Label: CHILDRENS PAIN AND FEVER- acetaminophen suspension
- NDC Code(s): 76281-526-23, 76281-526-24
- Packager: AptaPharma Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated February 2, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
Liver warning
This product contains acetaminophen. Severe liver
damage may occur nyour child takes: ■ more than 5
doses in 24 hours, which is the maximum daily amount
■ with other drugs containing acetaminophen
Allergy alert: acetaminophen may cause severe skin
reactions. Symptoms may include: ■ skin reddening
■ blisters ■ rash
If a skin reaction occurs, stop use and seek medical help
right away.
Sore throat warning
If sore throat is severe, persists for more than 2 days,
is accompanied or followed by ever, headache, rash.
nausea or vomiting, consult a doctor promptly. - DO NOT USE
- ASK DOCTOR
- ASK DOCTOR/PHARMACIST
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
OVERDOSAGE
Overdose warning:Taking more than the recommended dose (overdose)
may cause liver damage. In case of overdose, get
medical help or contact a Poison Control Center right
away. (1-800-222-1222) Quick medical attention is critical
for adults as well as for children even if you
do not notice any signs or symptoms. -
DOSAGE & ADMINISTRATION
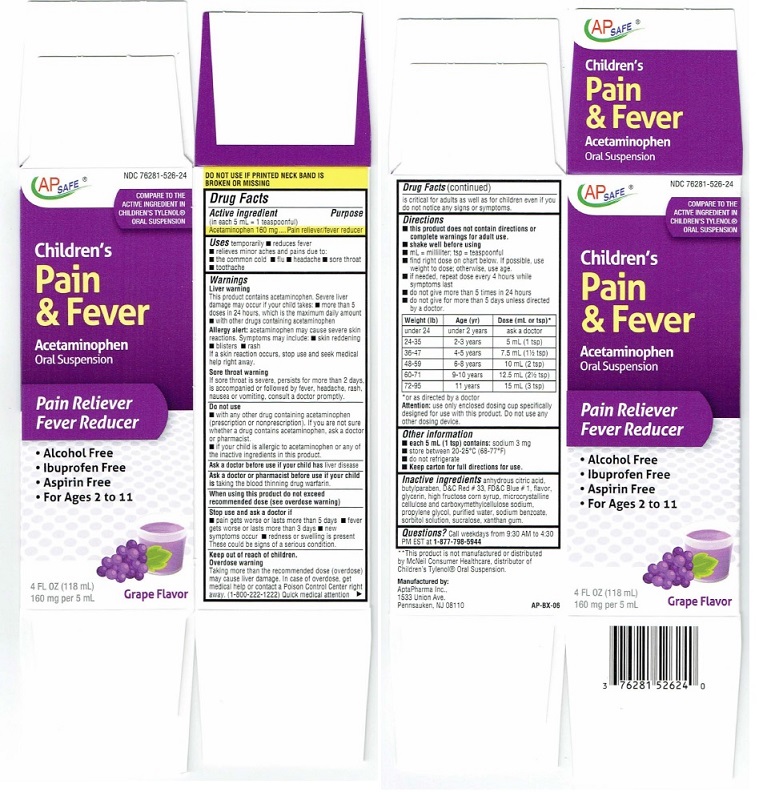
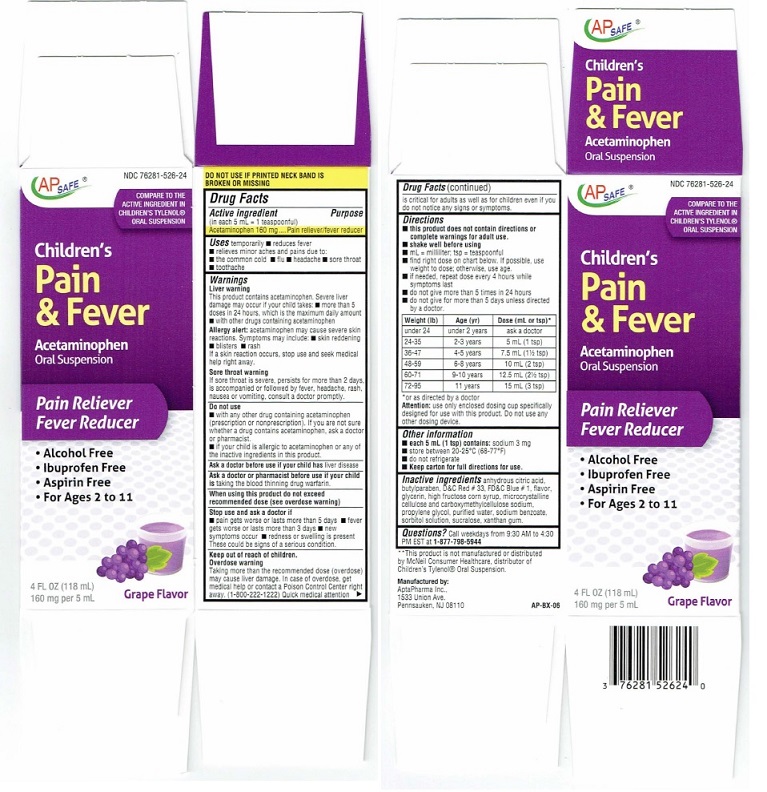
Directions (Children's Pain & Fever)
■ this product does not contain directions or complete warnings for adult use.
■ shake well before using
■ mL = milliliter; tsp = teaspoonful
