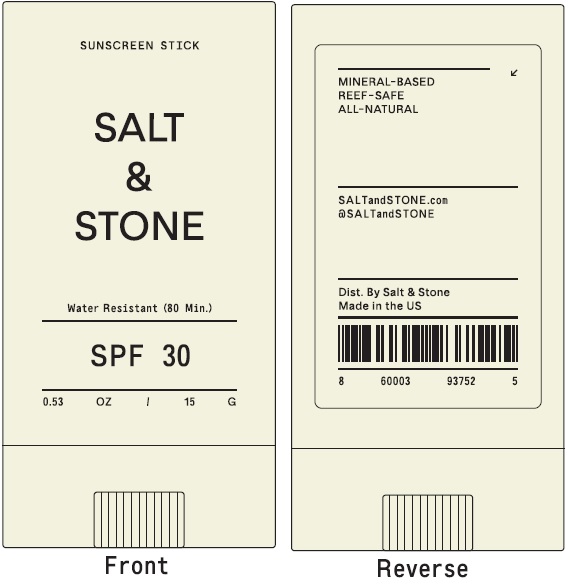
Label: SALT AND STONE SPF 30 SUNSCREEN STICK- zinc oxide stick
- NDC Code(s): 71585-141-00
- Packager: Salt and Stone
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 28, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DRUG FACTS
- Active Ingredients
- Uses
- Warnings
-
Directions
- Apply liberally 15 minutes before sun exposure.
- Reapply:
- After 80 minutes of swimming or sweating,
- Immediately after towel drying.
- At least every two (2) hours.
- Spending time in the sun increases the risk of skin cancer and early skin aging. To decrease the risk, regularly use: a suscreen with a broad-spectrum measures including: Sun Protection Measures
- Limiting time in the sun, especially from 10AM - 2PM.
- Wear long-sleeved shirts, pants, hats, and sunglasses.
- Children under 6 months: Ask a doctor.
- Other Information
- Inactive Ingredients
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
SALT AND STONE SPF 30 SUNSCREEN STICK
zinc oxide stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71585-141 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 225 mg in 1 g Inactive Ingredients Ingredient Name Strength SHEA BUTTER (UNII: K49155WL9Y) YELLOW WAX (UNII: 2ZA36H0S2V) CARNAUBA WAX (UNII: R12CBM0EIZ) COCONUT OIL (UNII: Q9L0O73W7L) SUNFLOWER OIL (UNII: 3W1JG795YI) MICA (UNII: V8A1AW0880) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) JOJOBA OIL (UNII: 724GKU717M) COCOA BUTTER (UNII: 512OYT1CRR) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71585-141-00 15 g in 1 TUBE; Type 0: Not a Combination Product 09/22/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 09/22/2021 Labeler - Salt and Stone (080683697)