL-4-HYDROXYPHENYL)PROPIONATE) (UNII: 255PIF62MS) RUTIN (UNII: 5G06TVY3R7) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69038-012-00 1 in 1 CARTON 11/14/2014 1 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:69038-012-01 3 in 1 BOX 11/14/2014 2 1 in 1 CARTON 2 30 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 11/14/2014 Labeler - Burberry Limited (210047627)