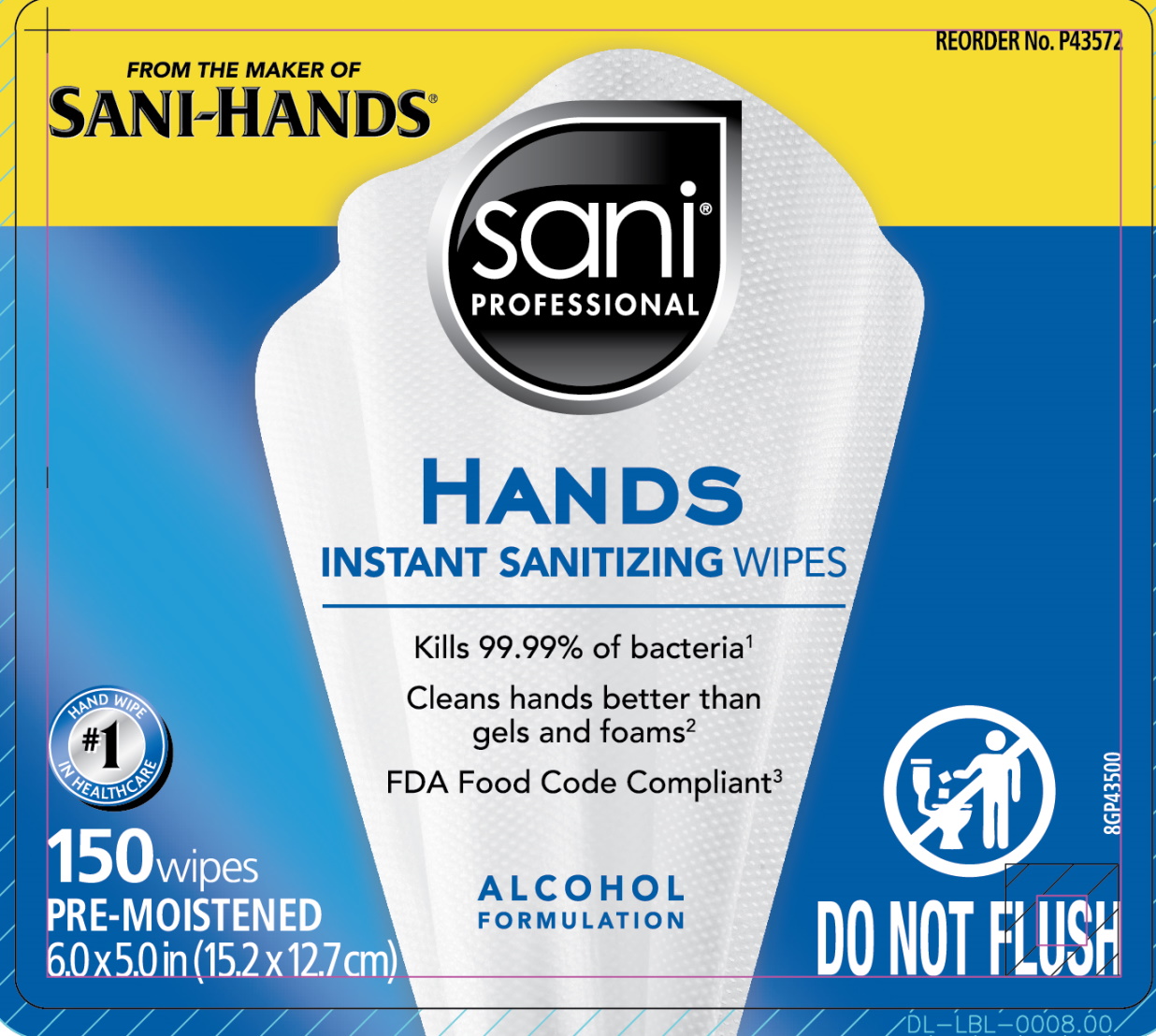
Label: SANI PROFESSIONAL HANDS INSTANT SANITIZING WIPES- alcohol cloth
- NDC Code(s): 10819-6006-1, 10819-6007-1, 10819-6008-1
- Packager: Professional Disposables International, Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 21, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
-
Warnings
For external use only
Flammable. Keep away from fire or flame
When using this product avoid contact with eyes. If contact occurs, rinse eyes thoroughly with water.
Do not use in or contact with eyes
Discontinue use if irritation and redness develop. If condition persists for more than 72 hours, consult a doctor.
