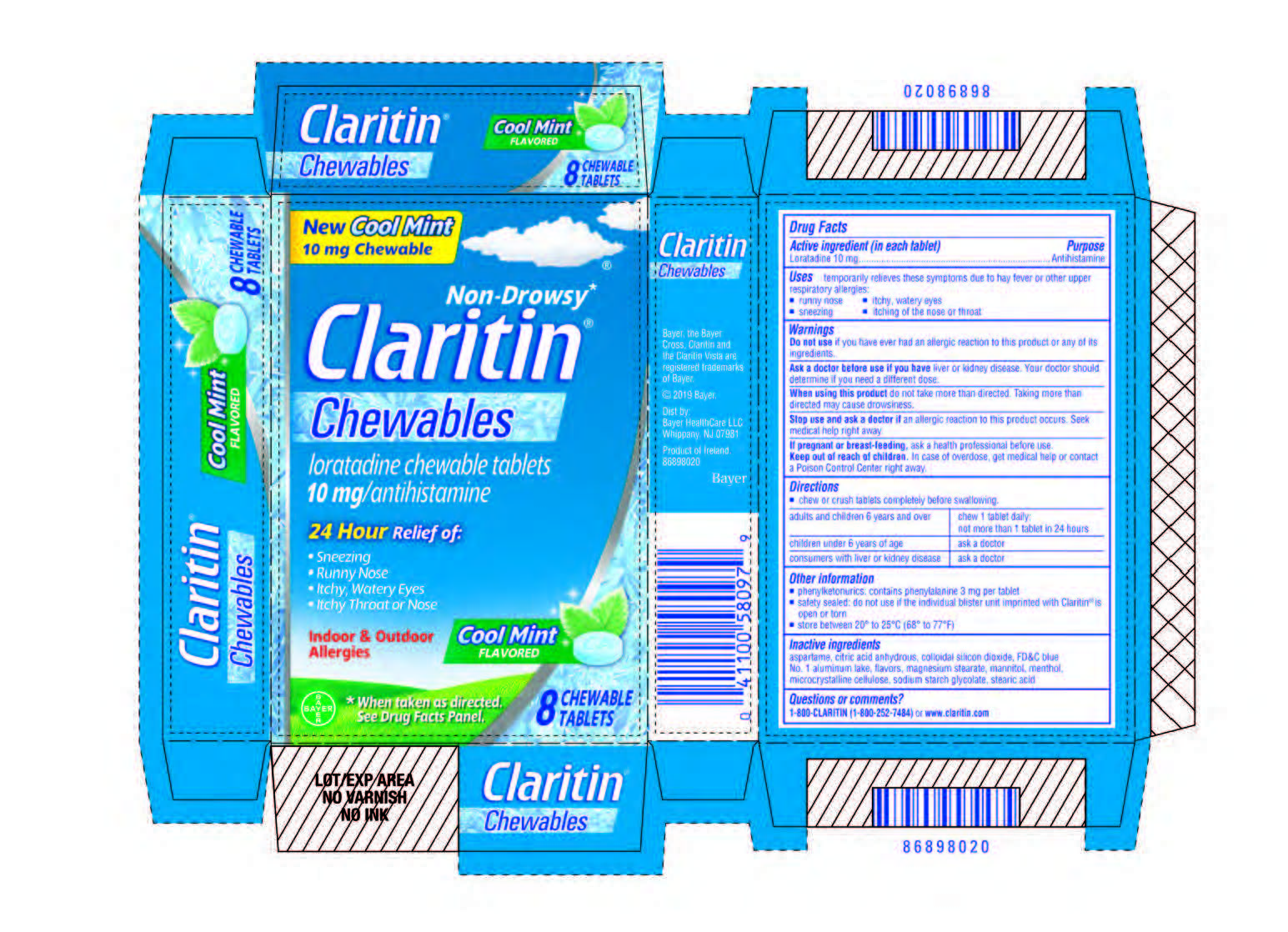
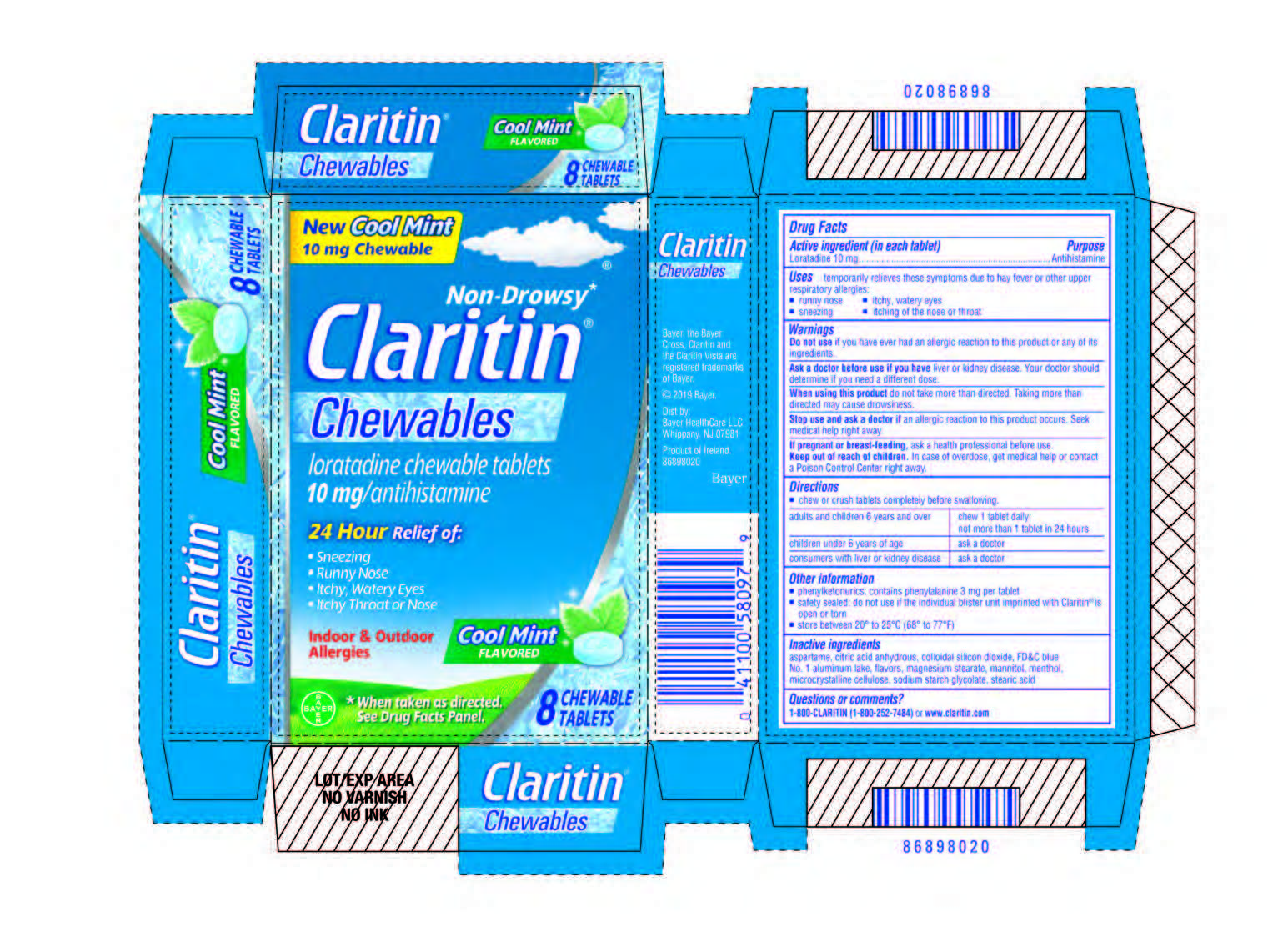
Label: CLARITIN CHEWABLE COOL MINT FLAVOR- loratadine tablet, chewable
-
NDC Code(s):
11523-4364-1,
11523-4364-2,
11523-4364-3,
11523-4364-4, view more11523-4364-5, 11523-4364-6, 11523-4364-7
- Packager: Bayer HealthCare LLC.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated December 13, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- Uses
-
WARNINGS
Warnings
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different
dose.
When using this product do not take more than directed. Taking more than directed may cause drowsiness.
- Directions
- Other information
- INACTIVE INGREDIENT
- Questions or comments
- Carton 8 count
-
INGREDIENTS AND APPEARANCE
CLARITIN CHEWABLE COOL MINT FLAVOR
loratadine tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11523-4364 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LORATADINE (UNII: 7AJO3BO7QN) (LORATADINE - UNII:7AJO3BO7QN) LORATADINE 10 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) SODIUM STARCH GLYCOLATE TYPE A CORN (UNII: AG9B65PV6B) STEARIC ACID (UNII: 4ELV7Z65AP) MAGNESIUM STEARATE (UNII: 