■ find the right dose on the chart below. If possible, use weight to dose, otherwise, use age.
■ if needed, repeat dose every 4 hours while symptoms last
■ do not give more than 5 times in 24 hours
■ do not give for more than 5 days unless directed by a doctor.Weight (lb) Age (yr) Dose (mL or tsp)* under 24 under 2 years ask a doctor 24-35 2 - 3 years 5 mL (1 tsp) 36-47 4 - 5 years 7.5 mL (1 1/2 tsp0 48-59 6 - 8 years 10 mL (2 tsp) 60-71 9 - 10 years 12.5. mL (2 1/2 TSP) 72-95 11 years 15 mL (3 tsp) *or as directed by a doctor
Attention: use only enclosed dosing cup specifically
designed for use with this product. Do not use any
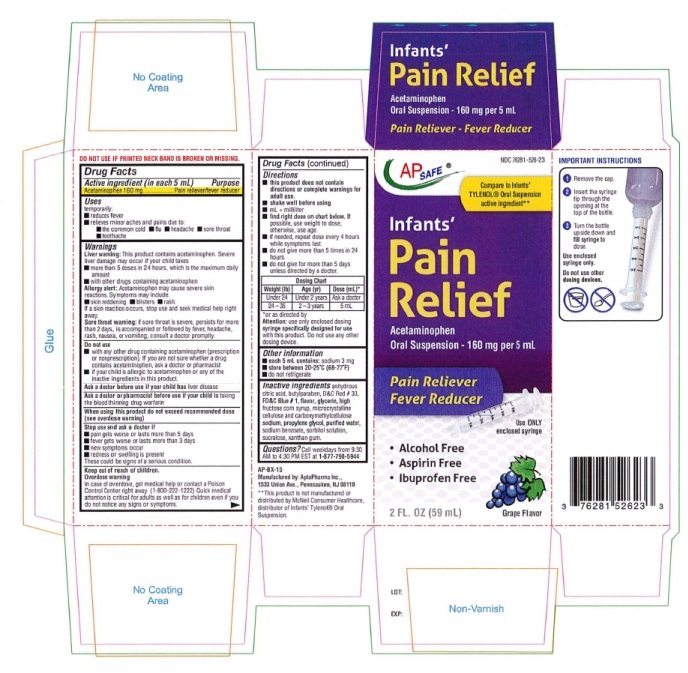
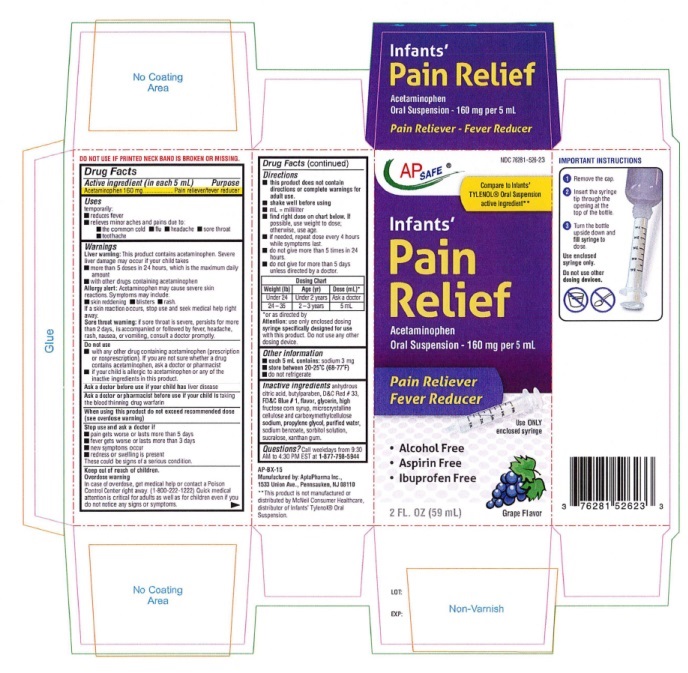
other dosing device.Directions (Infants' Pain Relief)
■ this product does not contain directions or complete warnings for adult use.
■ shake well before using
■ mL = milliliter; tsp = teaspoonful
■ find the right dose on the chart below. If possible, use weight to dose, otherwise, use age.
■ if needed, repeat dose every 4 hours while symptoms last
■ do not give more than 5 times in 24 hours
■ do not give for more than 5 days unless directed by a doctor.Weight (lb) Age (Yr) Dose (mL)* Under 24 Under 2 years Ask a doctor 24-35 2-3 years 5 mL *or as directed by a doctor
Attention: use only enclosed dosing syringe specifially
designed for use with this product. Do not use any
other dosing device. - OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
-
Product Label
AP SAFE® NDC 76821-526-24
COMPARE TO THE
ACTIVE INGREDIENT IN
CHILDREN'S TYLENOL®
ORAL SUSPENSION*
Children's
Pain