- Keep out of reach of children
-
Directions
Canister:
to dispense, lift cover, remove seal, pull center sheet from roll, twist to a point, feed through dispenser hole in cover. Keep cap closed to prevent moisture loss.
use entire wipe to thoroughly wipe hands, fingers, and wrists
for dirty hands, use first wipe to clean hands, then discard wipe; sanitize with second wipe
discard after single use
Packet:
open packet
use entire wipe to thoroughly wipe hands, fingers, and wrists
to sanitize, use multiple wipes
discard after single use
- Other information
- Inactive ingredients
- Principal display panel
-
INGREDIENTS AND APPEARANCE
SANI PROFESSIONAL HANDS INSTANT SANITIZING WIPES
alcohol clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10819-6006 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) ALOE VERA LEAF (UNII: ZY81Z83H0X) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10819-6006-1 3.23 mL in 1 PACKET; Type 0: Not a Combination Product 03/01/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 03/01/2020 SANI PROFESSIONAL HANDS INSTANT SANITIZING WIPES
alcohol clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10819-6007 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) WATER (UNII: 059QF0KO0R) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) GLYCERIN (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10819-6007-1 484 mL in 1 CANISTER; Type 0: Not a Combination Product 03/01/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 03/01/2020 SANI PROFESSIONAL HANDS INSTANT SANITIZING WIPES
alcohol clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10819-6008 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) WATER (UNII: 059QF0KO0R) ALOE VERA LEAF (UNII: ZY81Z83H0X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) GLYCERIN (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10819-6008-1 805.44 mL in 1 CANISTER; Type 0: Not a Combination Product 01/01/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 01/01/2020 Labeler - Professional Disposables International, Inc. (800777117) Establishment Name Address ID/FEI Business Operations Professional Disposables International, Inc. 800777117 manufacture(10819-6006, 10819-6007, 10819-6008)


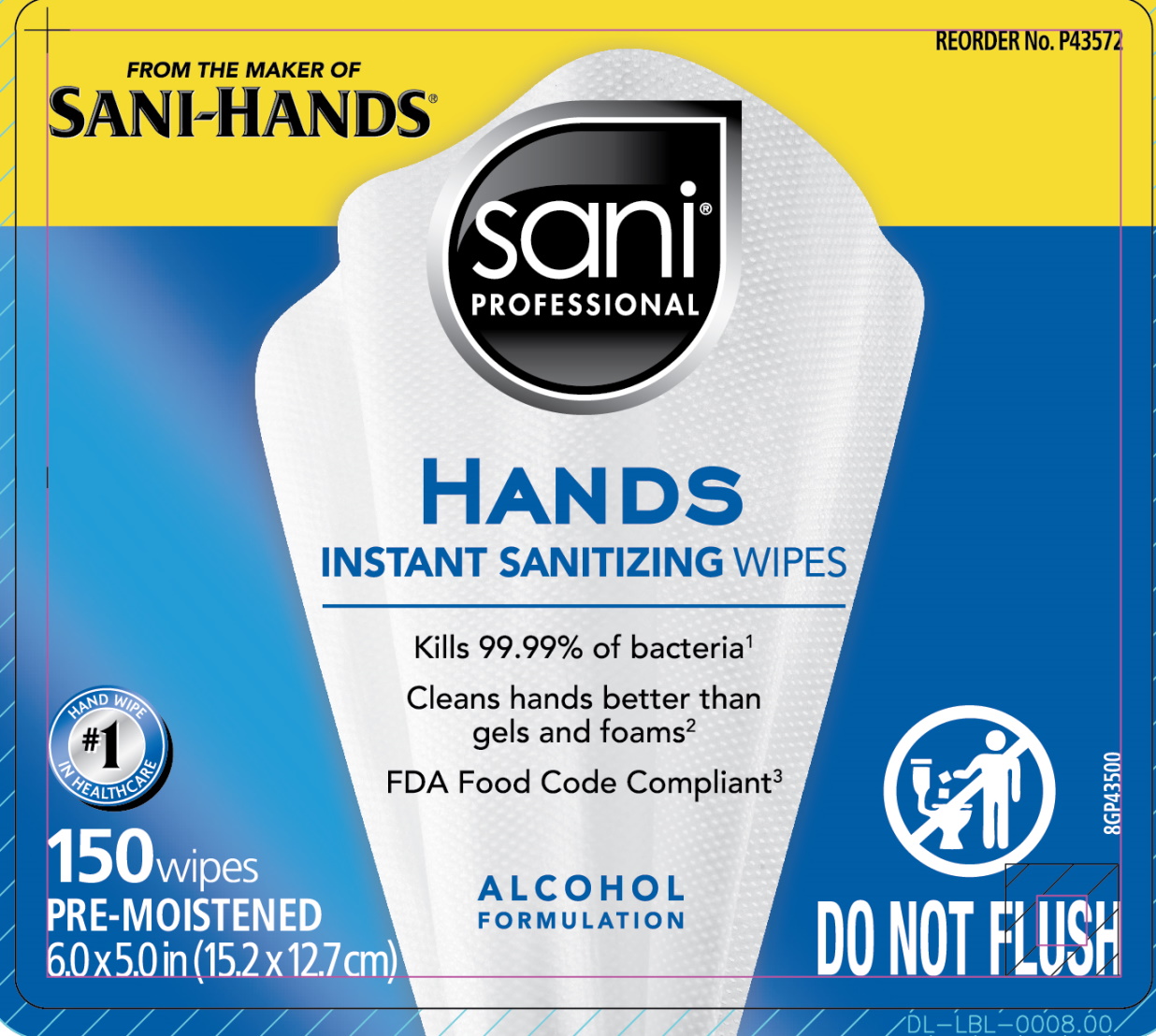



 Canister, 150ct
Canister, 150ct