70097M6I30) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) FD&C BLUE NO. 1--ALUMINUM LAKE (UNII: J9EQA3S2JM) MENTHOL (UNII: L7T10EIP3A) ASPARTAME (UNII: Z0H242BBR1) MANNITOL (UNII: 3OWL53L36A) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) Product Characteristics Color blue Score no score Shape ROUND Size 11mm Flavor MINT Imprint Code CA Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11523-4364-1 3 in 1 CARTON 12/09/2019 1 8 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:11523-4364-2 1 in 1 CARTON 12/09/2019 2 2 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC:11523-4364-3 1 in 1 CARTON 12/09/2019 3 4 in 1 BLISTER PACK; Type 0: Not a Combination Product 4 NDC:11523-4364-5 8 in 1 CARTON 12/09/2019 4 8 in 1 BLISTER PACK; Type 0: Not a Combination Product 5 NDC:11523-4364-6 1 in 1 CARTON 12/09/2019 5 8 in 1 BLISTER PACK; Type 0: Not a Combination Product 6 NDC:11523-4364-7 1 in 1 POUCH 12/09/2019 6 2 in 1 BLISTER PACK; Type 0: Not a Combination Product 7 NDC:11523-4364-4 7 in 1 CARTON 12/09/2019 7 8 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021891 12/09/2019 Labeler - Bayer HealthCare LLC. (112117283)