& Fever
Acetaminophen
Oral Suspension
Pain reliever/Fever Reducer
• Alcohol-Free
• Ibuprofen Free
• Aspirin Free
• For Ages 2 to 114 FL OZ (118 mL)
160 mg per 5 mL
Grape Flavor
DO NOT USE IF PRINTED NECK BAND IS
BROKEN OR MISSING**This product is not manufactured or distributed
by McNeil Consumer Healthcare, distributor of
Children's Tylenol®Oral Suspension.Manufactured by:
AptaPharma Inc.
1533 Union Ave.
Pennsauken. NJ 08110AP-BX-06
Carton
Bottle

Carton
Bottle

res
-
INGREDIENTS AND APPEARANCE
CHILDRENS PAIN AND FEVER
acetaminophen suspensionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76281-526 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 160 mg in 5 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) BUTYLPARABEN (UNII: 3QPI1U3FV8) D&C RED NO. 33 (UNII: 9DBA0SBB0L) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GLYCERIN (UNII: PDC6A3C0OX) HIGH FRUCTOSE CORN SYRUP (UNII: XY6UN3QB6S) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SORBITOL SOLUTION (UNII: 8KW3E207O2) SUCRALOSE (UNII: 96K6UQ3ZD4) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color purple Score Shape Size Flavor GRAPE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76281-526-24 1 in 1 CARTON 09/30/2020 1 118 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:76281-526-23 1 in 1 CARTON 01/30/2024 2 59 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 08/21/2013 Labeler - AptaPharma Inc. (790523323) Registrant - AptaPharma Inc. (790523323) Establishment Name Address ID/FEI Business Operations AptaPharma Inc. 790523323 manufacture(76281-526)